Continuous administration of recombinant human B-type natriuretic peptide can improve heart and renal function in patients after cardiopulmonary bypass surgery
Introduction
Any cardiac surgery under cardiopulmonary bypass (CPB) will induce ischemia-reperfusion injury especially in heart and kidney and systematic inflammatory response, which may cause a lot of complications such as cardiac stunning, low cardiac output syndrome, acute respiratory distress syndrome (ARDS), acute kidney injury (AKI) and so on (1-3). Interactions of these complications lead to exacerbation, even to death. In clinical practice, renal function will soon collapse when low cardiac output syndrome occurs and thus exacerbating the cardiac problem (4). Conventional therapy strategy is to use inotropes, diuretics and vasodilator drugs, yet, the therapeutic effects of which need to be improved (5).
B-type natriuretic peptide (BNP), one of the neuro-humor factors that produced and released from the ventricles that plays an important role in heart failure (6). With the in-depth study of the pathogenesis of heart failure in recent years, the functions of BNP have been further researched that not only does it have effects of diuretic and natriuretic, but it also can prevent myocardial remodeling and fibrosis through the side secretory pathway and inhibit the activation of renin–angiotensin–aldosterone system (RAAS) which can reduce both heart and renal function (7,8).
Recombinant human B-type natriuretic peptide (rhBNP), which has the same bioactivity as BNP, was approved by FDA in 2001 to treat severity of congestive heart failure and recommended in the acute decompensated heart failure (ADHF) treatment guidelines by the European Society of Cardiology (ESC) in 2005 (9). It can mediate natriuretic, diuretic and smooth muscle relaxant effects, and thus decrease in preload and afterload by venous and arterial vasodilation, which results in increasing cardiac output (10,11). In some clinical researches, rhBNP can improve heart function in treating heart failure by inhibiting RAAS and stimulating the sympathetic nervous system (5,11,12). Some researchers found that rhBNP had fewer adverse effects compared with nitroglycerin (11,13). In acute ST-elevation myocardial infarction (STEMI) patients, rhBNP therapy showed its protection of ventricular function and structure (14).
However, the effects of rhBNP on patients carried out CPB surgery are unknown. In this study, we will evaluate the effects of rhBNP in patients on heart and kidney after CPB surgery and try to find the possible mechanism.
Methods
Patient selection
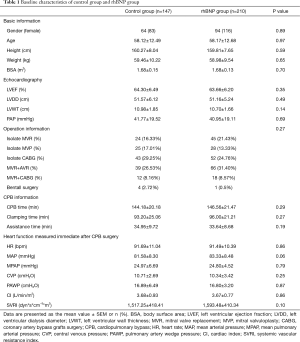
We retrospect patients carried out cardiac surgery between Jan 1st 2014 and Dec 31st 2015 from Department of Cardiothoracic Surgery of the second affiliated hospital of Second Military Medical University. Total of 357 patients were enrolled in the study, and the cardiac etiology included valve regurgitation or stenosis, coronary artery disease. Those who carried off-pump cardiac surgery such as OPCAB were excluded, as well as those who had chronic kidney disease and needed hemodialysis before surgery. The clinical data of the patients are given in Table 1.
Full table
According to the use of rhBNP, those patients were divided into two groups: rhBNP group (n=210) and control group (n=147). All patients were taken optimal conventional medical therapy after surgery but no diuretics. Patients in rhBNP group were received continuous recombinant human B-type natriuretic peptide (marketed as Xinhuosu, Chengdu Rhodiola Biopharmacy, Chengdu, China. Approval Number: Zhunzi S20050033) in 6 hours after CPB surgery, for a period of 72 h. rhBNP was administered as an intravenous bolus of 0.0075–0.01 µg/kg/min according to each patient’s clinical status.
Ethical approval was given by the Committee on Ethics of Biomedicine Research, Second Military Medical University, and all patients had signed the informed consent voluntary.
Hemodynamic study
All patients were monitored continuously with Philip MP70 monitor in CICU after surgery. A Swan-Ganz catheter was inserted into the jugular vein before surgery and was kept inserted during the first 24 h after surgery in all patients. Hemodynamic parameters, including heart rate (HR), mean invasive arterial pressure (MAP), mean pulmonary arterial pressure (MPAP), central venous pressure (CVP), cardiac index (CI), systemic vascular resistance index (SVRI) and pulmonary artery wedge pressure (PAWP), were measured immediately after CPB surgery, and then at 2, 4, 8, 12 and 24 h after surgery.
