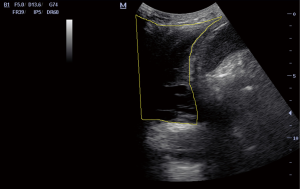
Correlation of pleural effusions’ grayscale sonographic parameters with fluid’s analysis results
Introduction
Until now, in the sonographic imaging of pleural effusions, the quantitative evaluation of the fluids characteristics was performed indirectly by using other anatomic structures of the thorax as a relative measure of comparison (1). Such quantitative methods are used to assess the fluid’s volume but to the best of our knowledge no method exists for the measurement of the fluids’ density and other qualitative values. The interpretation of the internal echogenicity of a pleural effusion is based upon the subjective observation and evaluation of each operator. According to the internal echogenicity, effusion can be subclassified as anechoic, complex non-septated, complex septated, and homogenously echogenic (2). Additionally, several visual patterns like swirling (3) or sonographic septations (4,5) have been proven useful as diagnostic predictors for pleural effusions (6). We suggest a quantitative method, based on the pixel density of the pleural effusion’s image, in order to evaluate the echogenicity of pleural effusion.
Methods
Pleural ultrasound (US) was performed prospectively in 62 patients with newly diagnosed pleural effusion. The patient demographics are shown in Table 1. All patients signed informed consent. Ethics approval was obtained from our institution (ID: 3648-27/05/14). Five consequent images of the pleural effusion were retrieved through axial view between the 9th and the 10th rib and one from the 10th rib through coronal view and converted into the high-resolution tagged image file format. We used a curvilinear probe (2.5–5 MHz) and the ultrasound settings were: Dynamic range 60 dBs and Depth 8 cm for all the patients. All images were transferred uncompressed to a computer and further processed with a widespread imaging analysis program, available for downloading from the public domain (Image J, 1.42q; National Institutes of Health, Bethesda, MD; http://rsb.info.nih.gov/ij). Pleural effusion echogenicity was measured with histogram analysis. Tissue echo levels were automatically calibrated to the value of 255 for the white pixels and 0 for the black pixels (Figure 1). The mean echo levels of all pixels of the pleural effusion and of the 10th rib were counted, and the hypoechogenicity index (HI) was calculated according to the following formula: HI = mean echo level of all pixels of the rib/mean echo levels of all pixels of pleural effusion. HI greater than 1 indicates pleural effusion’s hypoechogenicity. The HI calculation required less than a minute for every image taken. Diagnostic thoracocentesis was performed in all patients and biochemical markers were measured.
Full table
Statistical analysis
Data were expressed as mean ± standard deviation (SD) for continuous variables and percentages when reporting categorical variables. Normality was assessed via the one-sample Kolmogorov-Smirnov Test. Relationships were assessed via Pearson’s R or Spearman’s ρ correlation coefficients, where appropriate. Associations were assessed via simple linear regression; Curve estimation/Nonlinear regression was subsequently employed to determine the model that best fits the data. For all tests, a P value ≤0.05 was considered statistically significant. IBM SPSS 20.0 (IBM Corporation, San Diego, CA, USA).
Results
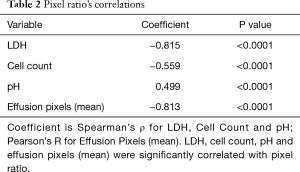
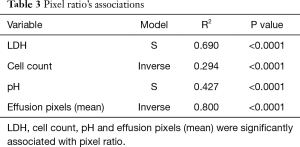
Pixel ratio’s correlations are presented on Table 2. As LDH, Cell Count and pH were not normally distributed, Spearman’s ρ was used to assess their relationship with pixel ratio. As displayed in Table 2, LDH, Cell Count, pH and Effusion Pixels (Mean) were significantly correlated with pixel ratio. As displayed in Table 3, LDH, Cell Count, pH and Effusion Pixels (Mean) were also significantly associated with pixel ratio. Specific nonlinear regression models are displayed in Table 3, as selected by a P value ≤0.05 and maximum R2 compared to the linear level. Conversely, pixel ratio was not correlated with any other thoracocentesis—derived parameter or biomarker.
Full table
Full table
Discussion
To the best of our knowledge, no quantitative method is being used in lung ultrasound imaging. We introduce an experimental method, based on the pixel density of the pleural effusion’s sonographic image. We employed the hypoechogenicity index, which has been widely used in ultrasound studies of the thyroid and its diseases but not in the lung (7). To achieve standardization in our method we used the same ultrasound depth and dynamic range for all our patients. To compensate for the variability of the gain we used the HI, a fraction of two pixel counts. Additionally, none of the patients had a history of osteoporosis, which could have affected the density of the rib. For better standardization of our method we used the same anatomic viewpoints for all our ultrasound images and 5 consequent images were taken to depict the dynamic movement of the fluid during the breathing cycle.
This method has also limitations regarding the margin of error in calculating the pixels near the solid surfaces due the low resolution. Additionally, obesity is another matter we have to take into consideration, since adipose tissue causes sound attenuation and can influence the depth and limit the observing ultrasound window. In our study, all of our patients had a body mass index below 30, nevertheless the HI index is a fraction and the needed adjustments of the ultrasound’s gain affect equally both parts of this fraction. This minimizes errors in measurement due to excess adipose tissue or pleural thickening (8).
This study demonstrated HI as new index, which could indicate the inflammation density of pleural effusions. Moreover, when used in combination with classical biomarkers, such as fluid LDH and cell counts, HI might be a useful adjunct for the discrimination of pleural transudate. This objective quantitative method could be integrated in the ultrasound’s software for more accurate measurements. More studies are needed in order to further investigate this quantitative method.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statements: All patients signed informed consent. Ethics approval was obtained from our institution (ID: 3648-27/05/14).
References
- Vignon P, Chastagner C, Berkane V, et al. Quantitative assessment of pleural effusion in critically ill patients by means of ultrasonography. Crit Care Med 2005;33:1757-63. [Crossref] [PubMed]
- Yang PC, Luh KT, Chang DB, et al. Value of sonography in determining the nature of pleural effusion: analysis of 320 cases. AJR Am J Roentgenol 1992;159:29-33. [Crossref] [PubMed]
- Chian CF, Su WL, Soh LH, et al. Echogenic swirling pattern as a predictor of malignant pleural effusions in patients with malignancies. Chest 2004;126:129-34. [Crossref] [PubMed]
- Chen HJ, Hsu WH, Tu CY, et al. Sonographic septation in lymphocyte-rich exudative pleural effusions: a useful diagnostic predictor for tuberculosis. J Ultrasound Med 2006;25:857-63. [Crossref] [PubMed]
- Shankar S, Gulati M, Kang M, et al. Image-guided percutaneous drainage of thoracic empyema: can sonography predict the outcome? Eur Radiol 2000;10:495-9. [Crossref] [PubMed]
- Bugalho A, Ferreira D, Dias SS, et al. The diagnostic value of transthoracic ultrasonographic features in predicting malignancy in undiagnosed pleural effusions: a prospective observational study. Respiration 2014;87:270-8. [Crossref] [PubMed]
- Banaka I, Kaltsas G, Antoniou S, et al. Prognostic value of vascularity index for the diagnosis of autoimmune thyroid disease. JBR-BTR 2011;94:185-90. [PubMed]
- Modica MJ, Kanal KM, Gunn ML. The obese emergency patient: imaging challenges and solutions. Radiographics 2011;31:811-23. [Crossref] [PubMed]