Echocardiography to guide fluid therapy in critically ill patients: check the heart and take a quick look at the lungs
Cardiac output (CO) is the major determinant of oxygen delivery (DO2) to organs. A low CO may be responsible for reduced DO2, which may lead, in turn, to tissue hypoperfusion and organ dysfunction and failure.
In cases of low output syndrome, one of the primarily therapeutic steps to improve CO is the administration of fluids (1). However, it is important to avoid administering fluids when critically ill patients are not fluid responders, as volume overload is associated with complications and poor outcome (2). Equally, it is important to avoid hypovolemia, which may potentially be associated with impaired organ perfusion and poor outcome (2).
A patient who responds to a fluid challenge is defined as one who will increase his baseline CO by more than 10% after volume loading, regardless of the method used to evaluate CO (3).
A number of invasive, less-invasive and non-invasive haemodynamic monitoring systems provide CO (2). In addition, many of these offer physicians some haemodynamic variables, which, under specific circumstances, are useful in identifying those patients who respond to a fluid challenge (2). These variables are called “dynamic indices of fluid responsiveness (FR)”. And it is in this context that echocardiography has emerged as an additional and useful tool for the assessment of cardiac function, CO and FR in intensive care unit (ICU) patients.
Echocardiography in the ICU
The use of echocardiography in a critical care setting has two distinguishable skill levels. The first of these is a basic-level use (4), which incorporates the standard technical expertise that every intensivist should possess. For instance, the physician is capable of demonstrating pleural or pericardial effusion and cardiac dysfunction. It has been shown that the use of echocardiography can improve outcomes, even when employed at the basic level (5). Kanji and colleagues, for instance, showed a significant reduction in acute kidney injury and mortality in critically ill patients who were monitored and managed with echocardiography (5).
The second, advanced level applies to intensivists using echocardiography as a haemodynamic tool (4). Here, the technique plays a pivotal role in monitoring unstable patients to assess their haemodynamic changes during fluid, inotropic and vasoactive drug administration (1).
One additional advantage when using echocardiography in the management of fluid administration in critically ill patients is the potential to exclude conditions that may affect the reliability of some dynamic indices of FR, such as pulse pressure variation (PPV), stroke volume variations (SVV) and systolic pressure variation (SPV). Specifically, echocardiography can reveal right ventricular dysfunction and cardiac tamponade, which, when present but unknown, are responsible for erroneous interpretations of the aforementioned dynamic indices (4).
Echocardiography to assess CO
Echocardiography allows physicians to estimate CO non-invasively in critically ill patients (1). Briefly, stroke volume (SV) can be calculated as the product of the aortic valve area [cross sectional area (CSA)] by the velocity time integral (VTI) of aortic blood flow (ABF) (6). To obtain CSA, the diameter of the left ventricular out-flow tract (LVOT) from the parasternal window is used. VTI is measured at the same site of the LVOT at the apical five-chamber view using pulsed-wave Doppler (6).
Echocardiography and preload and FR variables
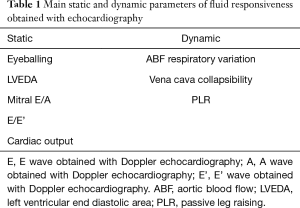
Echocardiography can provide static and dynamic preload and FR variables.
Static parameters
Static parameters include a number of variables that are measured under a single ventricular loading condition and allow the preload of the right and left ventricles (LVs) to be estimated. The assumption is that the lower the preload, the greater the probability that SV will increase following a fluid challenge. However, the consensus in the literature is that static parameters are not useful in predicting FR (2).
Static indexes obtained with echocardiography include volume and filling pressure parameters (Table 1). The fastest evaluation of the volume status of cardiac chambers is obtained by visual evaluation. An indication of severe hypovolemia is the “kissing papillary muscle sign”, where opposing papillary muscles come into contact with each other at the end of the systole (7). Among the volume parameters, the left ventricular end diastolic area (LVEDA) is popular in clinical practice due to its simplicity of measurement. An LVEDA of less than 10 cm2 suggests significant hypovolemia. Importantly, clinicians should bear in mind that severe concentric hypertrophy can reduce the LVEDA even without hypovolemia. Conversely, an LVEDA of more than 20 cm2 suggests volume overload (7). Unfortunately, only a few researchers have demonstrated that LVEDA is a reliable index of FR in critically ill patients. In addition, a cut-off value for the LVEDA is yet to be discovered.