Blood analysis
Blood samples were obtained immediately after surgery and thereafter once a day at 6:00 AM within the first 3 days after surgery. All blood samples were placed into EDTA-coated tubes and measured by clinical laboratory of the second affiliated hospital of Second Military Medical University.
Length of tracheal intubation time, chest drainage time, ICU stay and the volume of urine
The volume of urine was recorded at the next 6 AM and then every 24 h after surgery, and the length of tracheal intubation time, chest drainage time and ICU stay were also recorded.
Statistical analysis
Statistical analysis was performed with IBM SPSS Statistics 21.0 statistical software. For univariate analyses, two-sample t-tests were used to compare continuous variables, while the χ2 test or Fisher’s exact test was used to compare categorical variables between the two groups. The hemodynamic and blood parameters were compared over the time course using ANOVA for repeated measures. Continuous variables were described as mean±SEM, while discrete variables were reported as frequencies and percentage. Statistical significance was defined as P<0.05.
Results
Baseline characteristics
All the patients enrolled in the study were carried CPB surgery, and no one died in surgery or in the first 72 h after surgery. After surgery, all patients were given low dose of dopamine (0–5 µg/kg/min) and/or epinephrine (0–0.05 µg/kg/min). Doses of those inotropes were reduced based on the patients’ conditions, and usually stopped before patients converted out of ICU (mostly in 5 days). Five patients, 3 in control group and 2 in rhBNP group, died in hospital. Two patients (one each group) died of critical mediastinal bleeding at the 4th day and the 5th day after surgery, which may be caused by the chest tubes. The other three died of pneumonia induced sepsis shock. There were no significant differences in the baseline characteristics, including age (P=0.97), gender (P=0.89), left ventricular ejection fraction (LVEF) (P=0.35), pulmonary arterial pressure (PAP) (P=0.69), CPB time (P=0.29) or aortic clamping time (P=0.26) between the two groups. The baseline heart function and renal function of the patients was well balanced in rhBNP group and control group immediately after surgery (Table 1).
Changes in hemodynamic parameters
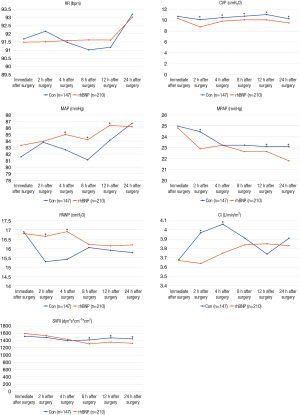
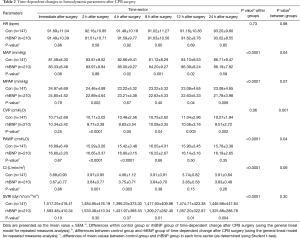
In both control group and rhBNP group, there were significant differences in the time-dependent changes of MAP (P<0.0001, within groups), MPAP (P<0.0001, within groups), PAWP (P<0.0001, within groups), CI (P<0.0001, within groups) and SVRI (P<0.0001, within groups) (Figure 1). MAP and CI increased by the time in rhBNP group, while MPAP, PAWP and SVRI decreased. But HR (P=0.73, within groups) and CVP (P=0.06, within groups) showed no difference in time-dependent changes in both groups (Table 2).
Full table
There were no significant differences in the time-dependent changes in HR (P=0.98, between groups), MPAP (P=0.07, between groups) or SVRI (P=0.30, between groups) between control group and rhBNP group. Our results showed that MAP (P=0.04, between groups) and PAWP (P=0.04, between groups) in rhBNP group were significantly higher than those in control group, while CVP (P=0.001, between groups) was lower in rhBNP group (Figure 1, Table 2).
