Reconstruction of mediastinal vessels for invasive thymoma: a retrospective analysis of 25 cases
Introduction
Thymoma, the most common primary mediastinal tumor, is a kind of slow-growing neoplasm with thymic epithelial cell origin and represents less possibility of hematogenous spread and regional lymph nodes metastasis (1,2). Although it constitutes nearly 20–50% of all mediastinal tumors in adults, the morbidity of thymoma is extremely low (3). However, according to World Health Organization (WHO) and the International Thymic Malignancy Interest Group (ITMIG), invasive thymoma is different from thymoma in biological characteristics, which is characterized by infiltrative growth and invasion into neighboring structures (4,5).
Currently, multidisciplinary treatment including surgery, radiation therapy and chemotherapy is often adopted in advanced thymoma or invasive thymoma (2). Among all the surgery options, complete resection, along with vessel replacement and reconstruction, has been proved to be the best option (6). Even though most prosthetic replacements were operated in patients suffered from advanced lung cancer, it still remains an important place in treating patients with invasive thymoma (7).
Despite that many researchers have presented their clinical experiences of mediastinal vascular prosthesis replacement, to our knowledge, the cases reported in literature remain scarce (8-11). Moreover, vessel reconstruction caused by primary lung cancer rather than invasive thymoma was commonly found in previous reports (6,12,13). Thus, we reviewed our experience with reconstruction of mediastinal vessels for invasive thymoma. It is the first time to research and report vessel reconstruction for invasive thymoma based on such number of data.
Methods
Population
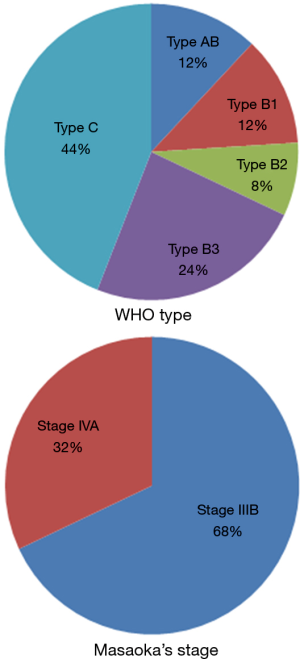
From April 2007 to August 2015, 28 consecutive cases underwent vessel reconstruction due to invasive thymomas in the department of thoracic surgery of Shanghai Chest Hospital, Shanghai Jiao Tong University. For better homogeneity of patient group, one case with arterial reconstruction, as well as another two cases with pneumonectomy was excluded. The remaining 25 cases were subjected to the study. All the patients had a thorough surgical risk evaluation and accurate diagnosis of primary affection. Preoperative examinations included physical examination, serological examination, chest X-ray, chest computed tomography (CT) scan or contrast-enhanced CT, abdominal CT scan, brain CT scan, bone scan and transabdominal ultrasound. PET-CT is also an option for excluding distant metastasis. Contrast-enhanced CT (Figure 1) displayed an important role in preoperative diagnosis. The Masaoka’s staging of 25 patients was: stage IIIB, 17; stage IVA, 8 (Figure 2).
A consent form was signed by each patient or their legal representatives. After obtaining the approval of institutional review board, the files for all patients were reviewed to collect corresponding clinicopathologic data including symptom, sex, age, smoking history, pathologic stage (WHO type and Masaoka’s stage) and treatment history. Follow-up (disease recurrence and survival) was obtained by outpatient visits or telephone.