Full table
In the last two decades, echocardiography has been used to estimate some indirect parameters of cardiac filling pressures. The study of the diastolic function of the LV in critically ill patients via the acquisition of mitral and pulmonary venous flow patterns is a means by which to investigate the loading conditions and filling pressures of the heart. This has significant diagnostic value in critically ill patients. For instance, for a patient in whom the analysis of the transmitral flow shows an alteration of the ratio between E and A waves (E/A ratio) and deceleration time (DT) (i.e., E/A >2 and DT <160 msec), the capillary wedge pressure is usually greater than 18 mmHg and exceeds 20 mmHg if DT is <120 msec (8). The E/A ratio hence seems to be a strong Doppler echocardiography parameter by which to predict the pulmonary artery occlusion pressure (PAOP) value (8). However, there has been little interest to date in estimating the filling pressures using echocardiography in ICU patients, except prior to 2012 when the technique was used to assess PAOP, with less than 18 mmHg being taken to denote acute respiratory distress syndrome (ARDS).
Dynamic parameters
In recent years, dynamic parameters have been successfully used to determine whether a patient’s heart is working on the ascending portion of the Frank-Starling curve (Table 1). In such conditions, it is possible that volume challenges may lead to an increase in SV, with a patient classified as “fluid responder” if their baseline SV increases by more than 10% (3). In addition, there is a physiological beat-by-beat change in the dynamic parameters related to variations in intrathoracic pressure secondary to either the mechanical or the spontaneous breathing cycle. Simply stated: the greater the tidal volume, the larger the changes in the dynamic indexes. This phenomenon is called “heart-lung interaction” and is used to monitor the variations of the dynamic indexes before and after a fluid challenge (2).
The most frequently used echocardiographic dynamic parameters of FR take into account the variations in ABF and velocity (V), determined by the heart-lung interaction. These are: (I) the variation of the maximal (Vpeak) Doppler velocity in the left ventricular outflow tract (ΔVmaxAo), calculated as (Vpeakmax − Vpeakmin)/(Vpealmax + Vpeakmin/2) ×100; and (II) the variation of aortic (ao) VTI, calculated as (VTIaomax − VTIaomin)/(VTIaomax + VTIaomin/2) ×100 (9,10).
Feissel and colleagues demonstrated that analysis of respiratory changes in aortic blood velocity (specifically the Vpeak) before volume expansion was an accurate method for predicting the haemodynamic effects of volume expansion in septic patients receiving mechanical ventilation (9). Similarly, Slama and co-workers simulated different preload conditions using a stepwise cumulative withdrawal and reinfusion of blood in an animal model (rabbit). Their results indicated the superiority of echocardiographic dynamic parameters of FR over static variables of cardiac preload as a means to predict FR under mechanical ventilation (10). It may hence be stated with some confidence that dynamic echocardiographic indexes are definitively and considerably superior to their static equivalents when it comes to predicting FR.
Variations in venae cavae diameter
Significant experience of echocardiography is required to evaluate the dynamic indexes of FR in clinical practice. In recent years, interest has been shown in using the variations in the diameter of the superior and inferior venae cavae as a means to assess the volume status of critically ill patients.
The respiratory variation of the superior vena cava (ΔSVC) is assessed using trans-oesophageal echocardiography (TEE). In mechanically ventilated patients, the inspiratory increase in intrathoracic pressure during lung inflation may produce a cyclic change in the diameter of the superior vena. ΔSVC has predicted FR in mechanically ventilated septic patients, with a cut-off value of 36% (11). Similarly, the respiratory variations of the inferior vena cava (ΔIVC) are measured using trans-thoracic echocardiography (TTE), with a cut-off value of 18% in the same category of patients (12).
A recent and interesting multicentre trial by Vignon and colleagues, published in American Journal of Respiratory and Critical Care Medicine (AJRCCM), enrolled the largest population of patients with acute circulatory failure using echocardiography to assess FR in 540 mechanical ventilated subjects (13). Echocardiography was performed within the first 24 hours of ICU admission in 78% of patients. The following echocardiographic indexes of FR were measured: ΔSVC, ΔIVC, respiratory variations of ΔVmaxAo, and PPVs, with patients in the semi-recumbent position. FR was assessed with a passive leg raising (PLR) manoeuvre, which mimics a preload challenge of around 300 mL of blood and induces a significant change in right and left cardiac preload. An increase of VTI ≥10% was defined as a positive response (i.e., a responder patient). In addition, VTI and LVOT diameter were used to calculate SV. The areas under the receiver operating characteristic (ROC) curve to predict FR were 0.755, 0.752, 0.675, and 0.635, for ΔSVC, ΔVmaxAo, PPV, and, ΔIVC, respectively. Importantly, in all subgroups of patients, ΔSVC always showed an AUC greater than ΔIVC. These figures call into question the “totemic status” that ΔIVC has acquired in recent years (14). Indeed, these ΔIVC results perfectly replicate those obtained using central venous pressure (CVP) to predict FR [CVP showed an area under the ROC curve of 0.56 (15)]. In light of this, the usefulness of ΔIVC to predict FR in mechanically ventilated patients has been queried by several authors (15,16). ΔSVC therefore seems more suitable than ΔIVC as a means to predict FR. However, its measurement requires TEE.