Though there were no significant differences in the time-dependent changes in MPAP, CI or SVRI between two groups, differences were showed in some specific points-in-time (Figure 1). MPAP in rhBNP group was lower at 2 h (24.46±4.99 vs. 22.89±4.64 mmHg, P=0.002), 12 h (23.08±4.65 vs. 22.63±5.33 mmHg, P=0.04) and 24 h (23.08±4.65 vs. 21.78±3.98 mmHg, P=0.006) after surgery. SVRI was also lower in rhBNP group at 8 h (1,417.93±409.98 vs. 1,309.27±282.40 dyn*s*cm-5*m2, P=0.01), 12 h (1,474.71±423.38 vs. 1,357.20±322.87 dyn*s*cm-5*m2, P=0.01) and 24 h (1446.98±447.84 vs. 1,325.88±288.78 dyn*s*cm-5*m2, P=0.004) after surgery. Though CI in rhBNP group was lower at 2 h (3.97±0.95 vs. 3.64±0.77 L/min/m2, P=0.001) and 4 h (4.06±1.12 vs. 3.75±0.71 L/min/m2, P=0.003), it increased swiftly since 8h after surgery (Table 2).
Changes in serum BNP, CK-MB, troponin and creatinine
There were significant differences in the time-dependent changes of serum BNP (P<0.0001, within groups), CK-MB (P<0.0001, within groups), troponin (P<0.0001, within groups) and creatinine (P<0.0001, within groups) in both groups. Serum BNP, CK-MB and troponin increased at the first day after CPB surgery and then started and kept decreasing slowly. Serum troponin fell more in rhBNP group especially at day 2 and day 3 (P<0.0001, between groups). Serum creatinine kept increasing after CPB surgery at the first 3 days in both groups, and it had a significant difference between control and rhBNP group (P<0.0001, between groups), which showed a less increase in rhBNP group in all the three days after surgery. Although serum BNP in rhBNP group is lower than that in control group (P<0.0001) only at day 2 after surgery, the overall changes of serum BNP between the two groups showed a significant difference. But serum CK-MB (P=0.23, between groups) seems to have no difference between the two groups (Figure 2, Table 3).
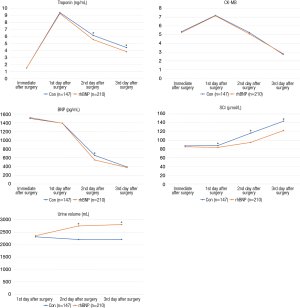
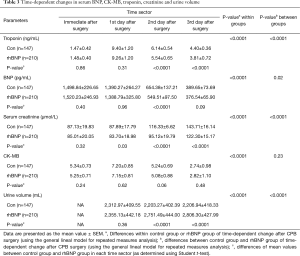
Full table
Length of tracheal intubation time, chest drainage time, ICU stay and the volume of urine
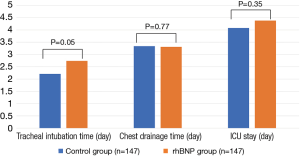
Patients who were continuously given intravenous rhBNP in 6 h after CPB surgery had more urine than those in control group (P<0.0001, between groups). While the volume of urine in rhBNP group increased significantly since day 2 after surgery and kept increasing, the volume of urine in control group seems to have a little decrease (Table 3). However, our results showed that patients in rhBNP group tended to wean off the respirator half a day later than those in control group (P=0.05). There was no significant difference in the length of chest drainage time (P=0.88) and intensive care unit (ICU) stay (P=0.64) between the two groups (Figure 3, Table 4).

Full table
Discussion
Cardiac stunning and acute kidney injury are two of the most common complications induced by CPB surgery (2). Interactions of these two complications will cause changes in hemodynamics, deterioration of heart and renal function, multiple organ disorder syndrome or death. Usually, we use inotropes, diuretics and vasodilator drugs, but the therapeutic effects remain unsatisfactory (5).
B-type natriuretic peptide (BNP) binds to the A-type natriuretic peptide receptor presented on the surface of vascular smooth muscle and endothelial cells (6), resulting in activation of guanylate cyclase and the subsequent accumulation of intracellular cyclic GMP in target tissues (10), which can mediate natriuretic, diuretic and smooth muscle relaxant effects. It can inhibit the renin-angiotensin-aldosterone system and the sympathetic nervous system (7,8), thus decreasing in systemic vascular resistance and cardiac preload and afterload by venous and arterial vasodilation and prevent myocardial remodeling and fibrosis, which results in increasing in cardiac output (10, 11).
