Tracheobronchial tuberculosis
Introduction
The prevalence of tuberculosis-induced tracheobronchial stenosis varies as a function of the prevalence of tuberculosis, and is estimated to be as high as 10% to 40% in patients with pulmonary tuberculosis.
Tracheobronchial tuberculosis (TBTB) is diagnosed on histopathological examination of bronchoscopically obtained specimens showing granulomatous inflammation with caseation necrosis and/or positive acid fast bacilli culture on the microbiological exam. Specimen for culture can also be obtained from sputum but it is less sensitive compared to bronchial washing, biopsy or bronchoalveolar lavage; as described by Qingliang et al. in China where only 3/22 (13%) patients were sputum acid fast bacilli positive, and the remainder required bronchoscopic sampling for the diagnosis (1,2). The diagnosis of TBTB is often delayed due to its non-specific clinical symptoms. The course of endobronchial tuberculosis is highly variable and can range from complete resolution of the disease to fibrotic central airway obstruction (3).
About 68% of the patients with TBTB develop some degree of bronchostenosis in the initial 4–6 months of the disease and up to 90% of patients have some degree of stenosis beyond this initial time period (2,4,5). Among patients with active disease involving the pulmonary parenchyma, 10–39% of patients are shown to have a component of TBTB (6). Initial treatment is a 4 drug regimen therapy as in pulmonary tuberculosis. However, due to a highly unpredictable disease progression when it involves the tracheobronchial tree, future interventions including bronchoscopic airway dilation or stent placement may be necessary.
Pathogenesis
Although the exact pathogenesis of TBTB is not well understood, at least four different pathways are proposed leading to infection of tracheobronchial mucosa by mycobacteria tuberculi. Anyone or combinations of these pathways may lead to infections. In 1951, Smart proposed different methods of infection which included:
- Direct spread of tubercle bacilli from parenchymal tuberculosis or cavitary lesions containing abundant tubercle bacilli (7-10);
- Dissemination of tubercle bacilli from peribronchial lymphatic channels draining pulmonary parenchymal tuberculosis. This is supported by the finding that endobronchial biopsy in this patient population shows intact mucosa but submucosal involvement is noted on biopsy (7);
- Disease spread from contiguous mediastinal lymph nodes to the bronchial mucosa, occasionally resulting in Broncho nodal fistula. This mode of infection is particularly known to affect the pediatric population due to weaker airway walls and smaller airway diameter (11,12);
- Direct implantation of inhaled mycobacterium tuberculosis into the bronchial wall mucosa (13).
TBTB can be clinically divided into active disease (bronchoscopic findings of active caseating material, ulceration or granulation tissue formation or histologic findings of caseating necrosis or a positive tubercle bacilli on culture) and fibrotic diseases (fibrosis seen on bronchoscopic view or biopsy-confirmed fibrostenotic tuberculosis often found to be culture negative) (7). During the healing phase of active endobronchial tuberculosis, cicatrization can cause mucosal ulceration, necrosis and fibrosis leading to stenosis (6).
Left bronchial involvement is seen more frequently than right bronchial or tracheal involvement (14). It is postulated that the left mainstem bronchus is anatomically compressed by the aortic arch and the left mediastinal lymph nodes tend to get infected faster than the right sided lymph nodes, resulting in increased vulnerability of the left main stem bronchus to endobronchial infection in TBTB.