Vignon and colleagues reported that only 15.5% of patients fulfilled the ideal pathophysiological and clinical conditions to assess FR (13)—a finding that applies to all uses of PPV, SPV, and SVV in the ICU (13). It has been shown that only about 2% of critically ill ICU patients match the ideal conditions necessary for the assessment of FR using dynamic parameters (17).
In the aforementioned study, only 229 (42%) of patients were classified as “fluid responders” after a PLR test. This result is in line with the FENICE study in which 50% of patients were classified as responders after fluid loading (18). Importantly, it should be noted that a number of patients do not respond to fluid administration, even if the dynamic indices (i.e., significant changes of PPV or SVV) suggest they should. However, this does not mean that the subjects’ CO will not increase after a fluid challenge. It simply means that they fall into a PPV value “grey zone” (19). Currently, critically ill patients with a PPV ranging between 9–12% have a 50% probability of being a fluid responder. This figure ranges from 25% to 62% in the operating room and the ICU, respectively (19,20).
Notably, in the study by Vignon, 470 patients (87%) were receiving catecholamines at the time of echocardiographic assessment, and the average administered volume during the preceding 24 hours was 2.5±2.7 litres. Thus, hypothesizing a mean patient weight of 70 kg, the studied population would have received, on average, about 38.6 mL/kg/h in the first 24 hours. Unfortunately, it has been demonstrated that the majority of hypotensive septic shock patients do not respond to fluid administration (21). Despite its listing in the Surviving Sepsis Campaign guidelines, there is no clear evidence that substantial (30 mL/kg) fluid resuscitation reliably improves end-organ perfusion in septic patients. In light of this, the early use of norepinephrine, as in the study by Vignon, suggests the timely use of intravenous vasopressors should be considered as a feasible alternative to haemodynamic optimisation in septic patients (13).
As previously stated, the term “fluid responder” refers to patients who increase their baseline CO by more than 10% after volume loading (5). If CO will not increase by more than 10%, this indicates that the patient’s heart is probably working on the flat portion of the Frank-Starling curve.
In light of the above, echocardiography could hence be used to assess FR on the basis of CO changes. However, there are conflicting opinions regarding the use of echocardiography as a reference tool for the measurement of CO (13,22). While not uncontroversial (23), the pulmonary artery catheter (PAC) is still considered the gold standard technique for assessing CO by means of the classic thermodilution (ThD) method (6). However, the ThD method does not provide SV on a beat-by-beat basis. Thus, its usefulness in assessing FR is very limited, unless the PAC is coupled to a calibrated pulse contour analysis device (e.g., transpulmonary ThD method). Finally, its invasiveness, the occurrence of complications, and inability to estimate CO on a beat-by-beat basis may explain the reduction in routine use of PAC.
In ICU patients, lung ultrasound evaluation provides further insights into pulmonary oedema and offers indirect information on likely systemic fluid overload. Volpicelli et al. showed that lung B-line artefacts, which reflect the loss of lung aerations, seem to be correlated with the amount of extra vascular lung water (EVLW) (24). Knowing the number of such artefacts would therefore allow physicians to administer fluids safely. Indeed, an increased number of lung B-line artefacts is an important warning to physicians to stop giving fluids, be fluid restrictive, or to switch to another therapeutic option (e.g., vasopressors, vasodilators, inotropics, diuretics) to increase organ perfusion. Based on this assumption, Lichtenstein described a protocol for fluid administration guided by lung sonography (25).