Recombinant human B-type natriuretic peptide (rhBNP), which has the same bioactivity as BNP, was approved by FDA in 2001 to treat severity of congestive heart failure and recommended in the ADHF treatment guidelines by the European Society of Cardiology in 2005 (9).
In many studies, it indicated that using rhBNP in heart failure or acute coronary syndrome patients resulted an improvement in hemodynamics and self-reported symptoms more effectively and with fewer adverse effects compared with nitroglycerin or dopamine (11-14). Most of the changes in hemodynamics in our study were similar to those in former studies, that rhBNP can significantly ameliorate the heart function, based on changes of hemodynamics. We also found that the application of rhBNP seemed to have benefits in some patients with right heart dysfunction, especially in those who got huge volume of right heart or severe tricuspid regurgitation, but those effects didn’t appear on each patient, and further research need to be continued.
But we found that the MAP of the patients in our study showed an increase in time course, which was different from most previous studies. The proper reason that causes the difference is that most previous studies focused on the patients with ADHF, and most of those patients will evidence an increase in cardiac afterload. On the contrary, patients after CPB surgery occurred lacking of circulation capacity and cardiac stunning, which are caused by bleeding in the surgery and CPB itself. So, if those patients were given enough fluid, their blood pressure would rise quickly like which was indicated in our study. We also found that the increase of MAP in rhBNP group was slenderer compared to that of the control group, which may be caused by the effect of diuretic and vasodilator of rhBNP. At the meantime, CVP in rhBNP group showed a significant lower than in control group also indicates those effects of rhBNP. The application of rhBNP can also reduce the serum troponin level faster, which seems to speed up the recovery from myocardial injury.
In the first 4 h after surgery, the level of CI of control group was significantly higher than that of rhBNP group, but tended to be equal after 8h, which indicates that the application of rhBNP right after cardiac surgery may cause the decrease of cardiac output. Further study should focus on the administration time of rhBNP.
Though some studies showed that there was no effect of low dose (0.001 µg/kg/min) or fixed-dose (0.01 µg/kg/min) rhBNP on 72 h cumulative urine volume or renal function in acute decompensated heart failure patients compared to placebo (15,16), the results in our study demonstrated that rhBNP infusion can increase the urine volume and decrease the increasing speed of serum creatinine level, and therefore improves the renal function after CPB surgery. But such effect appeared at the second day after surgery, probably related to the total bolus of rhBNP. The reasons of the differences between our study and previous studies may be concluded that rhBNP is more efficient in yellow race. It needs further studies, especially randomized controlled trials (RCTs) in different races and different doses, in order to prove this hypothesis.
Interestingly, although the administration of rhBNP could improve the heart and renal functions of patients after CPB surgery, it did not improve the prognosis. Contrarily, the tracheal intubation time was longer in rhBNP group unexpectedly, but the difference is only about half a day. Therefore, we thought the difference is only a borderline statistical significance with no clinical value.
In our studies, 3 patients in control group and 2 in rhBNP group finally dies in hospital, caused by pneumonia, sepsis shock or critical bleeding. Those 5 patients showed no special clinical manifestations to others in the first 3 days after surgery, so we didn’t exclude them from the study. Meanwhile the in-hospital death seemed to bear a little relation to the implementation of rhBNP. Several studies also indicated that rhBNP had fewer adverse effects and was safe in renal dysfunctional patients (13,17).
Study limitation
Several limitations to this study shouldn’t be ignored. In our study, most of the Swan-Ganz catheters were kept inserted in 24 hours. So, some hemodynamic parameters such as CI and SVRI could only be recorded in 24 hours, which may have an influence on the evaluation of the changes of heart function after rhBNP therapy. We only focused on the left ventricular function, instead on the correction of right heart function. That is what we need to analyze next time or in the future. Despite of these limitations, the improvement of heart and renal function in patients underwent CPB surgery in rhBNP group is affirmative.
Conclusions
We found that rhBNP could improve heart and renal function in patients underwent CPB surgery as well as accelerating the recovery from myocardial injury. However, the prognosis of the patients who were administrated rhBNP did not improve in our study.
Currently, the mechanisms of rhBNP in ADHF patients are well published. However, how rhBNP improves heart and renal function on patients underwent CPB surgery is still unknown. Further studies could focus on new protection mechanisms of rhBNP in patients carried out cardiac surgery.
