A non-randomized retrospective observational study on the subcutaneous esophageal reconstruction after esophagectomy: is it feasible in high-risk patients?
Introduction
Esophagectomy and esophageal reconstruction is a complex invasive procedure with high morbidity (40%) and mortality (5–10%) rates (1,2). According to previous studies, the presence of comorbidities such as old age, hypertension, diabetes, smoking status, congestive heart failure, coronary artery disease, poor preoperative pulmonary function, and high preoperative serum creatinine level worsens the clinical outcomes including anastomotic leakage, pulmonary complications, and overall survival (2-7). Therefore, additional consideration for lowering the operative morbidity and mortality is required for those patients with comorbidities.
Among the multiple options for the placement of the conduit during esophageal reconstruction, the posterior mediastinal route is most commonly used because of its lower rates of postoperative morbidity and mortality compared to other routes (8,9). On the other hand, esophageal reconstruction through the subcutaneous route is rarely performed as a primary surgical strategy because of its poor cosmetic appearance. It is usually considered as an option only after failure of previous esophageal reconstruction (10-12).
The reported rates of anastomotic leakage after cervical anastomosis are between 9% and 25% (13,14). Most cases of anastomotic leakage are easily controlled by simple drainage and conservative managements. However, while some cases may be asymptomatic or present with only local signs, other cases may present with respiratory symptoms due to pleural or mediastinal collections, or systemic septic conditions secondary to gastric necrosis (14,15). Major cervical anastomotic leakage, including that combined with graft necrosis, is considered an important predictor of subsequent death (15). Additionally, cervical anastomotic leakage may prolong hospital stay and lead to other complications (13).
The extrathoracic placement of the anastomosis site and the conduit graft in the subcutaneous esophageal reconstruction may prevent the leakage to the mediastinum or pleural cavity in case of anastomotic leakage, and thereby decrease the possibility of severe infection. Furthermore, prompt diagnosis of the graft status and easier correction of the leakage site may be possible because of the closer placement of the graft to the skin. In this aspect, we assumed that it may be a more conservative and safe method for avoiding disastrous conditions after esophageal reconstruction, especially in patients with comorbidities who may have a higher risk of postoperative anastomotic leakage and pulmonary complications.
Thus, to evaluate the safety and feasibility of esophageal reconstruction through the subcutaneous route, we analyzed the perioperative outcomes including the postoperative complications such as anastomotic leakages and also the patients’ postoperative clinical progression.
Methods
This was a retrospective, observational study of patients who underwent esophageal reconstruction through the subcutaneous route at Korea University Medical Center between February 2004 and May 2015. The Institutional Review Board of Korea University Medical Center approved this study (No. AN16343-001).
Inclusion and exclusion criteria
All patients diagnosed as having esophageal cancer and treated with esophageal reconstruction with cervical anastomosis through the subcutaneous route were included in our study. The selection criteria for the use of subcutaneous route were based on the surgeon’s choice depending on the presence of major preoperative comorbidities and the intraoperative findings. Preoperative comorbidities are described in Table 1. Past history of uncontrolled hypertension or diabetes, patient incompliance including chronic alcoholic abuse, heavy smoking, and intraoperative findings such as poor graft blood supply, poor graft color, and extensive tumor invasion requiring postoperative radiotherapy were the main reasons for deciding to perform the esophageal reconstruction through the subcutaneous route.

Full table
Only patients who had esophageal reconstruction through the subcutaneous route as their first operation were included and those who had it after a failure of previous attempt of esophageal reconstruction were excluded from our study considering the possibility of other implications on the outcomes.
