Unidirectionally progressive resection of lower right lung cancer under video-assisted thoracoscopy
Introduction
Video-assisted thoracoscopic surgery (VATS) has been widely applied in procedures such as resection of mediastinal tumors, wedge resection of lung masses, lobectomy, radical treatment of lung cancer, excavation of esophageal leiomyoma, and resection of esophageal cancer. In particular, VATS lobectomy has been recognized as the standard surgical option for treating lung cancer by the National Comprehensive Cancer Network and the American Association of Chest Physicians in their guidelines in 2006 and 2007, respectively. Thoracoscopic lobectomy is currently performed using the traditional or unidirectional approach. In 1994, He Jianxing first used the traditional lobectomy under thoracoscopy in China, making him one of first surgeons to implement this technique. In traditional lobectomy, the lobectomy is completed by handling the pulmonary fissure, the pulmonary artery, the pulmonary vein, and the bronchus in this order. The single-direction concept was first introduced by Liu L (1) in China and applied in clinical treatment, which eliminated the need for flipping the lobes back and forth, up and down. The surgical resection is generally completed unidirectionally from the anterior to posterior regions when removing the upper or middle lobes, or from the inferior to superior regions when dealing with the lower lobe. In view of the specific structure of the pulmonary hilum, the operation is progressed in the order of freeing and separating the pulmonary vein, the bronchus, the pulmonary artery, and the pulmonary fissure. Lobectomy of the affected lobes and mediastinal lymph node dissection constitute the standard radical operation for non-small cell lung cancer. Compared with the conventional open surgery, VATS radical resection for lung cancer is associated with less injury on the chest wall, decreased impact on the respiratory function, and reduced postoperative pain, and thereby lower risk of postoperative complications of the respiratory and circulatory systems, such as pneumonia, pulmonary atelectasis and cardiac arrhythmias. All these will in turn lead to better postoperative recovery. Double-lumen endotracheal intubation plays an important role in the successful application of VATS surgery. Villamizar and colleagues (2) have confirmed that the technique significantly reduces blood loss and the postoperative complication rate, with significantly shorter time of thoracic drainage and hospital stay compared with conventional thoracotomy. A number of studies (3,4) have demonstrated that VATS radical resection provides comparable long-term outcomes for lung cancer patients to open surgery.
Video description (Video 1)
Challenging steps
The key to success for VATS lobectomy of lung cancer includes thorough lymph node dissection, and proper management of major intraoperative bleeding. Advantages: the lobectomy should be followed by systemic lymph node dissection. The dead spot-free coverage and certain amplification of the surgical field in VATS allows further thorough lymph node dissection compared with traditional open surgery. However, obviously enlarged lymph nodes often indicate the presence of metastases, which are associated with a high risk of capsule rupture and potential tumor seeding during thoracoscopic resection. Therefore, we do not recommend this surgical technique for patients with known significant enlargement of hilar or mediastinal lymph nodes, or as confirmed by preoperative PET-CT or CT.
The surgery is performed under intravenous general anesthesia using double-lumen endotracheal intubation, with contralateral one-lung ventilation. The double-lumen tube intubation is essential for VATS surgery because it is mandatory to collapse the lung on the side of thoracoscopic surgery to allow smooth access.
The patient is placed in a 90-degree position lying on the unaffected side.
Ports
An approximately 1.5-cm observation port is created in the 7th intercostal space between the middle and anterior axillary lines, an approximately 4-cm working port in the 4th intercostal space between the anterior axillary line and the midclavicular line, and an approximately 1.5-cm auxiliary port in the 9th intercostal space between the posterior axillary line and the subscapular line. There is no need for rib retraction. The operator stands in front of the patient, manipulating the endoscopic instruments while watching the monitor.