Diagnosis
- Clinical symptoms: the prevalence of endobronchial tuberculosis is found to be twice as high in females as males. Lee and colleagues (6) hypothesized that female patient population tend to expectorate less frequently due to social customs and norms causing endobronchial stasis of the secretions and subsequent infection. These women are usually found to be in their second and third decade of life (4,8,10,15,16). Presenting symptoms are variable, non-specific and insidious in onset. Cough is the most common presenting symptoms followed by sputum, weight loss, hemoptysis, chest pain and dyspnea (4,17). Not uncommonly patients are treated with antibiotics for bacterial pneumonia which proves inefficacious. Due to the non-specific symptoms, of TBTB alone, and its common coincidence with pulmonary/parenchymal disease, diagnosis is challenging and often delayed. In both the initial stages of solitary airway involvement of tuberculosis as well as in the late stages of TBTB, there have been reports of misdiagnosis of asthma in these patients (18). Wheeze and dyspnea are commonly reported particularly when patients develop bronchostenosis. Asthma, pneumonia and malignancy are often in the differential diagnosis of TBTB;
- The diagnostic yield of sputum examination is very variable. Despite best efforts and appropriate collection techniques a highly variable diagnostic rate of 13.6–53% is reported in patients with TBTB (2). Ozkaya et al. (1) reported on their experience with 23 patients with biopsy proven endobronchial tuberculosis all of whom were sputum smear negative. Such reports have ben indicative of a high false negative rate of negative sputum for acid fast bacilli in the diagnosis of TBTB;
- Tuberculin skin test was found to be positive in only 59.1% (2). Its lack of sensitivity in immunocompromised patients and lack of specificity due to its cross reactivity with non-tuberculous mycobacterium makes it an ineffective test to rule in or rule out pulmonary tuberculosis (2). Interferon gamma release assay (IGRA) has sensitivity of 64–92% but it cannot be relied upon to rule out or rule in pulmonary tuberculosis (19);
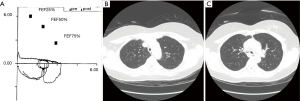
- Pulmonary function testing (PFT): The large airways contribute to approximately 50% of the total airway resistance (20,21). It is unclear what test most accurately quantifies the degree of upper airway obstruction. Spirometry typically shows limitation of maximal inspiratory and expiratory flows at high lung volumes. Miller and colleagues showed that lesions must narrow the tracheal lumen to <8 mm before symptoms occur (22-24). Moderate exercise limitation occurs when peak expiratory flow is reduced by about 40% and airway resistance is increased by about 600% (in which case FEV1 is still approximately 75% predicted). This limitation grossly corresponds to an upper airway that is narrowed to about 6 mm diameter. For all the reasons above, PFT tends to detect TBTB, after disease progression to the stage of fibrostenosis and significant central airway narrowing. If diffuse pulmonary disease accompanies TBTB, restrictive pattern disease may be present (25) (Figure 1);
- Chest imaging: chest imaging does not rule out endobronchial tuberculosis. Lee et al. (6) in a retrospective study found that 10% of the patients have normal chest X-ray. Pulmonary infiltrate is the most common findings seen on chest X-ray (18). In a study from south Korea (14) chest images of 121 patients with TBTB were reviewed. Seventy-one out of 121 patients (60%) had parenchymal infiltration while 24% had loss of volume. 8% of patients had cavitating lesions while surprisingly, 8% of patients had completely clear lung fields. Centrilobular nodules can be seen on CT of the chest. If patient develops tracheobronchial stenosis then depending on the airway involved, patient may have post-obstructive pneumonia or atelectasis in that region of the lung. CT imaging provides useful information about the extent of disease, length of bronchial involvement, any viable airway/lung parenchyma distal to the involved bronchus. Lee et al. (6) retrospectively looked at chest CT scans of 30 patients with lobar or complete lung atelectasis as a sequelae of TBTB in the setting of segmental, lobar or main stem bronchial stenosis. They concluded that chest CT scan can be useful in differentiating active TBTB from fibrotic stage disease. In active tuberculosis the bronchial walls look edematous and irregular while fibrotic stage disease appears smooth with no significant airway wall thickening or edema;
- Bronchoscopic view: Chung et al. (26) reported on their detailed bronchoscopic examination of patients with TBTB and classified the bronchoscopic view of endobronchial tuberculosis to seven subtypes: actively caseating, edematous-hyperemic, fibrostenotic, tumorous, granular, ulcerative and nonspecific bronchitic. These morphological features have prognostic significance; With the exception of the tumorous type, Chung and Lee suggested that the clinical outcome of these subtypes can be predicted in the first 3 months of treatment. In actively caseating type, bronchial mucosa is hyperemic and diffusely covered with whitish cheese like material. In edematous hyperemic type the cheese like appearance is absent but edema causes some degree of airway obstruction without any evidence of fibrosis. Fibrostenotic type results in marked narrowing of the bronchial lumen with fibrosis. Tumorous type presents with endobronchial exophytic tumor covered with caseous material and results in near complete occlusion of the bronchial lumen. They described granular type as endobronchial lesions that resemble scattered grains of boiled rice. Visible ulceration of bronchial mucosa is seen in the ulcerative type. In non-specific bronchitic type only mild mucosal swelling and or hyperemia were seen on bronchoscopy. Actively caseating, edematous-hyperemic type and the fibrostenotic type are more likely to proceed to bronchial stenosis despite treatment at 3 months. Frequently, healed endobronchial tuberculosis may leave black pigmentation within the airways. Multiple areas of black pigmentation can be seen on bronchoscopy. It is presumed that residue of mycobacterium tuberculosis leaves black pigments, a reportedly irreversible phenomenon (27).