Surgical procedure and perioperative care
For subcutaneous esophageal reconstruction, standard esophagectomy and abdominal mobilization of stomach or colon graft through the laparotomy was performed. Then, a left oblique cervical incision along the anterior border of the sternocleidomastoid muscle of about 8 cm with slight extension to the upper manubrium level was performed. The subcutaneous layer between the cervical and laparotomy incision was dissected with extreme caution not to miss any bleeding and also for an even dissection to prevent any possibility of skin necrosis. When a subcutaneous dissection of about 5 cm width was obtained, which enables a tension-free movement of the graft, the graft was pulled up through the subcutaneous route until the upper portion of graft reached the cervical opening where the esophagogastrostomy is later performed. The usual esophageal reconstruction through the posteromediastinal or retrosternal route was performed just like in the subcutaneous route, except that the conduit is passed through the intrathoracic routes. All patients were admitted in the intensive care unit (ICU) for the immediate postoperative period. Cervical anastomosis was monitored clinically, and oral diet was started between the 7th and 14th day postoperatively after confirmation by contrast radiography. The postoperative patients were followed up routinely between 1 and 2 weeks after discharge, 6 months, and yearly postoperatively at the out-patient clinic. And examinations were based on physical examination, esophagogastroduodenoscopy, and chest computed tomography.
Outcome measurements
Patient characteristics such as age, sex, tumor location, grade, histology, and preoperative comorbidities were analyzed. Data on the surgery technique and substitute organ used for reconstruction were obtained retrospectively from medical charts. Additionally, reasons for the application of esophageal reconstruction through the subcutaneous route were also analyzed and evaluated.
To analyze the clinical outcomes, postoperative complications, reoperation, 30 days in-hospital mortality, operative time, length of hospital stay, and ICU stay were evaluated. To evaluate the relative advantages and disadvantages of the subcutaneous technique, we compared the results with our overall results from the posteromediastinal and substernal routes.
Statistics
All data were entered into an Excel spreadsheet (Microsoft, Bellevue, WA, USA). Data were analyzed using IBM SPSS Statistics Version 20 (IBM SPSS Software, Armonk, NY, USA) to determine the perioperative clinical outcomes. Univariate data analysis included t-tests for continuous numerical variables, and Chi-square and Fisher’s exact test for discrete categorical variables. Multivariate analysis included logistic regression. Data are reported as the mean ± standard error of the mean. A P value <0.05 was considered statistically significant.
Results
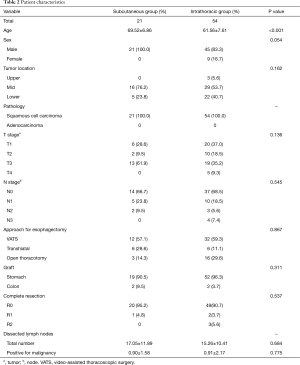
A total of 21 patients were included in this study. All patients underwent esophageal reconstruction with cervical anastomosis through the subcutaneous route. Preoperative patient characteristics including age, sex, pathology, tumor location, TNM staging, and additional surgical factors are described in Table 2.
Full table
The main reason for the selection of the subcutaneous route was the surgeon’s decision considering the intraoperative findings (8/21, 38.1%). Among the 8 patients, the subcutaneous route was selected in 4 (50%) patients because of the poor graft status including edema or unsatisfying color, or concurrent mucosal resection of early gastric cancer with a higher risk of postoperative anastomotic leakages. In the other 4 patients, the subcutaneous route was selected because of extensive tumor progression with suspicious invasion to surrounding tissues such as the main bronchus or enlarged lymph nodes probably requiring postoperative radiotherapy. The rest of the patients (13/21, 61.9%) had serious risk factors such as old age (>70 years), chronic kidney disease, chronic alcoholism with liver cirrhosis, heavy current smokers with emphysematous lung, or stroke history. The mean Charlson comorbidity index score of the subcutaneous group was 6.29±2.12.
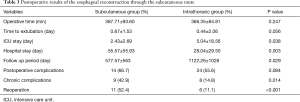
The mean operative time was 387.71±93.60 minutes, and the mean hospital stay was 55.57±55.93 days (Table 3). Among the 21 patients, 13 (61.9%) patients were extubated in the operating room and the other seven (33.3%) patients were extubated on postoperative day 1 without any complication. Only one (4.8%) patient had to go through prolonged ventilation for 7 days in the ICU because of poor pulmonary function. There was no early postoperative in-hospital mortality.
