Listen to patient’s complaints more than ECHO
The increasing amount of electronically stored clinical data in modern health care system has heralded an era of big medical data (1). Consequently, utilizing these big data has become a major interest in performing clinical research. Big data-driven clinical research is increasingly important and it is expected to be a viable alternative for prospective randomized controlled trial (RCT) in the future (2,3). Some researchers have argued that the conclusions from the analysis of big data may be more clinically relevant than those from RCT in a real world setting because strict experimental requirements for interventional protocols and subject selections in RCT are hardly reproducible during routine clinical practice (2).
Clinical researches using big data, however, have limitations inherent to their natures of observational study (2). Evaluating the risk of procedure [e.g. coronary artery bypass grafting (CABG)] for the adverse outcomes (e.g., mortality) according to the existence of morbidity [e.g., heart failure (HF), low ejection fraction (EF)] may be affected by measured and unmeasured confounders. As big data themselves cannot provide answers to such clinical questions directly, the key conclusions inferred from big data are heavily dependent on how they are collected and interpreted. Big data, however, are essentially complex, heterogeneous and incomplete to a varying extent; analyzing big data requires sophisticated statistical methods such as multiple imputation or sensitivity analysis to manage missing variables and uncertainty of data, as used in the study by Dalén and his colleagues (4).
In this recent study (4), Dalén and his colleagues report on the survivals of the 41,906 patients undergoing isolated CABG registered in the Swedish Nationwide Heart Disease Database (SWEDEHEART) during a 13-year period. The entire cohorts were categorized and analyzed based on a history of HF and left ventricular (LV) EF measured preoperatively. They demonstrated that preoperative diagnosis of HF as well as reduced EF (<50%) is associated with poor short- and long-term survivals. Furthermore, they concluded that a history of HF may be more closely implicated with poor long-term survival in combined with readmission for HF than reduced EF in this cohort, indicating that preoperative symptoms and signs of HF should be carefully addressed in assessing operative risks for patients undergoing CABG.
Dalén and his colleagues should be congratulated on pioneering an intriguing study from the use of big data. Of note, patients with a prior history of HF had more co-morbidities such as increased age, diabetes and impaired kidney function compared with patients with no HF in this study. In particular, patients with HF and preserved EF were more often female and presented more with a history of hypertension, prior percutaneous coronary intervention, atrial fibrillation and stroke. Also notable is that CABG was performed in a quite conventional manner; off-pump CABG was done in 3.7% of the patients only and multi-arterial grafts were used in 4.4%. A history of HF appears to be a stronger risk factor for long-term mortality than reduced EF in this cohort, whereas early mortality may be more affected by reduced EF rather than HF syndrome. These results were identical even after adjusting for other demographic factors and comorbidities affecting death. The authors availed themselves of a huge sample size of nationwide registry with accurate and complete follow-up for mortality to come to these conclusions supported by enough statistical power.
The selection of patients with HF in this study, however, was not made based on solid diagnostic criteria grounded by echocardiographic, radiologic (e.g., chest X-ray) or laboratory (e.g., B-type natriuretic peptide) evaluations. Rather, the patients were categorized to a HF group in this study if they were assigned a multitude of the International Classification of Disease (ICD) codes designated as representing HF (Table 1) (4). Although HF refers to a clinical syndrome which results from a complex blend of structural and functional impairment of ventricular function (5), patients who were selected by the use of heterogeneous ICD codes only may constitute the biased group not truly representing patients with HF. Conversely, for patients assigned to a no-HF group due to the lack of designated ICD codes, the diagnosis of HF may have been missed. Despite the author’s argument for the validity of diagnosis for HF which emphasized on the involvement of cardiologists in diagnosing the entire cohort in the registry, the heterogeneity of the group with or without HF compels readers to question the reliability of this study.
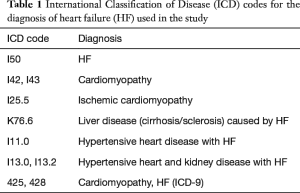
Full table
One of the main strengths in this study was to provide the prognostic insights of performing CABG for patients with HF and preserved EF from the analysis of big data with sufficient sample size. However, HF with preserved EF, predominantly caused by abnormalities in ventricular diastolic function, should be diagnosed not merely by ventricular contractility (EF), but by objective evidences supporting diastolic dysfunction (e.g., aberrant diastolic flow, elevated LV filling pressure) obtained by echocardiography or cardiac catheterization (6-8). In these regard, as agreed by the authors, the possibilities that HF with preserved EF is underdiagnosed or misdiagnosed by the lack of such supporting evidences are very likely. Thereby, the conclusions asserting the differences between the HF groups with preserved and reduced EF in this study may be altered by the introduction of new criteria into grouping the patients with HF and preserved EF.
Despite these issues, this study gives us a very important lesson: “listen to the patient’s complaints.” The authors raised noteworthy clinical implications: HF syndrome itself may be clinically more important than reduced EF in predicting the risk of death after CABG, therefore we should pay more attention to signs and symptoms suggestive of HF in addition to addressing ventricular contractility during preoperative evaluation of patients undergoing CABG. Current major risk models assessing early mortality after cardiac surgery, such as European System for Cardiac Operative Risk Evaluation (EuroSCORE) (9) and The Society of Thoracic Surgeons (STS) risk scores (10), require limited information in regard to HF such as left ventricular ejection fraction (LVEF), New York Heart Association (NYHA) functional classification or recent history of HF (within 2 weeks). In addition to these parameters, whether a history of HF should be incorporated into the risk models to help us with more accurate preoperative assessment warrants further investigations.
The advent of big data era is changing the overall aspects of clinical researches; analyzing data from a big sample size of population is becoming a prevailing way of doing clinical researches. Although clinical implications obtained from big data contribute to the process of making better decisions, the reliability of big data-driven research is still an issue. Researchers should endeavor to collect the best quality of data and analyze them based on objectively verifiable measures. These are also important lessons which authors have taught us in this study.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Lau E, Watson KE, Ping P. Connecting the Dots: From Big Data to Healthy Heart. Circulation 2016;134:362-4. [Crossref] [PubMed]
- Zhang Z. Big data and clinical research: perspective from a clinician. J Thorac Dis 2014;6:1659-64. [PubMed]
- Wang SD. Opportunities and challenges of clinical research in the big-data era: from RCT to BCT. J Thorac Dis 2013;5:721-3. [PubMed]
- Dalén M, Lund LH, Ivert T, et al. Survival After Coronary Artery Bypass Grafting in Patients With Preoperative Heart Failure and Preserved vs Reduced Ejection Fraction. JAMA Cardiol 2016;1:530-8. [Crossref] [PubMed]
- Jessup M, Brozena S. Heart failure. N Engl J Med 2003;348:2007-18. [Crossref] [PubMed]
- Aurigemma GP, Gaasch WH. Clinical practice. Diastolic heart failure. N Engl J Med 2004;351:1097-105. [Crossref] [PubMed]
- Redfield MM. Heart Failure with Preserved Ejection Fraction. N Engl J Med 2016;375:1868-1877. [Crossref] [PubMed]
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013;62:e147-239. [Crossref] [PubMed]
- Nashef SA, Roques F, Sharples LD, et al. EuroSCORE II. Eur J Cardiothorac Surg 2012;41:734-44. [Crossref] [PubMed]
- Shahian DM, O'Brien SM, Filardo G, et al. The Society of Thoracic Surgeons 2008 cardiac surgery risk models: part 1--coronary artery bypass grafting surgery. Ann Thorac Surg 2009;88:S2-22. [Crossref] [PubMed]