Treatment
When treating endobronchial tuberculosis, two important goals are of utmost importance: eradication of tubercle bacilli and prevention of the most undesirable consequence, stenosis (28). While anti-tuberculous chemotherapy is efficient in controlling the infection, it does not appear to be a successful method in preventing residual TBTB (15,26,29) In a study done in South Korea Um et al. retrospectively looked at the predictors of persistent airway stenosis in 67 patients with microbiologically or histologically confirmed endobronchial tuberculosis that had completed anti-tuberculosis therapy. Patients received at least two bronchoscopies and endobronchial lesions were characterized as one of the seven subtypes originally described by Chung et al. (26). They reported the treatment outcome based on the degree of stenosis that was seen between the first and the second bronchoscopy, the second bronchoscopy was done at the completion of anti-tuberculous therapy. Persistent bronchostenosis occurred in 41.8% of the patients. In multivariate regression analysis, age >45 years, pure or combined fibrostenotic subtype and duration from onset of chief complaint to the initiation of anti-tuberculosis chemotherapy >90 days were identified as independent predictors of persistent airway stenosis. Oral corticosteroids (prednisolone equivalent >30 mg/d) did not reduce the frequency of persistent airway stenosis.
Chung et al. (26) reviewed 62 cases with actively caseating, edematous-hyperemic, fibrostenotic, and tumorous endobronchial tuberculosis all of whom showed luminal narrowing of the bronchus at diagnosis. Of these 62 patients, 43 patients continued to have endobronchial tuberculosis by the end of treatment; and despite appropriate treatment with anti-tuberculous therapy approximately 50% (22/43 patients) of the patients developed bronchostenosis. This pattern is consistent with other previous studies (30-32). Among 19 cases with granular, ulcerative, and nonspecific bronchitic endobronchial tuberculosis that did not show luminal narrowing of the bronchus at diagnosis, 17 cases healed without bronchostenosis, while 2 cases of granular type still developed bronchial stenosis after treatment. They concluded that the evolution of endobronchial tuberculosis is determined by initial bronchoscopic findings and the subtypes.
Treatment of TBTB depends on the stage at which the diagnosis is made. In the active phase of the disease, the goal is to control the infection and prevent tracheobronchial stenosis. There is no clear evidence that any single mode of therapy can decrease the incidence of TBTB in this patient population (3). The following management strategies are proposed in the literature.
