Cardiac surgery using a single thoracic port—current status and future directions
Introduction
Cardiac surgery is almost universally performed through median sternotomy, an approach with all the known drawbacks, of which pain and life threatening infections are the most worrisome. Frequency of deep sternal wound infection (DSWI) has been estimated to range from 0.5% to 6% and its in-hospital mortality has varied from 7% to 35% (1). Several alternative approaches for cardiac surgery have been tried since cardiac surgery became mainstream. Of these, upper and lower mini-sternotomies and left and right thoracotomies represent attempts to diminish cardiac surgery invasiveness. Unfortunately, these approaches have not been completely successful in their objective—pain and infections still happen with mini-sternotomies, albeit less, and pain is still a very significant component of thoracotomies (2).
The development of mitral and right atrial surgery using a mini-thoracotomy has somewhat lessened the problem of pain, but the need for arterial and venous cannulation, aortic cross-clamping and the placement of a thoracoscope translates into at least two additional ports in the thorax besides the mini-thoracotomy.
Taking from the several lessons learned from our and other’s experience with single port lung surgery and seeing the huge benefit the patients reaped from uniportal lung surgery regarding post-operatory pain, shorter hospital stay and cosmesis (3), our group started to develop and implement techniques which we believe will allow cardiac single thoracic port surgery (CSTPS) to become mainstream in the near future.
It is important to acknowledge that cardiac surgery using extra-corporeal circulation needs arterial and venous femural cannulation, in order to keep the thoracic incision small and single, without use of rib retractors.
Off-pump coronary surgery may, in selected cases, be safely done with a singe thoracic incision, but rib retractor use is still mandatory.
It should be noted that external defibrillator pads must be placed and ready to be use in all these surgeries.
Selected types of single port thoracic cardiac surgery
Mitral, tricuspid and atrial surgery
Mitral, tricuspid, and atrial surgery, including valve pathology, left and right cardiac masses and inter-atrial septum defects have been for many years corrected by right mini-thoracotomy in selected centers (4,5). Worldwide experience with these techniques has been growing, and results have progressively shown this approach is equal, if not better, to standard sternotomy in this setting.
Our group started a minimally invasive mitral, tricuspid and atrial surgery in 2014, and we have been able to perform a steady number of these surgeries per month.
We use video in all of these surgeries, which are always done through a right mini-thoracotomy. Our standard set-up is similar to that previously described for these procedures by the Stuttgart group. We always perform arterial and venous cannulation through the femoral artery and vein, usually on the right side. The usual technique utilizes a right mini-thoracotomy incision in the 4th or 5th intercostal space, a 1 cm incision for the Chitwood clamp, a third incision for placement of the thoracoscopy trocar, and a forth anterior incision for the cardioplegia and root vent needle.
Frequently, due to obesity, large thorax dimensions, or tight intercostal space, a rib retractor is used.
Our attempt to perform single thoracic port cardiac surgery started with asking ourselves how could we offer the same surgery to the patient while removing all but the main thoracic incision. We are able to avoid using a rib retractor in many patients, at the cost of loss of direct visualisation of the heart—all surgery is done endoscopically, a technically more demanding option. We still use a soft tissue retractor in all patients. In the end, rib retraction (with frequent concomitant rib fractures, painful and prone to bleeding in a patient who underwent extra-corporeal circulation) is avoided.
Two thoracic incisions may be avoided if Heartport technology is used for clamping the aorta and delivering cardioplegia. This option allows us to avoid the clamp incision and the aortic cardioplegia needle/vent placement, one of the more risk prone gestures in mini-thoracotomy surgery, due to occasional internal mammary artery and vein disruption, and aortic purse-string suturing.
To eliminate the need for the thoracoscope incision, we simply put the thoracoscope through the mini-thoracotomy incision, at superior extreme (Figure 1). The soft tissue retractor is mandatory to keep the thoracoscope lens clean. The main difficulty with placing the thoracoscope through the mini-thoracotomy is that a 30° lens is preferable, because a 0° lens will not allow good surgeon positioning to complete the surgery.
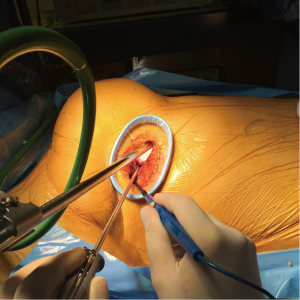
Using these modifications, we are able to, in selected patients, perform a CSTPS without any major modifications (other than the endoclamp utilization) to the usual right mini-thoracotomy mitral, tricuspid, and atrial surgery technique. In the end of the surgery, a 32 F chest drain is left through the mini-thoracotomy, as usual in the single port technique.
Coronary surgery
Minimizing the invasiveness of coronary surgery has been attempted many times, and minimally invasive direct coronary artery bypass (MIDCAB) has been practiced for many years worldwide. Endoscopical harvest of the internal mammary arteries has also been described and widely practiced. Both of these procedures have been accepted and performed in low numbers in most centers, due to being technically demanding, unavailability of appropriate instruments, and unsatisfactory results.
