Pathological upstaging and treatment strategy of clinical stage I small cell lung cancer following surgery
Overview
Small cell lung cancer (SCLC) represents approximately 10–15% of all lung cancers, and its incidence has been steadily decreasing in the past two decades, primarily because of reduction in cigarette smoking, which is the primary cause of this type of tumor (1). SCLC originates from neuroendocrine cell precursors and is characterized by rapid growth, early dissemination to regional lymph nodes, and distant metastasis, and resultant poor prognosis along with initial sensitivity to chemotherapy and radiotherapy (2,3).Current standard therapy for SCLC relies on chemotherapy or chemoradiotherapy, even for patients with “limited” disease. In contrast, the role of primary surgical resection in such patients remains controversial because only a minority of early stage SCLC patients presents without metastasis and are candidates for surgery. Recently, based on favorable surgical results reported in several large cohort studies for limited disease SCLC (4-7), the American College of Chest Physicians (ACCP) indicate surgical has recommended resection only for patients with clinical stage I SCLC (T1–2, N0), followed by chemotherapy (8). Similarly, the National Comprehensive Cancer Network (NCCN) guidelines recommend surgery with adjuvant chemotherapy for stage I disease alone and specify lobectomy as the preferred resection procedure (9).
Accurate staging provides prognostic information, which aids in the planning of treatment strategies for all types of lung cancer: non-small cell lung cancer (NSCLC) and SCLC. Use of tumor-node-metastasis (TNM) descriptors has been the basis of the NSCLC staging system since 1973 (10), but these descriptors are seldom used in SCLC staging. Because these systems have historically relied on surgical confirmation for their accuracy, and as stage earlier, patients with SCLC seldom present at a stage for which surgery is appropriate. Instead, most clinicians have used two group staging systems for SCLC, which define the limited stage as tumor being confined to one hemithorax with regional lymph node metastasis including both ipsilateral and contralateral hilar, supraclavicular, and mediastinal nodes, as well as ipsilateral pleural effusion, and these definitions are still most relevant for clinical decision making (11).
More recent studies and the NCCN guidelines use the American Joint Committee on Cancer (AJCC) tumor, node, and metastasis staging for more precise stratification of disease extent. Moreover, the International Association for the Study of Lung Cancer (IASLC) recommendations for SCLC staging notes the significance of T and N stage on survival (12). The current lung cancer TNM staging system was developed by IASLC and adopted by the AJCC Cancer Staging Manual 7th edition; this staging system is applicable to both NSCLC and SCLC based on studies by the IASLC that showed the prognostic significance of various stage designations in both diseases (4,12-14). The current TNM staging system is based on 8,088 SCLC patients and provides better prognostic information and more precise nodal staging, which is required for conformal radiation techniques and intensity-modulated radiation therapy. The former term “limited stage” would now include T1–4, N0–3, and M0 tumors, whereas metastatic tumors encompass former extensive-stage patients. In addition, T1/T2 N0/N1 M0 tumors, previously described as “very limited stage” tumors, were identified as a group with more favorable outcomes compared with patients with N2/N3 tumors, which must be considered for curative-intent surgery according to expert consensus.
Upstaging
Clinical staging is often applied in order to guide management and pathological staging to predict prognosis, although the two aims are often not mutually exclusive. Approximately 5% of patients with SCLC present with T1/2 N0/1 M0 tumors; such patients have more favorable outcomes with 5-year survival rates of approximately 50% (6,15); moreover, surgical approaches are justified in such patients after ruling out mediastinal lymph node involvement. Despite advances in the diagnosis and preoperative staging of lung cancer using positron emission tomography-computed tomography (PET-CT) (16), pathological upstaging of early-stage disease remains a common finding (17,18). A recent review suggested that with PET-CT, 9% of patients are upstaged whereas 4% are downstaged; thus, PET-CT findings that could affect treatment decisions should be pathologically confirmed (19). The current staging protocols may underestimate the disease extent in up to 28% of patients with clinical stage I NSCLC (20,21). On the other hand, the overall concordance between clinical and pathological TNM staging was 58% in IASLC data bases of SCLC (4). Before curative-intent surgery, invasive mediastinal staging such as endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) (22) and surgical mediastinoscopy (8) is recommended to evaluate for regional and distant disease conditions, particularly for SCLC, because nodal involvement designation has traditionally been considered as a contraindication to surgery. Therefore, the present manuscript must be meaningful, as well as previous studies, and one of the largest nationally representative data pool that could identify the incidence of pathological upstaging (19.7–25%) after surgical resection of SCLC (Table 1).