- Corticosteroids: corticosteroids have been used in children (33) and adults (3) without any benefit. As mentioned above, tracheobronchial stenosis is the most common long term complication despite adequate treatment. Um et al. (3) described 67 patients which were divided in two groups, patients who received steroids (37 patients, >30 mg equivalent prednisone/day) and the ones who did not receive steroids as part of the treatment. Both groups were treated with standard anti-tuberculosis therapy. The age, sex, smoking history, duration of treatment with anti-tuberculosis chemotherapy, pulmonary function test were similar in both groups. There was no difference in the outcome. Interestingly, patients who received steroids had symptoms longer than the group that did not receive steroids (155.8±27.3 vs. 67.6±8.3 days, P=0.004);
- Medical therapy: medical treatment is of no benefit in the fibrotic stage (3). Restoring the airway patency and relieving the symptom is the key to the treatment in fibrotic stage. Patients with TBTB caused by organisms known or presumed to be drug susceptible are treated in the same way as pulmonary tuberculosis. In general, antituberculous regimens consists of two phases: an intensive phase (2 months), followed by a continuation phase (4 to 7 months); most patients receive 6 months of treatment (intensive phase of 2 months and continuation phase of 4 months (34,35). Directly observed therapy (DOT) by enforcing compliance, has shown to improve the outcome in patients with pulmonary tuberculosis and thus decreasing resistance (34,35). However effect of DOT is not studied on patients with TBTB (4). As discussed earlier, Chung et al. showed that patients who received anti-tuberculous chemotherapy >90 days after diagnosis of tuberculosis and airway involvement, were more likely to develop persistent airway stenosis. This may suggest that medical therapy has a significant role in the progression of disease to fibrosis in the airway; however, there are no randomized trials that have looked at the effect of anti-tuberculous chemotherapy on TBTB progression;
- Bronchoscopic: in tracheobronchial stenosis, bronchoscopic intervention is not standardized and patients are treated on case by case basis depending on the symptoms, PFT, co-morbidity, chest imaging and bronchoscopic findings (3,26). Bronchoscopic techniques include balloon dilatation alone, stent placement following balloon dilatation, laser photoresection, argon plasma coagulation and cryotherapy (36-38) (Figure 2). Interventional bronchoscopy has largely replaced surgical resection and bronchoplastic reconstruction which have been standard treatment in the past (29). Endobronchial ultrasound has been used to evaluate the underlying cartilaginous structure in the setting of tracheobronchomalcia in this patient population (38-40). It is recommended that patients with suspected tracheobronchial stenosis have an evaluation of the extent of their disease by pulmonary function tests with flow-volume loop followed by chest imaging when possible. Once subjective and objective data suggesting TBTB is obtained, patients will need flexible bronchoscopy and inspection of the airways. This can be done after anti-tuberculosis chemotherapy has completed. Endobronchial biopsy can be performed to rule out active endobronchial tuberculosis at this time.
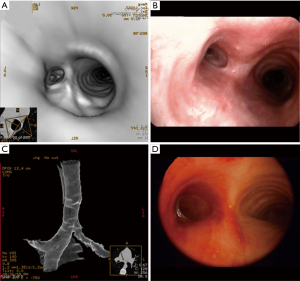
Iwamoto et al. (38) retrospectively assessed the efficacy of bronchoscopic techniques in treating airways stenosis in 30 patients due to TBTB. Twelve of 30 patients continued to have dyspnea, cough and repeated obstructive pneumonia due to tracheobronchial stenosis. One of 12 patients required pneumonectomy and the remainder underwent bronchoscopic interventions. The left main stem was the most involved site (8 patients). Tracheobronchial stents were inserted in 6/11 patients, 3 patients receiving silicone straight stents and the remaining three patients received silicone Y-stent. The remaining five patients only required balloon dilatation. There was significant improvement in symptoms in all patients. Minor complications including stent migration and granulation tissue were reported in the patients who had stent placement. The investigators did not report data on stent removal and long term follow up. As shown in this study, in about 30–40% of patients (38,41) balloon dilatation alone is sufficient. Radial endobronchial ultrasound is useful in examining the underlying cartilage, and any evidence of damaged cartilage warrants consideration of stent placement to support the airway as tracheobronchomalcia is a common finding in this patient population (38). Argon plasma coagulation, laser and cryoprobe can be used to remove the granulation tissue before or after dilatation and before stent placements.
When airway stenting is required, use of silicone stents is preferred. In the setting of benign airway stenosis such as TBTB, we recommend against the use of expandable metallic stents, particularly due to their increased risk of airway wall trauma and perforation, fatigue or rupture of the metallic stent and difficulty in retrieving the stent (39,40). Silicone stents instead are found to be removed easier and are less prone to granulation tissue formation. However, there are reports of self-expandable metallic alloy nitinol stents used for tuberculosis induced tracheobronchial stenosis (36-38,42).