Surgical management
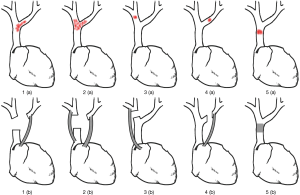
After general anesthesia, the patient was placed in the supine position and then median sternotomy was performed. Afterwards, the internal mammary artery was ligated and sternum spreader was used, allowing the surgeon to explore the tumor location, infiltrating situation and intrathoracic metastasis and to have an easy access to mediastinal great vessels. Radical resection of tumor along with other involved neighboring structures was undertaken. If not involved, the phrenic nerve should be carefully preserved. With regarding to the involved vessels, when the involvement was less than 30% circumference or no more than 2 cm in length, the vessels could be sutured directly or fixed by patches. Otherwise, vessel reconstruction should be performed. Potts vascular clamp and auricular clamp were used to block bleeding in reconstruction procedure. The central venous pressure and facial swelling should be monitored by anesthetists. If needed, an external shunt could provide a favorable condition for vessel reconstruction. Before the blocking of involved vessels, local or systemic heparinization should be necessary. Based on the tumor location, the methods used for vessel reconstruction varies from person to person (Figure 3). Subsequently, thoracic washing was commonly performed after hemostasis. A re-exploration was necessary for ensuring no active hemorrhage and then two or three chest drainage tubes were inserted before closing the incision.
In the 25 cases, all the vessels involved were reconstructed with ringed polytetrafluoroethylene (PTFE) grafts. Furthermore, before prosthetic replacement, the ringed PTFE grafts were soaked in heparin-salt solution to reduce the risk of thrombosis.
Postoperative complications and graft patency
Adverse events occurring in hospital or within 30 days after surgery were defined as postoperative complications. Graft patency was accessed by postoperative CT.
Statistical analysis
All the clinicopathologic data were analyzed using SPSS 19.0 software package (SPSS Inc., Chicago, IL). The survival curves including relapse-free survival (RFS) and overall survival (OS) were established by Prism 5 (Graph Pad Software Inc., La Jolla, CA), as well as the 3- and 5-year survival rates.
Results
A total of 25 patients underwent reconstruction of mediastinal vessels, including 17 (68%) men and 8 (32%) women, ranging in age from 28 to 75 years (median, 52.4 years). Nineteen patients were admitted with the problem of breathlessness, cough, drooping eyelid or neck and facial edema while six were detected incidentally in routine health examination. The average postoperative hospital stay and ICU stay were 12.5 and 4.7 days, respectively (Table 1).
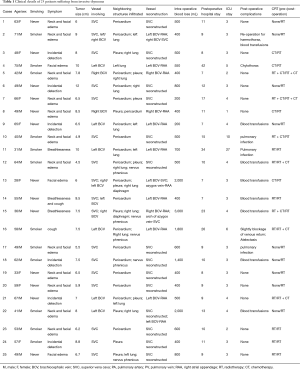
Full table
Surgical procedure
Twenty of 25 patients (80%) underwent R0 resection of the tumor and involved neighboring structures. The average intra-operative blood loss was 944 mL. Concerning vessel reconstruction, 13 patients underwent superior vena cava (SVC)-SVC reconstruction, 11 patients underwent brachiocephalic vein (BCV)-right atrial appendage (RAA) reconstruction and 1 patient underwent complex vessel reconstruction (more than one graft) (Figure 3). The mean clamping time was 16 minutes and no patient needed external shunt.
As for infiltrated adjacent structures, there were 6 cases with pulmonary wedge resection, 7 cases with lobectomy, 19 cases with pericardiectomy, 10 cases with pleurectomy, 4 cases with phrenicectomy, and 2 cases with diaphragm patching, respectively.
Postoperative complications and graft patency
The overall postoperative complication rate was 52% (including postoperative blood transfusion) due to the complexity of vessel reconstruction and radical tumor resection. Three cases suffered from pulmonary infection. Besides, chylothorax and atelectasis occurred in one case respectively, leading to long-term hospitalization (P<0.001).
One patient needed a second operation for haemothorax. Because of the large haemorrhage amount during operation, eight patients (32%) required blood transfusions.
Postoperative CT showed all the grafts had good patency except two patients, who had irregular use of warfarin.
Survival analysis
Neither WHO type nor Masaoka’s stage was revealed as a significant predictor in the univariable analysis.