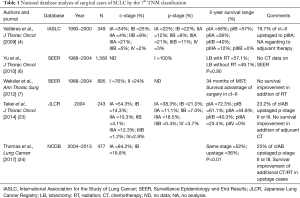
Full table
Treatment modality
Despite changes in demographics and treatment, the median and 5-year survival rates for patients with SCLC have not improved remarkably over the past 15 years. Based on analysis of 68,611 patients with SCLC in NCDB, the median survival of patients with ESCLC and LSCLC was 6.1 and 12.9 months in 2007, respectively, which did not improve markedly from the median survival reported between 1992 and 2002 (25). Nevertheless, in small surgical series of patients with SCLC, the TNM staging system has been prognostic of outcomes, particularly in the era of adjuvant chemotherapy. The average 5-year survival reported in this large series of stage I SCLC after resection is 52%, which is in line with the corresponding values reported in current large-scale studies (Table 1) (4,6,7,23). Based on analysis of these databases, surgery seems to offer reasonable survival in patients who are node negative and undergo lobectomy.
Data regarding clinical outcomes of patients who are pathologically upstaged because of nodal metastases is limited, which is critical to define postoperative recommendations for adjuvant therapy. Moreover, in patients with nodal disease identified at the time of surgical resection, recommendations for the use of adjuvant radiation therapy in addition to chemotherapy are based upon limited evidence. Nevertheless, additional chemoradiotherapy seems reasonable for patients with nodal disease identified at the time of surgical resection as well as those with LSCLC, for which chemoradiotherapy is the standard treatment. Although such patients should receive four cycles of adjuvant chemotherapy and postoperative thoracic radiotherapy when staged pN1 or pN2 in the present manuscript, no improvement in survival has been observed in such patients according to analyses of other large national data bases (Table 1).
Guidelines for treatment of patients with pathological nodal disease after surgery are based on limited evidence. Several studies have investigated the role of surgery combined with chemotherapy and radiation therapy to improve local recurrence rates but have reported mixed results (26-28). In contrast, patients with limited disease can be successfully treated with combined concomitant chemoradiotherapy, known to provide a cure rate of 15–20% (29) but a local recurrence rate of as high as 50% (30). Therefore, NCCN and European Society for Medical Oncology (ESMO) guidelines recommend adjuvant chemotherapy and radiation therapy for patients found to have nodal disease (pN+) following curative resection; however, both groups cite these recommendations based on insufficient or lower level of evidence (9,31).
Limitations
Limitations of this analysis include a retrospective study, no randomization for adjuvant treatment, lack of preoperative histopathological diagnosis data, lack of information regarding preoperative staging methods, no information regarding aims of preoperative treatment and whether induction treatment was followed by surgery or salvage surgery. Selection bias in selecting patients who should or should not receive radiation therapy remains unknown. Finally, there was no central pathology review. It must be emphasized that “resectable” SCLC represents only a small proportion of patients with SCLC, which tends to magnify the effects of selection.