Ryu et al. (36) in a retrospective study evaluated the role of bronchoscopic interventions including silicone stenting at a tertiary referral hospital in South Korea. Eighty patients underwent bronchoscopic intervention for the treatment of post tuberculous tracheobronchial stenosis over a period of 4 years. These interventions included balloon dilatation, neodymium-yttrium aluminum garnet laser resection as first line methods of airway dilation. The median luminal diameter was 9.5 mm in the trachea and 5 mm in bronchus when the lumen was measured by bronchoscopy.
Silicone stents were required in 75/80 (94%) patients. Interventions were performed in patients who were symptomatic from post tuberculous tracheobronchial stenosis (progressive dyspnea, recurrent post-obstructive pneumonia or atelectasis of the lung or reduced lung function). There was a female predominance and median of 33 years of age and the most common involved location was the left main stem bronchus (65%).
Stents were removed in 54/75 (72%) patients after a median of 14 months. Fourthy nine of 54 patients had a successful stable clinical course during a follow up period of 36 months. However, 5 patients showed recurrence and needed re-stenting. These 5 patients along with remaining 18 patients (total 23 patients) required prolonged stent placement (median 32 months). Three patients showed no response to bronchoscopic intervention and needed surgical resection involving end to end anastomosis. During a median follow up period of 41 months, clinical outcome was unchanged in all patients.
Subgroup analysis between those who had their stents successfully removed and those who continued to have stents showed that patients with prolonged stent placement had a lower baseline FEV1 and need for more re-intervention. Granulation tissue and re-stenosis were also more common in patients who required re-stenting and those stented for longer duration.
Minor complications such as cough, mucus plugging and blood tinged sputum was seen in majority of the patients. Eight of 80 (10%) patients experienced major complications which included massive bleeding leading to respiratory failure and requiring blood transfusion and mechanical ventilation, Pneumothorax occurred in five patients and pneumomediastinum in two patients and there was no mortality.
Similarly, Wan et al. (43) published their experience with silicone stents in patients with tuberculosis-induced tracheobronchial stenosis over 7 years. Seven patients with a mean age of 43 years underwent a total of 11 dilatations with placement of 10 straight stents and 1 Y stent. Stents were left in situ for a mean of 32 months. One patient had pneumothorax and two patients had migration of the stents requiring stent revision. There was no mortality. These and other studies conclude that silicone stents are safe in the management of patients with tuberculosis-induced tracheobronchial stenosis (36). There are no definite guidelines on the required duration of stent placement in the airway. Based on published data from investigators in high prevalent areas with tuberculosis, an attempt can be made to remove stents in 8–16 months after placement (36,44). These studies highlight the role of advanced therapeutic bronchoscopy procedures in the management of chronic fibrostenosis in the central airways following tuberculosis and anti-tuberculous therapy. Airway stent placement can serve as a temporary therapeutic intervention followed by successful stent removal with no need of re-intervention.
Conclusions
TBTB can present with and without evidence of parenchymal disease on imaging. The course of the disease in unpredictable but delay in medical therapy is shown to increase the likelihood of progression to fibrostenosis. Early diagnosis and treatment is often missed due to its varied clinical presentation. Patients with pulmonary tuberculosis presenting with continued respiratory symptoms after treatment should be evaluated for tracheobronchial stenosis. Interventional bronchoscopic approaches should be considered to restore airway patency.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Ozkaya S, Bilgin S, Findik S, et al. Endobronchial tuberculosis: histopathological subsets and microbiological results. Multidiscip Respir Med 2012;7:34. [Crossref] [PubMed]
- Qingliang X, Jianxin W. Investigation of endobronchial tuberculosis diagnoses in 22 cases. Eur J Med Res 2010;15:309-13. [Crossref] [PubMed]
- Um SW, Yoon YS, Lee SM, et al. Predictors of persistent airway stenosis in patients with endobronchial tuberculosis. Int J Tuberc Lung Dis 2008;12:57-62. [PubMed]
- Kashyap S, Solanki A. Challenges in endobronchial tuberculosis: from diagnosis to management. Pulm Med 2014;2014:594806.