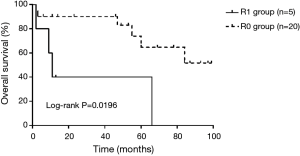
During the follow-up, two patients died for acute respiratory distress syndrome in 2 and 3 months after the surgery, respectively. Within the remaining patients, 11 cases (44%) experienced a relapse and finally 8 (32%) patients died (Figure 4). Compared to R1 resection group, R0 resection group had a better prognosis (Log-rank P=0.0196) (Figure 5). Local recurrence, as the most common site of first recurrence, was observed in seven patients. Besides, 5 patients had distant metastases at the first time of recurrence (4 cases in the lung and 1 case in the bone). The 3- and 5-year survival rates were 79.6% and 59.1%, with median survival time of 84 months.

Discussion
It has been observed that 20–30% of thymomas are malignant, which commonly invade neighboring structures such as pleura (30%), pericardium (25%) and lung (8%), whereas rarely involve mediastinal great vessels (1,14). SVC syndrome is commonly related to advanced malignancy. Many cases with SVC syndrome caused by invasive thymoma were found in previous reports (10,11,15). All the 25 cases in our series were advanced WHO type (Stage III/IV). Similar to previous reports, most of our patients suffered from SVC syndrome such as neck and facial edema, drooping eyelid and dyspnea, while 6 patients seemed asymptomatic preoperatively.
Radical resection of the tumor and adjacent involved structures is indispensable for better prognosis (16). Therefore, in order to obtain long-term survival for patients, it is required to reconstruct mediastinal vessels involved by invasive thymoma. The operation indication for prosthetic graft replacement is when the involvement is more than 30% circumference or half of the circumference (at least 2 cm in length) and all the patients met the requirement in our study (13,17). In our study, when compared to R1 resection group, R0 resection group had a better prognosis (Log-rank P=0.0196). Even though some studies emphasized cardiopulmonary bypass (CPB) should be necessary in some cases when en bloc resection can be completed, none of our patients required it (2). During the surgery, Arvind et al. (7) reported, in their cases, the average blood loss was 400 mL, which was smaller than ours (944 mL). The main reason maybe our operations with various surgical methods provided a lot more complexity.
A variety of materials, such as autologous or bovine jugular vein, Dacron and PTFE, have been reported to be used in reconstruction of vessels, which caused lots of debate (18). Grafts made by autologous or bovine jugular vein have produced encouraging clinical results in SVC reconstruction, but without support, pericardial grafts would develop stenosis (19,20). Many surgeons are in favor of using autologous vein such as saphenous vein as an ideal graft for reconstruction (21). However, the process of preparation is time-consuming and more sutures used in surgery may result in thrombosis. A high risk of thrombosis cannot be avoided even using bovine jugular vein instead (22). Chen and his colleagues (17) have used Dacron conduits to perform SVC replacement and acquired promising results. Similar outcome has been proved by Amirghofran et al. (23). Nevertheless, PTFE grafts are still widely used for the advantages of availability in caliber and length with long-term patency and excellent clinical results (24-26). In our series, all the grafts we used were ringed PTFE conduits for vessel reconstruction. Twenty-three patients (92%) had good graft patency during follow-up period.
Regarding to postoperative complications, the risk of prosthetic thrombosis did exist. Thus, surgeons need to soak the ringed PTFE grafts in heparin-salt solution before implanting and controlling perioperative international normalized ratio (INR) of patients, so that the thrombotic risk is minimized. As for cerebral damage due to the backflow obstruction, the SVC should be clamped above the level of azygos vein when the SVC clamping is needed during operation, while a SVC bypass is considered if the clamping was below azygos vein or required longer surgical time. In addition, much more attention also should be taken to avoid such intraoperative injury as nerve or artery injuries. In our series, the mean clamping time was 16 minutes and no patient needed external shunt.
Despite surgical resection acts as mainstay of treatment for thymoma, radiotherapy still plays an important role in treating invasive thymoma in terms of reducing local recurrence and prolonging survival while chemotherapy controls pleural and distant metastasis (27). Recently, to patients with advanced invasive thymoma, multidisciplinary treatment including radical resection, pre- or post-operative chemotherapy and radiotherapy, has been considered effective (28). In our study, postoperatively, all the patients underwent routinely prophylactic radiotherapy, but chemotherapy or preoperative radiotherapy depends on their individual situations (chemotherapy criteria: locally advanced thymoma; solitary metastasis or ipsilateral pleural metastasis; extrathoracic metastasis. Preoperative radiotherapy criteria: unresectable disease; locally advanced thymoma).
