Clinicopathological features of Chinese lung cancer patients with epidermal growth factor receptor mutation
Introduction
Epidermal growth factor receptor (EGFR) gene locates at chromosome 7 and has 28 exons. Mutations in EGFR gene are associated with different kinds of tumor including lung cancer. The EGFR protein which is a member of the protein kinase superfamily is a transmembrane glycoprotein. It is a cell surface protein receptor that binds to epidermal growth factor (EGF). The ligand and receptor binding induces dimerization and tyrosine autophosphorylation of EGFR (1). EGFR regulates cellular signaling pathways, promotes tumor cell differentiation, proliferation, maintenance, invasion and metastasis (2,3).
EGFR contains extracellular domain, transmembrane domain and intracellular domain which had tyrosine kinases (TK) domain and autophosphorylation domain. The TK domain which is the functional core of the protein consists of a smaller N-terminal and a larger C-terminal lobe (4). It stretches from exon 18 to exon 24. In lung cancer, the EGFR mutation sites center on exons 18–21 (5). Increasing catalytic activity of TK domain caused by EGFR mutation results in greatly over-expressed EGFR (3,6). Meanwhile it also provides a specific therapeutic strategy. Tyrosine kinases inhibitors (TKIs) targeted to TK domain have been approved for the treatment of NSCLC (7,8). Several studies suggest that the application of TKIs improved response rates and progression-free survival of lung cancer patients with EGFR mutations (9,10). The sensitivity of lung cancer patients to TKIs is associated with the mutation type. Patients with deletions in exon 19 and L858R in exon 21 responded positively. In this respect, the detection of EGFR mutations is the premise to the treatment of lung cancer patients. But in clinic the quantity of biopsy samples were not enough to fulfill the entire mutation screening. The phenotypic traits summary could help clinicians make judgement beforehand. Furthermore, most previous studies on EGFR mutations mainly focused on lung adenocarcinoma, few studies have evaluated the EGFR mutations in other lung cancer type in large scale. In the current study, we analyzed the EGFR mutation spectrum in Chinese lung cancer patients and summarized the clinicopathological characters of patients with EGFR gene mutations.
Methods
Ethical approval
This study was approved by the Institutional Review Board (IRB) of Shanghai Pulmonary Hospital affiliated Tongji University (No. 2014-016). Written informed consents were obtained from all participants. The methods were carried out in accordance with the approved guidelines.
Patients and specimen collection
The consecutive primary lung cancer patients who were admitted into the Shanghai Pulmonary Hospital affiliated Tongji University from Jun. 2014 to Oct. 2015 were recruited. No choose or correct was performed on patients’ collection. None of these patients received any anticancer therapies prior to surgery. The recurrent or metastatic patients were excluded. The samples which contained more than 50% tumor cells were qualified. Fresh primary tumor tissues were collected during the surgery.
Clinical and pathological data which was gathered for analysis included gender, age at diagnosis, pathological TNM stage, histological type, tumor morphology and location, visceral pleural invasion as well as smoking status. Tumors were staged pathologically according to the Union International Contre le Cancer (UICC-7) staging system for lung cancer (11).
Candidate gene mutation analysis
According to the manufacturer’s instruction, genomic DNA and total RNA were extracted from fresh tumor tissues using QIAamp DNA Tissue Kit and RNeasy Kit (Qiagen, Germany) respectively. EGFR mutations were detected by Amoy Diagnostics kits (Xiamen, China) which were based on amplification refractory mutation system (ARMS) real-time PCR. Twenty-nine mutations in exons 18–21 of EGFR gene were detected including T790M, L858R, L861Q, S768I, G719S, G719A, G719C, three types of insertions in exon 20, and 19 kinds of deletions in exon 19.
Statistical analysis
χ2 test was used to analyze the association between the EGFR mutation type and other clinicopathology data. All data were analyzed by the SPSS package for Windows (Version 18.0, Chicago, IL). P value <0.05 was considered statistically significant.
Results
Mutation spectrum
In total, 1,034 lung cancer patients were recruited in this study (Table 1), 515 of them had the EGFR gene mutations, 51.26% of them had L858R mutation and 39.61% had deletions in exon 19. G719X and L861Q were detected in less than 2% patients respectively. Besides, it was noteworthy that 20 patients had complex mutation, 19 del and L858R together.
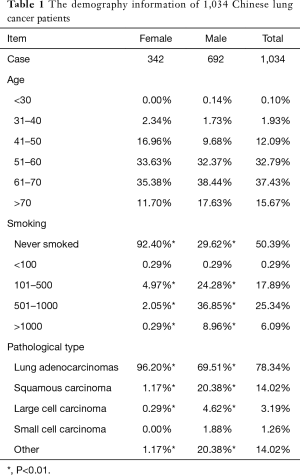
Full table
The phenotype and genotype associations
Compared to the wild-type EGFR patients, EGFR L858R mutation patients showed evident differences in the tumor site, pathological stage and type, tobacco using status, tumor size and visceral pleura invasion status (Table 2). Most tumors with EGFR L858R mutation located on the right side while tumors with non-EGFR mutation were on the left sides. Although the age profile looked similar between these two groups, it still had a little difference if it was analyzed by gender. L858R mutation female patients were elder than that of non-EGFR mutation.