- Han JK, Im JG, Park JH, et al. Bronchial stenosis due to endobronchial tuberculosis: successful treatment with self-expanding metallic stent. AJR Am J Roentgenol 1992;159:971-2. [Crossref] [PubMed]
- Lee JY, Yi CA, Kim TS, et al. CT scan features as predictors of patient outcome after bronchial intervention in endobronchial TB. Chest 2010;138:380-5. [Crossref] [PubMed]
- Moon WK, Im JG, Yeon KM, et al. Tuberculosis of the central airways: CT findings of active and fibrotic disease. AJR Am J Roentgenol 1997;169:649-53. [Crossref] [PubMed]
- Ip MS, So SY, Lam WK, et al. Endobronchial tuberculosis revisited. Chest 1986;89:727-30. [Crossref] [PubMed]
- Smith LS, Schillaci RF, Sarlin RF. Endobronchial tuberculosis. Serial fiberoptic bronchoscopy and natural history. Chest 1987;91:644-7. [Crossref] [PubMed]
- SMART J. Endo-bronchial tuberculosis. Br J Tuberc Dis Chest 1951;45:61-8. [Crossref] [PubMed]
- Natkunam R, Tse CY, Ong BH, et al. Carinal resection for stenotic tuberculous tracheitis. Thorax 1988;43:492-3. [Crossref] [PubMed]
- Daly JF, Brown DS, Lincoln EM, et al. Endobronchial tuberculosis in children. Dis Chest 1952;22:380-98. [Crossref] [PubMed]
- Mariotta S, Guidi L, Aquilini M, et al. Airway stenosis after tracheo-bronchial tuberculosis. Respir Med 1997;91:107-10. [Crossref] [PubMed]
- Lee JH, Park SS, Lee DH, et al. Endobronchial tuberculosis. Clinical and bronchoscopic features in 121 cases. Chest 1992;102:990-4. [Crossref] [PubMed]
- Shim YS. Endobronchial tuberculosis. Respirology 1996;1:95-106. [Crossref] [PubMed]
- Lee JH, Lee DH, Park SS. Endobronchial tuberculosis: clinical and bronchofiberscopic features. Korean J Intern Med 1986;1:229-32. [Crossref] [PubMed]
- Sahin F, Yıldız P. Characteristics of endobronchial tuberculosis patients with negative sputum acid-fast bacillus. J Thorac Dis 2013;5:764-70. [PubMed]
- Argun Baris S, Onyilmaz T, Basyigit I, et al. Endobronchial Tuberculosis Mimicking Asthma. Tuberc Res Treat 2015;2015:781842.
- Dheda K, van Zyl Smit R, Badri M, et al. T-cell interferon-gamma release assays for the rapid immunodiagnosis of tuberculosis: clinical utility in high-burden vs. low-burden settings. Curr Opin Pulm Med 2009;15:188-200. [Crossref] [PubMed]
- Hyatt RE, Wilcox RE. Extrathoracic airway resistance in man. J Appl Physiol 1961;16:326-30. [PubMed]
- Ferris BG Jr, Mead J, Opie LH. Partitioning of respiratory flow resistance in man. J Appl Physiol 1964;19:653-8. [PubMed]
- Miller RD, Hyatt RE. Obstructing lesions of the larynx and trachea: clinical and physiologic characteristics. Mayo Clin Proc 1969;44:145-61. [PubMed]
- Melissant CF, Lammers JW, Demedts M. Relationship between external resistances, lung function changes and maximal exercise capacity. Eur Respir J 1998;11:1369-75. [Crossref] [PubMed]
- Shojaee S, Tilluckdharry L, Manning H. Tuberculosis-induced tracheobronchial stenosis during pregnancy. J Bronchology Interv Pulmonol 2012;19:211-5. [Crossref] [PubMed]
- Lee JH, Chung HS. Bronchoscopic, radiologic and pulmonary function evaluation of endobronchial tuberculosis. Respirology 2000;5:411-7. [Crossref] [PubMed]