Overall, apart from 2 cases died from acute respiratory distress syndrome, 11 patients (44%) relapsed, and among them, 8 died during the follow-up. The 3- and 5-year survival rates were 79.6% and 59.1%, respectively, with median survival time of 84 months. Ogawa and his colleagues (29) have demonstrated that if no surgery taken, radiotherapy only resulted in 44% 5-year survival rate. Another study revealed that the 5- and 10-year survival rates were 53% and 44% if patients with stage III thymoma only received radiotherapy (30). It appears that among patients with advanced thymoma, complete resection with preoperative radiotherapy is much better than radiotherapy only.
There are several limitations in this study. First, a small sample size was taken in this study, although we reported a rare disease invasive thymoma in such a number. Second, some patients had their adjuvant radiochemotherapy in their local hospitals because of economic reasons, and the specific postoperative treatments were not available.
In conclusion, our experiences have proven that reconstruction of mediastinal vessels for invasive thymoma is a feasible technology method. Radical resection of the tumor with involved neighboring structures is the key to prolong OS in patients suffered from invasive thymoma.
Acknowledgements
Funding: This work was supported by grants from special funds of science and technology innovation of Shanghai Jiao Tong University (No.YG2015MS71) and Shanghai municipal commission of health and family planning (No. 2013ZYJB0004, No. 201540139).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by institutional ethics committee of Shanghai Chest Hospital, Shanghai Jiao Tong University [No. KS (P1701)]. Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Cohen DJ, Ronnigen LD, Graeber GM, et al. Management of patients with malignant thymoma. J Thorac Cardiovasc Surg 1984;87:301-7. [PubMed]
- Pastorino U, Yang XN, Francese M, et al. Long-term survival after salvage surgery for invasive thymoma with intracardiac extension. Tumori 2008;94:772-6. [PubMed]
- Engels EA, Pfeiffer RM. Malignant thymoma in the United States: demographic patterns in incidence and associations with subsequent malignancies. Int J Cancer 2003;105:546-51. [Crossref] [PubMed]
- Travis WD, Brambilla E, Müller-Hermelink HK, et al. (editors). Organization Classification of Tumours. Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart. Lyon: IARC Press, 2004:146-7.
- Marx A, Ströbel P, Badve SS, et al. ITMIG consensus statement on the use of the WHO histological classification of thymoma and thymic carcinoma: refined definitions, histological criteria, and reporting. J Thorac Oncol 2014;9:596-611. [Crossref] [PubMed]
- Leo F, Bellini R, Conti B, et al. Superior vena cava resection in thoracic malignancies: does prosthetic replacement pose a higher risk? Eur J Cardiothorac Surg 2010;37:764-9. [Crossref] [PubMed]
- Arvind K, Roman D, Umashankkar K, et al. Resection and reconstruction of mediastinal great vessels in invasive thymoma. Indian J Cancer 2010;47:400-5. [Crossref] [PubMed]
- Dong YQ, Liang JS, Zhang XM, et al. Surgical treatment of an invasive thymoma extending into the superior vena cava and right atrium. World J Surg Oncol 2014;12:6. [Crossref] [PubMed]
- Funakoshi Y, Ohta M, Maeda H, et al. Extended operation for invasive thymoma with intracaval and intracardiac extension. Eur J Cardiothorac Surg 2003;24:331-3. [Crossref] [PubMed]
- Konstantinov IE, Saxena P, Koniuszko M, et al. Superior vena cava obstruction by tumour thrombus in invasive thymoma: diagnosis and surgical management. Heart Lung Circ 2007;16:462-4. [Crossref] [PubMed]