Conclusions
Indications for surgical resection of SCLC have been extremely limited. Because “resectable” SCLC represents only a small proportion of patients with SCLC, studies comparing surgical resection among a large number of patients are unlikely to be conducted. This study presents data on >1,000 patients with clinical stage I SCLC who underwent resection, indicating the national practice patterns and outcomes, and thus provides additional insight into the role of surgery in such cases. Based on this analysis, surgery did seem to have a vital role in the management of selected patients with clinical stage I SCLC who revealed favorable survival outcomes. Based on these findings, four cycles of adjuvant chemotherapy should be administered after surgery. On the other hand, postoperative radiotherapy should be considered in cases of unforeseen N2 or N1 or in patients who have not undergone systematic nodal dissection.
Further evaluation of this question is warranted before an actionable recommendation can be made; in fact, additional studies to confirm this are underway. This should be the aim of continued clinical studies, and prospective randomized clinical trials are required to assess the effects of adjuvant chemoradiotherapy after surgery in patients with pathological stage II–III tumors, which may more clearly identify patient and tumor characteristics associated with improved outcomes after primary resection. Additional studies are needed to improve the existing clinical classification models for patients who are potentially suitable for surgery and to develop better prognostic models in patients undergoing complete resection for SCLC.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Govindan R, Page N, Morgensztern D, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol 2006;24:4539-44. [Crossref] [PubMed]
- van Meerbeeck JP, Fennell DA, De Ruysscher DK. Small-cell lung cancer. Lancet 2011;378:1741-55. [Crossref] [PubMed]
- Rudin CM, Ismaila N, Hann CL, et al. Treatment of Small-Cell Lung Cancer: American Society of Clinical Oncology Endorsement of the American College of Chest Physicians Guideline. J Clin Oncol 2015;33:4106-11. [Crossref] [PubMed]
- Vallières E, Shepherd FA, Crowley J, et al. The IASLC Lung Cancer Staging Project: proposals regarding the relevance of TNM in the pathologic staging of small cell lung cancer in the forthcoming (seventh) edition of the TNM classification for lung cancer. J Thorac Oncol 2009;4:1049-59. [Crossref] [PubMed]
- Lim E, Belcher E, Yap YK, et al. The role of surgery in the treatment of limited disease small cell lung cancer: time to reevaluate. J Thorac Oncol 2008;3:1267-71. [Crossref] [PubMed]
- Yu JB, Decker RH, Detterbeck FC, et al. Surveillance epidemiology and end results evaluation of the role of surgery for stage I small cell lung cancer. J Thorac Oncol 2010;5:215-9. [Crossref] [PubMed]
- Weksler B, Nason KS, Shende M, et al. Surgical resection should be considered for stage I and II small cell carcinoma of the lung. Ann Thorac Surg 2012;94:889-93. [Crossref] [PubMed]
- Jett JR, Schild SE, Kesler KA, et al. Treatment of small cell lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e400S-19S.
- Kalemkerian GP, Akerley W, Bogner P, et al. Small cell lung cancer. J Natl Compr Canc Netw 2013;11:78-98. [PubMed]
- Goldstraw P, Chansky K, Crowley J, et al. The IASLC Lung Cancer Staging Project: Proposals for Revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM Classification for Lung Cancer. J Thorac Oncol 2016;11:39-51. [Crossref] [PubMed]
- Stahel RA. Diagnosis, staging, and prognostic factors of small cell lung cancer. Curr Opin Oncol 1991;3:306-11. [Crossref] [PubMed]
- Shepherd FA, Crowley J, Van Houtte P, et al. The International Association for the Study of Lung Cancer lung cancer staging project: proposals regarding the clinical staging of small cell lung cancer in the forthcoming (seventh) edition of the tumor, node, metastasis classification for lung cancer. J Thorac Oncol 2007;2:1067-77. [Crossref] [PubMed]
- Ignatius Ou SH, Zell JA. The applicability of the proposed IASLC staging revisions to small cell lung cancer (SCLC) with comparison to the current UICC 6th TNM Edition. J Thorac Oncol 2009;4:300-10. [Crossref] [PubMed]
- Goldstraw P, Crowley J, Chansky K, et al. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol 2007;2:706-14. [Crossref] [PubMed]
- Schreiber D, Rineer J, Weedon J, et al. Survival outcomes with the use of surgery in limited-stage small cell lung cancer: should its role be re-evaluated? Cancer 2010;116:1350-7. [Crossref] [PubMed]
- Bradley JD, Dehdashti F, Mintun MA, et al. Positron emission tomography in limited-stage small-cell lung cancer: a prospective study. J Clin Oncol 2004;22:3248-54. [Crossref] [PubMed]
- D'Cunha J, Herndon JE 2nd, Herzan DL, et al. Poor correspondence between clinical and pathologic staging in stage 1 non-small cell lung cancer: results from CALGB 9761, a prospective trial. Lung Cancer 2005;48:241-6. [Crossref] [PubMed]
- Wilson JL, Louie BE, Cerfolio RJ, et al. The prevalence of nodal upstaging during robotic lung resection in early stage non-small cell lung cancer. Ann Thorac Surg 2014;97:1901-6; discussion 1906-7.