In conclusion, echocardiography is a useful tool for the intensivist to assess either cardiac function or FR in the majority of ICU patients. Unfortunately, a certain number of critically ill patients do not fulfil the criteria to have FR assessed with standard haemodynamic monitoring (i.e., with PPV, SVV, and SPV). In this context, increased attention has been given to transthoracic lung ultrasonography, which may allow clinicians to obtain additional information regarding the effect of fluid loading on haemodynamics, efficacy and tolerance of therapeutic changes.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Vincent JL, Rhodes A, Perel A, et al. Clinical review: Update on hemodynamic monitoring—a consensus of 16. Crit Care 2011;15:229. [Crossref] [PubMed]
- Vincent JL, Pelosi P, Pearse R, et al. Perioperative cardiovascular monitoring of high-risk patients: a consensus of 12. Crit Care 2015;19:224. [Crossref] [PubMed]
- Marik PE, Cavallazzi R. Does the central venous pressure predict fluid responsiveness? An updated meta-analysis and a plea for some common sense. Crit Care Med 2013;41:1774-81. [Crossref] [PubMed]
- Expert Round Table on Ultrasound in ICU. International expert statement on training standards for critical care ultrasonography. Intensive Care Med 2011;37:1077-83. [Crossref] [PubMed]
- Kanji HD, McCallum J, Sirounis D, et al. Limited echocardiography-guided therapy in subacute shock is associated with change in management and improved outcomes. J Crit Care 2014;29:700-5. [Crossref] [PubMed]
- Scolletta S, Franchi F, Romagnoli S, et al. Comparison Between Doppler-Echocardiography and Uncalibrated Pulse Contour Method for Cardiac Output Measurement: A Multicenter Observational Study. Crit Care Med 2016;44:1370-9. [Crossref] [PubMed]
- Beaulieu Y, Marik PE. Bedside ultrasonography in the ICU: part 1. Chest 2005;128:881-95. [Crossref] [PubMed]
- Boussuges A, Blanc P, Molenat F, et al. Evaluation of left ventricular filling pressure by transthoracic Doppler echocardiography in the intensive care unit. Crit Care Med 2002;30:362-7. [Crossref] [PubMed]
- Feissel M, Michard F, Mangin I, et al. Respiratory changes in aortic blood velocity as an indicator of fluid responsiveness in ventilated patients with septic shock. Chest 2001;119:867-73. [Crossref] [PubMed]
- Slama M, Masson H, Teboul JL, et al. Respiratory variations of aortic VTI: a new index of hypovolemia and fluid responsiveness. Am J Physiol Heart Circ Physiol 2002;283:H1729-33. [Crossref] [PubMed]
- Vieillard-Baron A, Chergui K, Rabiller A, et al. Superior vena caval collapsibility as a gauge of volume status in ventilated septic patients. Intensive Care Med 2004;30:1734-9. [Crossref] [PubMed]
- Barbier C, Loubières Y, Schmit C, et al. Respiratory changes in inferior vena cava diameter are helpful in predicting fluid responsiveness in ventilated septic patients. Intensive Care Med 2004;30:1740-6. [Crossref] [PubMed]
- Vignon P, Repessé X, Bégot E, et al. Comparison of Echocardiographic Indices Used to Predict Fluid Responsiveness in Ventilated Patients. Am J Respir Crit Care Med 2016. [Epub ahead of print]. [Crossref] [PubMed]
- Hilton AK, Bellomo R. Totem and taboo: fluids in sepsis. Crit Care 2011;15:164. [Crossref] [PubMed]
- Marik PE, Baram M, Vahid B. Does central venous pressure predict fluid responsiveness? A systematic review of the literature and the tale of seven mares. Chest 2008;134:172-8. [Crossref] [PubMed]
- Via G, Tavazzi G, Price S. Ten situations where inferior vena cava ultrasound may fail to accurately predict fluid responsiveness: a physiologically based point of view. Intensive Care Med 2016;42:1164-7. [Crossref] [PubMed]
- Mahjoub Y, Lejeune V, Muller L, et al. Evaluation of pulse pressure variation validity criteria in critically ill patients: a prospective observational multicentre point-prevalence study. Br J Anaesth 2014;112:681-5. [Crossref] [PubMed]
- Cecconi M, Hofer C, Teboul JL, et al. Fluid challenges in intensive care: the FENICE study: A global inception cohort study. Intensive Care Med 2015;41:1529-37. [Crossref] [PubMed]
- Cannesson M, Le Manach Y, Hofer CK, et al. Assessing the diagnostic accuracy of pulse pressure variations for the prediction of fluid responsiveness: a “gray zone” approach. Anesthesiology 2011;115:231-41. [Crossref] [PubMed]
- Biais M, Ehrmann S, Mari A, et al. Clinical relevance of pulse pressure variations for predicting fluid responsiveness in mechanically ventilated intensive care unit patients: the grey zone approach. Crit Care 2014;18:587. [Crossref] [PubMed]
- Monge García MI, Guijo González P, Gracia Romero M, et al. Effects of fluid administration on arterial load in septic shock patients. Intensive Care Med 2015;41:1247-55. [Crossref] [PubMed]
- Wetterslev M, Møller-Sørensen H, Johansen RR, et al. Systematic review of cardiac output measurements by echocardiography vs. thermodilution: the techniques are not interchangeable. Intensive Care Med 2016;42:1223-33. [Crossref] [PubMed]
- Yang XX, Critchley LA, Joynt GM. Determination of the precision error of the pulmonary artery thermodilution catheter using an in vitro continuous flow test rig. Anesth Analg 2011;112:70-7. [Crossref] [PubMed]
- Volpicelli G, Skurzak S, Boero E, et al. Lung ultrasound predicts well extravascular lung water but is of limited usefulness in the prediction of wedge pressure. Anesthesiology 2014;121:320-7. [Crossref] [PubMed]
- Lichtenstein D. FALLS-protocol: lung ultrasound in hemodynamic assessment of shock. Heart Lung Vessel 2013;5:142-7. [PubMed]