- Rosa GR, Takizawa N, Schimidt D, et al. Surgical treatment of superior vena cava syndrome caused by invasive thymoma. Rev Bras Cir Cardiovasc 2010;25:257-60. [Crossref] [PubMed]
- Spaggiari L, Leo F, Veronesi G, et al. Superior vena cava resection for lung and mediastinal malignancies: a single-center experience with 70 cases. Ann Thorac Surg 2007;83:223-9; discussion 229-30. [Crossref] [PubMed]
- Politi L, Crisci C, Montinaro F, et al. Prosthetic replacement and tangential resection of the superior vena cava in chest tumors. J Cardiovasc Surg (Torino) 2007;48:363-8. [PubMed]
- Large SR, Shneerson JM, Stovin PG, et al. Surgical pathology of the thymus: 20 years’ experience. Thorax 1986;41:51-4. [Crossref] [PubMed]
- Kamikubo Y, Shiiya N, Kubota S, et al. Thymic carcinoma with tumor thrombus into the superior vena cava. Jpn J Thorac Cardiovasc Surg 2001;49:327-9. [Crossref] [PubMed]
- Yagi K, Hirata T, Fukuse T, et al. Surgical treatment for invasive thymoma, especially when the superior vena cava is invaded. Ann Thorac Surg 1996;61:521-4. [Crossref] [PubMed]
- Chen KN, Xu SF, Gu ZD, et al. Surgical treatment of complex malignant anterior mediastinal tumors invading the superior vena cava. World J Surg 2006;30:162-70. [Crossref] [PubMed]
- Spaggiari L, Magdeleinat P, Kondo H, et al. Results of superior vena cava resection for lung cancer. Analysis of prognostic factors. Lung Cancer 2004;44:339-46. [Crossref] [PubMed]
- Warren WH, Piccione WJ Jr, Faber LP. As originally published in 1990: Superior vena caval reconstruction using autologous pericardium. Updated in 1998. Ann Thorac Surg 1998;66:291-2; discussion 292-3. [Crossref] [PubMed]
- Lü WD, Yu FL, Wu ZS. Superior vena cava reconstruction using bovine jugular vein conduit. Eur J Cardiothorac Surg 2007;32:816-7. [Crossref] [PubMed]
- Doty JR, Flores JH, Doty DB. Superior vena cava obstruction: bypass using spiral vein graft. Ann Thorac Surg 1999;67:1111-6. [Crossref] [PubMed]
- Schoof PH, Koch AD, Hazekamp MG, et al. Bovine jugular vein thrombosis in the Fontan circulation. J Thorac Cardiovasc Surg 2002;124:1038-40. [Crossref] [PubMed]
- Amirghofran AA, Emaminia A, Rayatpisheh S, et al. Intracardiac invasive thymoma presenting as superior vena cava syndrome. Ann Thorac Surg 2009;87:1616-8. [Crossref] [PubMed]
- Van Putten JW, Schlosser NJ, Vujaskovic Z, et al. Superior vena cava obstruction caused by radiation induced venous fibrosis. Thorax 2000;55:245-6. [Crossref] [PubMed]
- Spaggiari L, Thomas P, Magdeleinat P, et al. Superior vena cava resection with prosthetic replacement for non-small cell lung cancer: long-term results of a multicentric study. Eur J Cardiothorac Surg 2002;21:1080-6. [Crossref] [PubMed]
- Garcia-Rinaldi R, Zamora JL, Torres-Salichs M, et al. Four-year patency of PTFE grafts after replacement of the superior vena cava and the innominate veins. Tex Heart Inst J 1988;15:192-4. [PubMed]
- Nonaka T, Tamaki Y, Higuchi K, et al. The role of radiotherapy for thymic carcinoma. Jpn J Clin Oncol 2004;34:722-6. [Crossref] [PubMed]
- Yokoi K, Miyazawa N, Mori K, et al. Invasive thymoma with intracaval growth into the right atrium. Ann Thorac Surg 1992;53:507-9. [Crossref] [PubMed]
- Ogawa K, Toita T, Uno T, et al. Treatment and prognosis of thymic carcinoma: a retrospective analysis of 40 cases. Cancer 2002;94:3115-9. [Crossref] [PubMed]
- Graeber GM, Tamim W. Current status of the diagnosis and treatment of thymoma. Semin Thorac Cardiovasc Surg 2000;12:268-77. [Crossref] [PubMed]