Full table
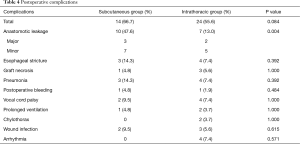
There were 14 (66.7%) patients with postoperative complications (Table 4). Anastomotic leakage (10 cases, 47.6%) was one of the main problems. Three (14.3%) of the anastomotic leakages were major and resulted in chronic cutaneous fistula, requiring lower neck reconstruction using a pectoralis muscle flap and radial forearm fasciocutaneous free flap (RFFF) coverage. The other 7 (33.3%) were minor leakages, among which 5 (23.8%) required reoperation with simple primary repair, and 2 (9.5%) were resolved by spontaneous wound healing after a certain time.
Full table
The mean follow up period of the subcutaneous group was 25.5±26.06 months. Regarding the chronic complications, 9 patients (42.9%) developed chronic complications including 3 (14.3%) cases of esophageal stricture requiring endoscopic balloon dilatation, 2 (9.5%) cases of pneumonia treated with antibiotics, 2 (9.5%) cases of mild dysphagia, 1 (4.8%) case of esophagocutaneous fistula treated with RFFF coverage, and 1 (4.8%) case of vocal cord palsy due to tumor recurrence at the subaortic lymph node which was treated with injection laryngoplasty.
Discussion
Despite favorable trends toward reduced incidence and morbidity of esophageal leaks, esophagogastric anastomotic failure with a reported prevalence of 0.57–53% remains an important source of postoperative morbidity and mortality (16,17). Preoperative comorbidities such as hypertension, diabetes, serum levels of creatinine, old age (>75 years), congestive heart failure, coronary artery disease, peripheral vascular disease, renal insufficiency, and heavy smoking were reported to be strong predictors of major morbidity after esophagectomy (2,5,6). Therefore, when planning for esophagectomy and reconstruction for patients with comorbidities, careful attention should be paid to the possibility of anastomotic failure.
There are multiple options for the placement of the gastric or colon conduit. Among them, the retrosternal and posterior mediastinal route are the most commonly used for the reconstruction after esophagectomy (9). In our study, we focused on patients who had esophageal reconstruction through the subcutaneous route. The main reasons for the selection of the subcutaneous route were the intraoperative findings such as poor blood supply, unsatisfactory graft color, edematous change of the graft, and preoperative comorbidities such as old age (>70 years), hypertension, diabetes, heavy current smoking with emphysematous lung, chronic alcohol abuse with liver cirrhosis, chronic kidney disease, or stroke histories (Table 1). Our decisions were based on the idea that such preoperative and intraoperative findings may act as the risk factors for major morbidities including anastomotic leakages as reported in many previous studies (2,5,6). Therefore, we assumed that this rather less cosmetic and unorthodox method could be more appropriate for high-risk patients in terms of perioperative safety.
The incidence of postoperative anastomotic leakage in our subcutaneous group was 47.6% (10/21 cases). Even though the results coincide with our expectation of higher anastomotic failure in patients with comorbidities, it is still quite unsatisfactory when compared to the results of other previously reported studies (5,13,17,18), or even when compared to the overall anastomotic leakage rate in our department (7/54, 13%, P=0.004). After an analysis of the risk factors between the subcutaneous group and the rest of the patients operated in our department, we have concluded that several factors may have been associated with the high rate of anastomotic failure in the subcutaneous group. First, all patients included in this study were high-risk patients with at least more than one major preoperative comorbidity, probably increasing the possibility of anastomotic leakage and other postoperative complications. A significantly higher proportion of patients with hypertension, heavy smoking, kidney disease, and old age were included in the subcutaneous group compared with the overall esophageal reconstruction patients in our department who had esophageal reconstruction through the intrathoracic route (Table 1). Since the above factors are considered as main risk factors for increasing postoperative comorbidities, this may explain the higher anastomotic leakage rate in our subcutaneous group. When we applied the multivariate logistic regression to the results, the negative intraoperative findings such as poor blood supply to the graft, graft hematoma or edema, and gross tumor invasion to surrounding tissues acted as an independent predictor of overall postoperative complications, while previous history of pneumonia or pulmonary tuberculosis acted as an independent predictor of anastomotic leakage as summarized in Table 5. Second, the cervical anastomosis itself which is considered an important risk factor for increasing the anastomotic leakage rate (14,19), together with the longer distance of the subcutaneous route compared to the posteromediastinal and retrosternal route may have also affected the results. As a result, esophageal reconstruction with cervical anastomosis through the subcutaneous route seems to result in higher anastomotic failures. The higher postoperative leakage rate also increased the duration of hospital stay (55.57±55.93 days), which is significantly longer than the overall hospital stay of the esophageal reconstruction patients in our department (28.04±29.50 days, P=0.003).