More recently, the development of specific instruments, stabilizers and proximal aortic anastomosis devices such as the Heart String (R) has allowed off-pump coronary surgery using a left mini-thoracotomy to be performed. This technique has been developed and championed by Drs. McGinn and Lapierre (6). Using the aforementioned devices, safe full coronary revascularization has been possible in selected patients, with very good long term results (6-9).
We started developing skills for this technique in 2015. We now currently use it as a first option in planned single left internal mammary artery-left anterior descending (LIMA-LAD) anastomosis.
Set-up for minimally invasive LIMA-LAD anastomosis
The patient is placed in prone position, with a padded pillow under the right hemithorax in a longitudinal fashion. The inguinal region is kept easily accessible and prepped bilaterally.
A submammary thoracic incision of about 7 cm to introduce the rib retractor blade is done. A soft tissue retractor is not used since all of the operation is done under direct vision. LIMA harvesting is done in the usual way using the rib retractor’s blades number 1 or 2, the Rultract and the navy arm.
Standard off-pump technique is utilized to perform the LIMA-LAD anastomosis, using Octopus epicardial stabilizer and intra-coronary shunts.
Performing posterior descending artery or obtuse marginal coronary anastomosis is more complex and normally needs additional ports (one in the right hemithorax and one subxiphoid) due to the need to lift the heart for access to the inferior and lateral walls. Besides, in about 30% of cases, cardiopulmonary bypass support is needed (done through femural cannulation).
Bilateral mammary artery and radial artery grafting through this technique has been described by some centers, with very satisfying results (7).
Currently the LIMA-LAD anastomosis is a single thoracic uniportal procedure, since there is but one thoracic incision. A rib retractor is mandatory in all but very selected cases because, while endoscopic LIMA harvesting is feasible and reproducible through the uniportal incision with a soft tissue retractor, the coronary anastomosis construction needs a perfect visualization of the LAD—proper choice of the incision placement is key to avoid rib retractor utilization. In thin patients with wide intercostal spaces, endoscopic harvesting of the LAD coupled with a thoracic incision right on top of the LAD may allow full single port surgery with the avoidance of a rib retractor.
In the end of the surgery, a 32 F chest drain is left in the pleural cavity, as usual in the single port technique.
Aortic valve surgery, septal myectomy and ascending aortic surgery
Minimally invasive aortic valve surgery has traditionally been done by a upper mini-sternotomy or a right anterior mini thoracotomy (2,10).
Our group normally utilizes the mini-sternotomy approach, with which we have accumulated significant experience. We currently perform a L-upper sternotomy and use a small sternal retractor. Arterial and venous cannulation is performed centrally in most cases, but in selected cases we have utilized femural cannulation. To facilitate air purging before aortic cross-clamp removal, we utilize CO2 injection in the surgical field (as in minimally invasive mitral surgery). Normally we place the subxiphoid chest drain during the induction cardioplegia (with an empty heart), and then connect the CO2 delivery system through the chest drain, with the gas being injected in the pericardium without additional cannulas obstructing the surgical field.
Due to chest drains being mandatory but not feasible through a mini-sternotomy approach (we usually place them through a subxiphoid approach), this surgical modality is not a complete single thoracic port cardiac surgery.
Rapid deployment valves have brought back the interest for the mini-sternotomy approach worldwide, and we have also utilized them in several cases, since they simplify and speed the valve implantation. Their only drawback nowadays seems to be the price (about 4 times the price of the standard stented aortic valves).
We also have used the L-mini sternotomy approach for ascending aortic surgery, with no significant technical modification. Arch surgery is possible with mini-sternotomy, but the skin incision must be extended in the cephalad direction in many cases for a good exposure of the aortic arch branches.
Video is used in septal miectomies or in left ventricle outflow tract (LVOT) tumors, allowing for perfect visualization of the LVOT.
Right anterior mini-thoracotomy has also been extensively used for aortic valve replacement surgery. While we find this a very interesting approach, frequently the right internal mammary artery and veins are sacrificed and at least one of the rib cartilages is separated from the sternum. In the end, chest drains are not placed through the incision, so this option is also far from being a true CSTPS.
Atrial fibrillation ablation
Currently we perform atrial fibrillation ablation by multi-trocar video assisted thoracic surgery (VATS), using CO2 to collapse the lung. Due to the need for a bilateral approach and fairly complex manipulation of delicate heart structures without extracorporeal circulation (since the procedure is all done off-pump), perfect cardiac stabilization is mandatory, and three trocars on each side (one for the camera, two for instruments) with different angles are at this point necessary. For the moment it’s difficult to think how to perform this technique with a single thoracic port.
Left atrial appendage closure
We have been performing left atrial appendage closure with a left sided two port approach. As in atrial fibrillation ablation procedures, left atrial appendage occlusion is an off-pump procedure which involves one of the most delicate heart structures, in which proper and secure manipulation is key. Still, we are developing techniques to transform this procedure into a complete single thoracic port cardiac surgery.