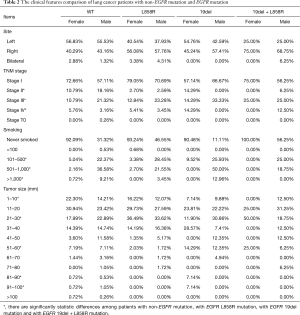
Full table
Tumors with EGFR 19 del mutation were also inclined to occur on the right side. This group had more advanced tumor (stage III) compared to the EGFR L858R mutation and wild-type patients. The invasion to visceral pleura was also common in this group.
Discussion
Lung cancer with the highest prevalence around the world caused great damage to patients. The burden of it was more and more serious. An effective treatment was urgently needed. In the past few years, the application of TKIs had greatly improved the treatment effect of lung cancer (9). But not all lung cancer patients were sensitive to TKIs. Previous reports suggested Asian female adenocarcinoma patients with no-smoking were sensible to TKIs (12,13). But it is still controversial (9). Therefore patient stratification is critical for successful application of TKIs. Accumulated evidences had suggested that the identification of EGFR mutations in lung adenocarcinomas was essential before administering TKIs (10).
EGFR is the major causative gene of lung cancer. But the mutation frequency is highly dependent on ethnicity. Patients with Asian origin have a higher prevalence compared with Caucasian. It was reported that EGFR mutation was detected in 34.3% of lung patients in Hong Kong (14) and 18.9% in South Korea (15). It was only 8–15% in patients from western European countries and 12–16% in American patients (16). In this study, we found the EGFR mutation rate was around 50% in our patients which was higher than the previous reported prevalence.
The most common EGFR mutations with lung cancer are 19 del and L858R mutation. Mutant EGFR protein results in constitutive activation of EGFR and promotes EGFR-mediated pro-survival and anti-apoptotic signal pathways (17). In our study, L858R was the most common type of EGFR mutation which was consistent with the previous report (18). But exon 19 del mutation was the dominant type in other reports (19). The inconsistent results might be caused by ethnic variations.
The concomitant mutation was reported occasionally. We found 1.93% investigated patients had both exon 19 del and L858R mutations simultaneously. Compared to patients with exon 19 del or L858R mutations solely, they were inclined to have small size (1–20 mm) adenocarcinomas which occurred in bilateral and invaded the visceral pleura.
The most serious issue with the TKIs treatment was drug resistant. Part of patients who had sensitive mutations will inevitably develop disease progression (20). This is frequently due to the development of a second-site mutation in the EGFR gene (21,22). Exon 20 insertion and T790M was confirmed as two major TKIs resistant mutations which account for up to 50% of acquired resistance (23,24). EGFR with T790M lose the binding ability through steric hindrance to TKIs (25). Most T790M mutations were developed after TKI using and the de novo T790M mutation was rare. In this study, we found 11 patients with exon 20 ins. Most of them were 51–70 years old. Right side, stage I lung adenocarcinoma was the character of tumor in this group. But our sample size was too small. Further large scale studies are needed to validate our results.
Tobacco using is one of the major aetiological factors of lung cancer and affects the EGFR mutation frequency. The EGFR mutation prevalence was 42.5% vs. 8.5% in never smoked lung cancer patients and patients with more than 26 pack-years of smoking in Americans (16). Similarly, in our study, 47.59% patients with wild-type EGFR never smoked, while more than 70% patients with different kinds of EGFR mutations were non-smokers, especially for patients with both exon 19 del and L858R mutation.
The major limitations of our study were the small sample size and short observation time. We would continue to follow up these patients and report the treatment response of them.
In a word, EGFR gene was also a major causative gene for Chinese lung cancer patients. L858R in exon 21 were the most common types. Patients with bilateral, small sized (1–20 mm) adenocarcinomas accompanied by the visceral pleura invasion were more likely to have both exon 19 del and L858R mutation. The clinicopathological characters summary will be helpful for clinicians to make treatment plan.
Acknowledgements
We appreciate the patients and their families for participating in this study.
Funding: This work was supported by grants from the National Natural Scientific Foundation of China (81602412 and 81370107), China Postdoctoral Science Foundation (2016M601510) and Scientific research project of Shanghai Municipal Commission of Health and Family Planning (2016Y0121; 2013ZYJB0401 and 2013sy023).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by the Institutional Review Board (IRB) of Shanghai Pulmonary Hospital affiliated Tongji University (No. 2014-016). Written informed consents were obtained from all participants.