Full table
The anastomotic leakages vary in magnitude, and the treatment strategy depends on the severity of the leakage. Previous reports have shown conflicting results regarding the impact of anastomotic leakage on the in-hospital mortality, which seems to depend on the location of the anastomosis. Rutegard et al. reported that the postoperative death rate following intrathoracic anastomotic leakage increased by 3-fold when compared to that without such complication (17). On the other hand, Aminian et al. reported that cervical leakage is not associated with mortality (2). Moreover, the fatality rate from major esophageal leaks was reported to be between 21–35% in many studies (15,20). While some anastomotic leakages can be treated conservatively, others may require immediate reoperation for graft removal and secondary reconstruction (17). Considering the poor nutritional status and the preoperative comorbidities of patients with postoperative anastomotic leakage, they may not be in proper conditions for tolerating additional major reoperations. Furthermore, even though the patients may tolerate the reoperation, such major reoperations may result in increased perioperative risks. So, performing a major reoperation after anastomotic leakage following esophageal reconstruction is quite a challenge not only for the patient but also for the surgeon.
Despite its higher leakage rate, the esophageal reconstruction through the subcutaneous route may have some minor advantages in some aspects. One of the advantages of esophagogastrostomy through the subcutaneous route is that the postoperative monitoring and management of the anastomotic leakage is quite simple and less burdensome to both the patient and the surgeon when compared to the cervical esophagogastrostomy via intrathoracic route. The anastomosis and graft status can be monitored easily postoperatively just by observing and palpating the cervical wound and the subcutaneously tunneled area of the anterior chest wall. If there is any sign of leakage, it can be drained very easily through the cervical wound preventing any further progress of infection. There were 10 patients with cervical anastomotic leakage in our study, but no patient showed any sign or symptom of mediastinitis or sepsis. Consequently, this resulted in zero in-hospital mortality related to postoperative anastomotic leakage. Among the 10 leakage patients, 7 had minor leakages of which 2 were resolved spontaneously just by additionally restraining the oral intake. Five patients required reoperation because of failure of spontaneous healing. However, even in those patients, the exploration of the leakage site and confirmation of the graft status through the cervical wound were very easy when compared with the leaks following the Ivor-Lewis operation or cervical esophagogastrostomy through the posterior mediastinal route, because of the more superficial location of the anastomosis site. After confirmation of the viable tissues including mucosa near the leakage site and the graft status through the cervical wound, the leaks could be managed with a simple interrupted suture repair. This way, the reoperation could be performed in a short operative time and did not require either one-lung ventilation, thoracotomy, or laparotomy which could be a burden to the already weakened patients. As for the result, all five patients were discharged showing sufficient oral intake without any additional complications.