Transapical transcatheter aortic valve implantation (TAVI)
Transapical TAVI is a cardiac procedure in which a single thoracic port is sufficient for all patients, even the obese or with narrow intercostal spaces (11).
The patient is placed in a supine position with a pad underneath his left hemithorax.
The location of the left ventricle apex is determined using transthoracic echocardiography. A standard 6 cm thoracotomy is made in the apex mark, and the apex is easily localized and cannulated after a double purse string is constructed.
We normally use a soft tissue retractor and no rib retractor. In the end a single 32 F chest drain is left in the usual single port technique.
Transaortic TAVI
Currently, transaortic TAVI is done with a right anterior mini-thoracotomy. To have a good access to the ascending aorta allowing the construction of an aortic purse string suture, we still perform rib cartilage section and use a rib retractor. In the end, mandatory pericardial drainage using a single drain may be obtained through the mini-thoracotomy incision as in standard single port technique.
In this technique, the need for a very secure aortic purse string does not allow for the moment rib retractor avoidance. In the near future, automated devices of small dimension similar to ones already in use for apical cannulation may allow safe aortic cannulation without the need for rib section and spreading.
Recently, Kiser et al. have shown suprasternal notch arch access to be a very interesting option for transaortic TAVI, in a true uniportal fashion (12).
Conclusions
Nowadays, full single thoracic port cardiac surgery with avoidance of a rib spreader already exists and is being practiced, albeit in small numbers (Table 1).

Full table
Mitral, tricuspid, and atrial surgery can be done through a real single thoracic port technique using endoclamp technology.
Coronary surgery, especially LIMA-LAD anastomosis, may be done through a real single thoracic port with avoidance of rib spreaders, but it is very technically demanding and only applicable to very selected patients.
Transapical TAVI is another cardiac surgery which can easily be performed through a full single thoracic port approach.
Aortic valve, LVOT pathology and ascending aortic surgery are still far from being a full single thoracic port surgery even if femural cannulation is used, due to the universal need for a sternal retractor in mini-sternotomy or a rib retractor in mini-thoracotomy.
AF ablation and left atrial appendage closure are also still multiport VATS procedures, although the latter may, in the next years, be converted to a single thoracic port procedure.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Cotogni P, Barbero C, Rinaldi M. Deep sternal wound infection after cardiac surgery: Evidences and controversies. World J Crit Care Med 2015;4:265-73. [Crossref] [PubMed]
- Iribarne A, Easterwood R, Chan EY, et al. The golden age of minimally invasive cardiothoracic surgery: current and future perspectives. Future Cardiol 2011;7:333-46. [Crossref] [PubMed]
- Ng CS, Gonzalez-Rivas D, D'Amico TA, et al. Uniportal VATS-a new era in lung cancer surgery. J Thorac Dis 2015;7:1489-91. [PubMed]
- Czesla M, Götte JM, Doll N. How to establish video assisted, minimally invasive mitral valve surgery. Heart 2012;98:1172-8. [Crossref] [PubMed]
- Seeburger J, Borger MA, Falk V, et al. Minimal invasive mitral valve repair for mitral regurgitation: results of 1339 consecutive patients. Eur J Cardiothorac Surg 2008;34:760-5. [Crossref] [PubMed]
- McGinn JT Jr, Usman S, Lapierre H, et al. Minimally invasive coronary artery bypass grafting: dual-center experience in 450 consecutive patients. Circulation 2009;120:S78-84. [Crossref] [PubMed]
- Kikuchi K, Une D, Endo Y, et al. Minimally Invasive Coronary Artery Bypass Grating Using Bilateral In Situ Internal Thoracic Arteries. Ann Thorac Surg 2015;100:1082-4. [Crossref] [PubMed]
- Barsoum EA, Azab B, Patel N, et al. Long-term Outcome after Percutaneous Coronary Intervention Compared with Minimally Invasive Coronary Artery Bypass Surgery in the Elderly. Open Cardiovasc Med J 2016;10:11-8. [Crossref] [PubMed]
- Lemma M, Atanasiou T, Contino M. Minimally invasive cardiac surgery-coronary artery bypass graft. Multimed Man Cardiothorac Surg 2013;2013:mmt007. [Crossref] [PubMed]
- Glauber M, Ferrarini M, Miceli A. Minimally invasive aortic valve surgery: state of the art and future directions. Ann Cardiothorac Surg 2015;4:26-32. [PubMed]
- Cheung A, Lichtenstein KM. Illustrated techniques for transapical aortic valve implantation. Ann Cardiothorac Surg 2012;1:231-9. [PubMed]
- Kiser AC, O'Neill WW, de Marchena E, et al. Suprasternal direct aortic approach transcatheter aortic valve replacement avoids sternotomy and thoracotomy: first-in-man experience†. Eur J Cardiothorac Surg 2015;48:778-83; discussion 784. [Crossref] [PubMed]