References
- Benter IF, Yousif MH, Hollins AJ, et al. Diabetes-induced renal vascular dysfunction is normalized by inhibition of epidermal growth factor receptor tyrosine kinase. J Vasc Res 2005;42:284-91. [Crossref] [PubMed]
- Cheng L, Zhang S, Alexander R, et al. The landscape of EGFR pathways and personalized management of non-small-cell lung cancer. Future Oncol 2011;7:519-41. [Crossref] [PubMed]
- Mitsudomi T, Yatabe Y. Epidermal growth factor receptor in relation to tumor development: EGFR gene and cancer. FEBS J 2010;277:301-8. [Crossref] [PubMed]
- Yasuda H, Kobayashi S, Costa DB. EGFR exon 20 insertion mutations in non-small-cell lung cancer: preclinical data and clinical implications. Lancet Oncol 2012;13:e23-31. [Crossref] [PubMed]
- Xing K, Zhou X, Zhao X, et al. A novel point mutation in exon 20 of EGFR showed sensitivity to erlotinib. Med Oncol 2014;31:36. [Crossref] [PubMed]
- Inamura K, Ninomiya H, Ishikawa Y, et al. Is the epidermal growth factor receptor status in lung cancers reflected in clinicopathologic features? Arch Pathol Lab Med 2010;134:66-72. [PubMed]
- Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med 2009;361:947-57. [Crossref] [PubMed]
- Kalikaki A, Koutsopoulos A, Hatzidaki D, et al. Clinical outcome of patients with non-small cell lung cancer receiving front-line chemotherapy according to EGFR and K-RAS mutation status. Lung Cancer 2010;69:110-5. [Crossref] [PubMed]
- Wang S, Wang Z. EGFR mutations in patients with non-small cell lung cancer from mainland China and their relationships with clinicopathological features: a meta-analysis. Int J Clin Exp Med 2014;7:1967-78. [PubMed]
- Ping W, Xia C, Fu S, et al. Immunohistochemistry with a novel mutation-specific monoclonal antibody as a screening tool for the EGFR L858R mutational status in primary lung adenocarcinoma. Tumour Biol 2015;36:693-700. [Crossref] [PubMed]
- Kassis ES, Vaporciyan AA, Swisher SG, et al. Application of the revised lung cancer staging system (IASLC Staging Project) to a cancer center population. J Thorac Cardiovasc Surg 2009;138:412-8.e1-2.
- Tanaka T, Matsuoka M, Sutani A, et al. Frequency of and variables associated with the EGFR mutation and its subtypes. Int J Cancer 2010;126:651-5. [Crossref] [PubMed]
- Li M, Zhang Q, Liu L, et al. The different clinical significance of EGFR mutations in exon 19 and 21 in non-small cell lung cancer patients of China. Neoplasma 2011;58:74-81. [Crossref] [PubMed]
- Yung TK, Chan KC, Mok TS, et al. Single-molecule detection of epidermal growth factor receptor mutations in plasma by microfluidics digital PCR in non-small cell lung cancer patients. Clin Cancer Res 2009;15:2076-84. [Crossref] [PubMed]
- Han SW, Kim TY, Hwang PG, et al. Predictive and prognostic impact of epidermal growth factor receptor mutation in non-small-cell lung cancer patients treated with gefitinib. J Clin Oncol 2005;23:2493-501. [Crossref] [PubMed]
- Dogan S, Shen R, Ang DC, et al. Molecular epidemiology of EGFR and KRAS mutations in 3,026 lung adenocarcinomas: higher susceptibility of women to smoking-related KRAS-mutant cancers. Clin Cancer Res 2012;18:6169-77. [Crossref] [PubMed]
- Sordella R, Bell DW, Haber DA, et al. Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science 2004;305:1163-7. [Crossref] [PubMed]
- Chan SK, Gullick WJ, Hill ME. Mutations of the epidermal growth factor receptor in non-small cell lung cancer—search and destroy. Eur J Cancer 2006;42:17-23. [Crossref] [PubMed]
- Bircan S, Baloglu H, Kucukodaci Z, et al. EGFR and KRAS mutations in Turkish non-small cell lung cancer patients: a pilot study. Med Oncol 2014;31:87. [Crossref] [PubMed]
- Jiang X, Yang B, Lu J, et al. Pemetrexed-based chemotherapy in advanced lung adenocarcinoma patients with different EGFR genotypes. Tumour Biol 2015;36:861-9. [Crossref] [PubMed]
- Mitsudomi T, Yatabe Y. Mutations of the epidermal growth factor receptor gene and related genes as determinants of epidermal growth factor receptor tyrosine kinase inhibitors sensitivity in lung cancer. Cancer Sci 2007;98:1817-24. [Crossref] [PubMed]
- Yamamoto H, Toyooka S, Mitsudomi T. Impact of EGFR mutation analysis in non-small cell lung cancer. Lung Cancer 2009;63:315-21. [Crossref] [PubMed]
- Shih JY, Gow CH, Yang PC. EGFR mutation conferring primary resistance to gefitinib in non-small-cell lung cancer. N Engl J Med 2005;353:207-8. [Crossref] [PubMed]
- Heuckmann JM, Rauh D, Thomas RK. Epidermal growth factor receptor (EGFR) signaling and covalent EGFR inhibition in lung cancer. J Clin Oncol 2012;30:3417-20. [Crossref] [PubMed]
- Yun CH, Mengwasser KE, Toms AV, et al. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc Natl Acad Sci U S A 2008;105:2070-5. [Crossref] [PubMed]