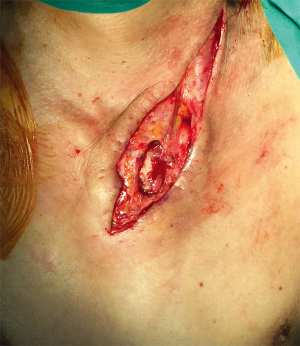
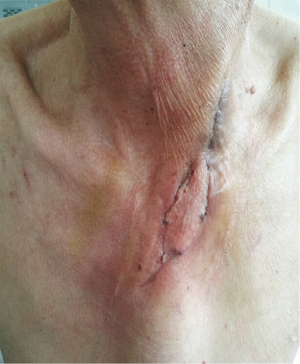
The advantage of subcutaneous esophageal reconstruction was even more remarkable when dealing with the major leakage complications. In the subcutaneous group, there were three patients with major anastomotic leakage which resulted in chronic cutaneous fistula (Figure 1). Unlike the cases of conduit necrosis in the intrathoracic route, we could examine the status of the gastric conduit more easily by exploring the neck wound including the upper anterior chest wall. In all three patients, only the small upper portion of the graft near the anastomosis site was necrotic, and the rest of the graft including the mucosa was viable. Therefore, instead of graft take down and reconstruction using a colon or jejunum graft, debridement of the necrotic site and coverage of the cutaneous fistula at the lower neck using a pectoralis major muscle or RFFF was performed after about 5 to 6 weeks after the esophagectomy (Figures 2,3). All three patients recovered and were discharged with sufficient oral intake without any additional major complications.
Our study has several limitations. First, this study is limited by its retrospective nature and data based on medical chart reviews. Moreover, the heterogeneous preoperative characteristics including the comorbidities between the subcutaneous group and the rest of our patients may have affected both the acute and chronic postoperative progression of the patients, preventing any possibility of direct comparison between the subcutaneous group and the overall results from the rest of our patients. Also, considering that the subcutaneous group had relatively more risk factors, it would be hard to conclude that the high leakage rate resulted mainly due to the subcutaneous reconstruction. Finally, our study included only a small number of cases; therefore, our findings should be interpreted with caution as the generalizability of our results is limited. Further studies with a larger sample size are warranted to confirm the safety and effectiveness of the subcutaneous route in esophageal reconstruction surgery.
In conclusion, patients with serious comorbidities who underwent esophageal reconstruction through the subcutaneous route showed a higher rate of postoperative anastomotic leakages and a longer hospital stay when compared to the overall esophageal reconstruction patients. Given the high anastomotic leakage rate, we have to be careful in choosing the subcutaneous route. However, unlike the leakages from the grafts placed by the intrathoracic route, which may cause sepsis through mediastinitis or require a major reoperation with graft removal and reconstruction with another graft, an easier correction could be obtained by simple primary repair or flap reconstruction resulting in lower perioperative mortality. Therefore, considering the disadvantages such as poor cosmetic aspect and higher risk of anastomotic leakage, we do not recommend esophageal reconstruction through the subcutaneous route as a routine primary option. In highly selected patients with unfavorable preoperative comorbidities or intraoperative findings, especially those with poor blood supply to the graft, graft hematoma or edema, or gross tumor invasion to surrounding tissues, esophageal reconstruction through the subcutaneous route may carefully be considered as an alternative to the conventional surgical techniques.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The Institutional Review Board of Korea University Medical Center approved this study (No. AN16343-001).