- Chung HS, Lee JH. Bronchoscopic assessment of the evolution of endobronchial tuberculosis. Chest 2000;117:385-92. [Crossref] [PubMed]
- Tunsupon P, Panchabhai TS, Khemasuwan D, et al. Black bronchoscopy. Chest 2013;144:1696-706. [Crossref] [PubMed]
- Rikimaru T. Endobronchial tuberculosis. Expert Rev Anti Infect Ther 2004;2:245-51. [Crossref] [PubMed]
- Hoheisel G, Chan BK, Chan CH, et al. Endobronchial tuberculosis: diagnostic features and therapeutic outcome. Respir Med 1994;88:593-7. [Crossref] [PubMed]
- Calpe JL, Chiner E, Larramendi CH. Endobronchial tuberculosis in HIV-infected patients. AIDS 1995;9:1159-64. [Crossref] [PubMed]
- Kim YH, Kim HT, Lee KS, et al. Serial fiberoptic bronchoscopic observations of endobronchial tuberculosis before and early after antituberculosis chemotherapy. Chest 1993;103:673-7. [Crossref] [PubMed]
- Kurasawa T, Kuze F, Kawai M, et al. Diagnosis and management of endobronchial tuberculosis. Intern Med 1992;31:593-8. [Crossref] [PubMed]
- Toppet M, Malfroot A, Derde MP, et al. Corticosteroids in primary tuberculosis with bronchial obstruction. Arch Dis Child 1990;65:1222-6. [Crossref] [PubMed]
- Nahid P, Dorman SE, Alipanah N, et al. Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: Treatment of Drug-Susceptible Tuberculosis. Clin Infect Dis 2016;63:e147-95. [Crossref] [PubMed]
- Nahid P, Dorman SE, Alipanah N, et al. Executive Summary: Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America Clinical Practice Guidelines: Treatment of Drug-Susceptible Tuberculosis. Clin Infect Dis 2016;63:853-67. [Crossref] [PubMed]
- Ryu YJ, Kim H, Yu CM, et al. Use of silicone stents for the management of post-tuberculosis tracheobronchial stenosis. Eur Respir J 2006;28:1029-35. [Crossref] [PubMed]
- Low SY, Hsu A, Eng P. Interventional bronchoscopy for tuberculous tracheobronchial stenosis. Eur Respir J 2004;24:345-7. [Crossref] [PubMed]
- Iwamoto Y, Miyazawa T, Kurimoto N, et al. Interventional bronchoscopy in the management of airway stenosis due to tracheobronchial tuberculosis. Chest 2004;126:1344-52. [Crossref] [PubMed]
- Kato R, Kakizaki T, Hangai N, et al. Bronchoplastic procedures for tuberculous bronchial stenosis. J Thorac Cardiovasc Surg 1993;106:1118-21. [PubMed]
- Hsu HS, Hsu WH, Huang BS, et al. Surgical treatment of endobronchial tuberculosis. Scand Cardiovasc J 1997;31:79-82. [Crossref] [PubMed]
- Bolliger CT, Probst R, Tschopp K, et al. Silicone stents in the management of inoperable tracheobronchial stenoses. Indications and limitations. Chest 1993;104:1653-9. [Crossref] [PubMed]
- Ducic Y, Khalafi RS. Use of endoscopically placed expandable nitinol tracheal stents in the treatment of tracheal stenosis. Laryngoscope 1999;109:1130-3. [Crossref] [PubMed]
- Wan IY, Lee TW, Lam HC, et al. Tracheobronchial stenting for tuberculous airway stenosis. Chest 2002;122:370-4. [Crossref] [PubMed]
- Martinez-Ballarin JI, Diaz-Jimenez JP, Castro MJ, et al. Silicone stents in the management of benign tracheobronchial stenoses. Tolerance and early results in 63 patients. Chest 1996;109:626-9. [Crossref] [PubMed]