- Thomson D, Hulse P, Lorigan P, et al. The role of positron emission tomography in management of small cell lung cancer. Lung Cancer 2011;73:121-6. [Crossref] [PubMed]
- López-Encuentra A, García-Luján R, Rivas JJ, et al. Comparison between clinical and pathologic staging in 2,994 cases of lung cancer. Ann Thorac Surg 2005;79:974-9; discussion 979. [Crossref] [PubMed]
- Boffa DJ, Kosinski AS, Paul S, et al. Lymph node evaluation by open or video-assisted approaches in 11,500 anatomic lung cancer resections. Ann Thorac Surg 2012;94:347-53; discussion 353. [Crossref] [PubMed]
- Wada H, Nakajima T, Yasufuku K, et al. Lymph node staging by endobronchial ultrasound-guided transbronchial needle aspiration in patients with small cell lung cancer. Ann Thorac Surg 2010;90:229-34. [Crossref] [PubMed]
- Takei H, Kondo H, Miyaoka E, et al. Surgery for small cell lung cancer: a retrospective analysis of 243 patients from Japanese Lung Cancer Registry in 2004. J Thorac Oncol 2014;9:1140-5. [Crossref] [PubMed]
- Thomas DC, Arnold BN, Rosen JE, et al. Defining outcomes of patients with clinical stage I small cell lung cancer upstaged at surgery. Lung Cancer 2017;103:75-81. [Crossref] [PubMed]
- Gaspar LE, McNamara EJ, Gay EG, et al. Small-cell lung cancer: prognostic factors and changing treatment over 15 years. Clin Lung Cancer 2012;13:115-22. [Crossref] [PubMed]
- Fox W, Scadding JG. Treatment of oat-celled carcinoma of the bronchus. Lancet 1973;2:616-7. [Crossref] [PubMed]
- Lad T, Piantadosi S, Thomas P, et al. A prospective randomized trial to determine the benefit of surgical resection of residual disease following response of small cell lung cancer to combination chemotherapy. Chest 1994;106:320S-323S. [Crossref] [PubMed]
- Eberhardt W, Stamatis G, Stuschke M, et al. Prognostically orientated multimodality treatment including surgery for selected patients of small-cell lung cancer patients stages IB to IIIB: long-term results of a phase II trial. Br J Cancer 1999;81:1206-12. [Crossref] [PubMed]
- Turrisi AT 3rd, Kim K, Blum R, et al. Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med 1999;340:265-71. [Crossref] [PubMed]
- Pignon JP, Arriagada R, Ihde DC, et al. A meta-analysis of thoracic radiotherapy for small-cell lung cancer. N Engl J Med 1992;327:1618-24. [Crossref] [PubMed]
- Früh M, De Ruysscher D, Popat S, et al. Small-cell lung cancer (SCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2013;24 Suppl 6:vi99-105. [Crossref] [PubMed]