References
- Ben-David K, Sarosi GA, Cendan JC, et al. Decreasing morbidity and mortality in 100 consecutive minimally invasive esophagectomies. Surg Endosc 2012;26:162-7. [Crossref] [PubMed]
- Aminian A, Panahi N, Mirsharifi R, et al. Predictors and outcome of cervical anastomotic leakage after esophageal cancer surgery. J Cancer Res Ther 2011;7:448-53. [Crossref] [PubMed]
- Murphy CC, Incalcaterra JR, Albright HW, et al. Pretreatment patient comorbidity and tobacco use increase cost and risk of postoperative complications after esophagectomy at a high-volume cancer center. J Oncol Pract 2013;9:233-9. [Crossref] [PubMed]
- Zingg U, Smithers BM, Gotley DC, et al. Factors associated with postoperative pulmonary morbidity after esophagectomy for cancer. Ann Surg Oncol 2011;18:1460-8. [Crossref] [PubMed]
- Kassis ES, Kosinski AS, Ross P Jr, et al. Predictors of anastomotic leak after esophagectomy: an analysis of the society of thoracic surgeons general thoracic database. Ann Thorac Surg 2013;96:1919-26. [Crossref] [PubMed]
- Wright CD, Kucharczuk JC, O'Brien SM, et al. Predictors of major morbidity and mortality after esophagectomy for esophageal cancer: a Society of Thoracic Surgeons General Thoracic Surgery Database risk adjustment model. J Thorac Cardiovasc Surg 2009;137:587-95; discussion 596. [Crossref] [PubMed]
- Dolan JP, Kaur T, Diggs BS, et al. Impact of comorbidity on outcomes and overall survival after open and minimally invasive esophagectomy for locally advanced esophageal cancer. Surg Endosc 2013;27:4094-103. [Crossref] [PubMed]
- Gawad KA, Hosch SB, Bumann D, et al. How important is the route of reconstruction after esophagectomy: a prospective randomized study. Am J Gastroenterol 1999;94:1490-6. [Crossref] [PubMed]
- Chan ML, Hsieh CC, Wang CW, et al. Reconstruction after esophagectomy for esophageal cancer: retrosternal or posterior mediastinal route? J Chin Med Assoc 2011;74:505-10. [Crossref] [PubMed]
- Dahn I, Huldt B. Anthethoracic subcutaneous esophagus reconstruction with the stomach in esophageal cancer. Nord Med 1971;86:882-3. [PubMed]
- Dawson JH. Subcutaneous placement of the left colon for esophageal bypass. Am Surg 1969;35:574-9. [PubMed]
- Kent MS, Gayle L, Hoffman L, et al. A new technique of subcutaneous colon interposition. Ann Thorac Surg 2005;80:2384-6. [Crossref] [PubMed]
- Cooke DT, Lin GC, Lau CL, et al. Analysis of cervical esophagogastric anastomotic leaks after transhiatal esophagectomy: risk factors, presentation, and detection. Ann Thorac Surg 2009;88:177-84; discussion 184-5. [Crossref] [PubMed]
- Larburu Etxaniz S, Gonzales Reyna J, Elorza Orúe JL, et al. Cervical anastomotic leak after esophagectomy: diagnosis and management. Cir Esp 2013;91:31-7. [Crossref] [PubMed]
- Alanezi K, Urschel JD. Mortality secondary to esophageal anastomotic leak. Ann Thorac Cardiovasc Surg 2004;10:71-5. [PubMed]
- Díaz de Liaño Argüelles A, Sánchez García G, Yárnoz Irazábal C, et al. Oesophagogastric anastomosis complications in the Ivor Lewis operation. Cir Esp 2011;89:175-81. [Crossref] [PubMed]
- Rutegård M, Lagergren P, Rouvelas I, et al. Intrathoracic anastomotic leakage and mortality after esophageal cancer resection: a population-based study. Ann Surg Oncol 2012;19:99-103. [Crossref] [PubMed]
- Orringer MB, Marshall B, Chang AC, et al. Two thousand transhiatal esophagectomies: changing trends, lessons learned. Ann Surg 2007;246:363-72; discussion 372-4. [Crossref] [PubMed]
- Markar SR, Arya S, Karthikesalingam A, et al. Technical factors that affect anastomotic integrity following esophagectomy: systematic review and meta-analysis. Ann Surg Oncol 2013;20:4274-81. [Crossref] [PubMed]
- Junemann-Ramirez M, Awan MY, Khan ZM, et al. Anastomotic leakage post-esophagogastrectomy for esophageal carcinoma: retrospective analysis of predictive factors, management and influence on longterm survival in a high volume centre. Eur J Cardiothorac Surg 2005;27:3-7. [Crossref] [PubMed]