Acknowledgements
Funding: This work was supported by the National Nature Science Foundation of China (No. 81172228) and Shanghai Foundation for Development of Science and Technology (No. 12140901600). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by the Committee on Ethics of Biomedicine Research, Second Military Medical University, and written informed consent was obtained from all patients.
References
- O'Neal JB, Shaw AD, Billings FT 4th. Acute kidney injury following cardiac surgery: current understanding and future directions. Crit Care 2016;20:187. [Crossref] [PubMed]
- Haase M, Shaw A. Acute kidney injury and cardiopulmonary bypass: special situation or same old problem? Contrib Nephrol 2010;165:33-8. [Crossref] [PubMed]
- Anselmi A, Abbate A, Girola F, et al. Myocardial ischemia, stunning, inflammation, and apoptosis during cardiac surgery: a review of evidence. Eur J Cardiothorac Surg 2004;25:304-11. [Crossref] [PubMed]
- Ronco C, Haapio M, House AA, et al. Cardiorenal syndrome. J Am Coll Cardiol 2008;52:1527-39. [Crossref] [PubMed]
- Brunner-La Rocca HP, Kaye DM, Woods RL, et al. Effects of intravenous brain natriuretic peptide on regional sympathetic activity in patients with chronic heart failure as compared with healthy control subjects. J Am Coll Cardiol 2001;37:1221-7. [Crossref] [PubMed]
- Arora S, Clarke K, Srinivasan V, et al. Effect of nesiritide on renal function in patients admitted for decompensated heart failure. QJM 2007;100:699-706. [Crossref] [PubMed]
- Omland T, Hagve TA. Natriuretic peptides: physiologic and analytic considerations. Heart Fail Clin 2009;5:471-87. [Crossref] [PubMed]
- Kawakami R, Saito Y, Kishimoto I, et al. Overexpression of brain natriuretic peptide facilitates neutrophil infiltration and cardiac matrix metalloproteinase-9 expression after acute myocardial infarction. Circulation 2004;110:3306-12. [Crossref] [PubMed]
- Nieminen MS, Böhm M, Cowie MR, et al. Executive summary of the guidelines on the diagnosis and treatment of acute heart failure: the Task Force on Acute Heart Failure of the European Society of Cardiology. Eur Heart J 2005;26:384-416. [Crossref] [PubMed]
- Michaels AD, Klein A, Madden JA, et al. Effects of intravenous nesiritide on human coronary vasomotor regulation and myocardial oxygen uptake. Circulation 2003;107:2697-701. [Crossref] [PubMed]
- Dunavant S. Nesiritide vs nitroglycerin for decompensated congestive heart failure. JAMA 2002;288:571-2; author reply 572-3. [Crossref] [PubMed]
- Pan HY, Zhu JH, Gu Y, et al. Comparative effects of recombinant human brain natriuretic peptide and dobutamine on acute decompensated heart failure patients with different blood BNP levels. BMC Cardiovasc Disord 2014;14:31. [Crossref] [PubMed]
- Publication Committee for the VMAC Investigators (Vasodilatation in the Management of Acute CHF). Intravenous nesiritide vs nitroglycerin for treatment of decompensated congestive heart failure: a randomized controlled trial. JAMA 2002;287:1531-40. [PubMed]
- Gong X, Mou Z, Shao L, et al. Human recombinant-B-type natriuretic peptide protect ventricular function and structure in ST-elevation myocardial infarction. Int J Clin Exp Pathol 2015;8:11622-8. eCollection 2015.
- Chen HH, Anstrom KJ, Givertz MM, et al. Low-dose dopamine or low-dose nesiritide in acute heart failure with renal dysfunction: the ROSE acute heart failure randomized trial. JAMA 2013;310:2533-43. [Crossref] [PubMed]
- Witteles RM, Kao D, Christopherson D, et al. Impact of nesiritide on renal function in patients with acute decompensated heart failure and pre-existing renal dysfunction a randomized, double-blind, placebo-controlled clinical trial. J Am Coll Cardiol 2007;50:1835-40. [Crossref] [PubMed]
- Owan TE, Chen HH, Frantz RP, et al. The effects of nesiritide on renal function and diuretic responsiveness in acutely decompensated heart failure patients with renal dysfunction. J Card Fail 2008;14:267-75. [Crossref] [PubMed]