Surgical sequence
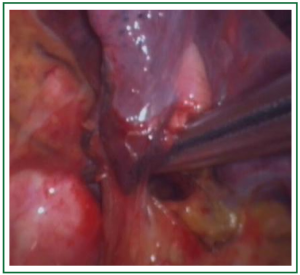
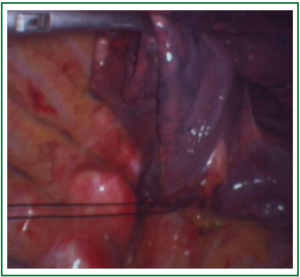
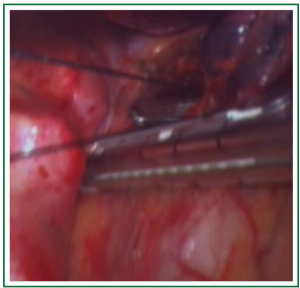
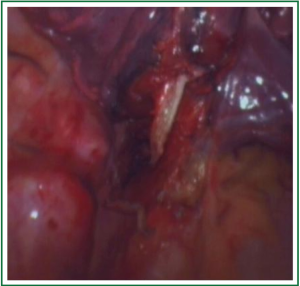
For traditional lobectomy, the following structures are operated in this order: (I) the right upper lobe: pulmonary vein—pulmonary artery—horizontal fissure—posterior ascending branch of the pulmonary artery—poorly developed fissure—bronchus; (II) the right middle lobe: pulmonary vein—junction between the oblique and horizontal fissures—pulmonary artery-poorly developed fissure-bronchus; (III) the right lower lobe: pulmonary vein—oblique fissure—pulmonary artery—poorly developed fissure —bronchus; (IV) the left upper lobe: pulmonary vein—pulmonary artery—oblique fissure—posterior ascending branch of the pulmonary artery—poorly developed fissure—bronchus; (V) the left lower lobe: pulmonary vein—oblique fissure—pulmonary artery—poorly developed fissure—bronchus. In the unidirectionally progressive lobectomy, the operation proceeds from the soft tissue at the hilum to deeper structures through the working port, in which the layers are freed and separated successively until the fissure in a single direction. This eliminates the need for flipping the lobes back and forth, up and down. The resection is completed unidirectionally from the anterior to posterior regions when removing the upper or middle lobes, or from the inferior to superior regions when dealing with the lower lobe. The pulmonary vein, bronchi, pulmonary artery and the poorly developed fissure of the right lower lobe are treated successively during lobectomy. In view of the specific structure of the pulmonary hilum, the operation is progressed in the order of freeing and separating the pulmonary vein, the bronchus, the pulmonary artery, and the pulmonary fissure. In the case of resection of the right upper lobe, the superior pulmonary vein is initially freed. A stapler is inserted through the auxiliary port for dissection of the right superior pulmonary vein, and separation of all branches of the superior pulmonary artery, which are cut with the stapler or ligated with suture. The upper lobe bronchus is then freed towards the posterior region, and cut with the stapler through the auxiliary port. At last, based on the development conditions, the fissure is freed with an electrotome or the stapler. The hilar structures are mainly freed with the electrotome in combination with a suction device. The vessels, bronchi and fissures are cut using an endoscopic linear stapler or the Hemolock clips.
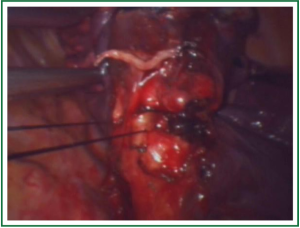
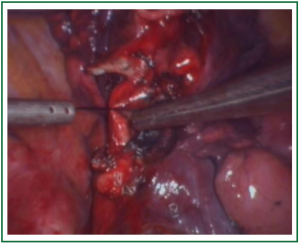
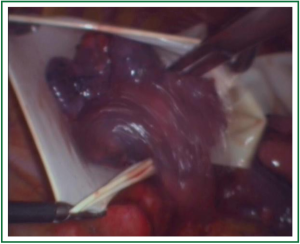
In this case, the right inferior pulmonary vein in the right inferior pulmonary ligament is first separated (Figures 1,2,3,4,5,6,7,8,9,10,11).
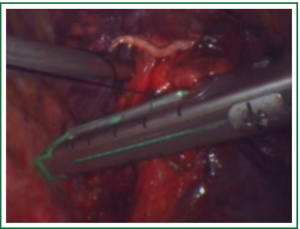
Comments
VATS resection of lung cancer is characterized in that it is performed with incisions as small as only 3 to 5 cm, avoiding rib retraction and extensive transection of chest muscles. In this way, it enables better protection of the neuromuscular system, significantly reduced postoperative pain, blood loss, as well as the incidence of postoperative complications, and increased benefits for postoperative recovery. The minimally invasive approach is also more easily accepted by patients, and empowers them with enhanced confidence in recovery. The key to success for VATS lobectomy of lung cancer includes thorough lymph node dissection, and proper management of major intraoperative bleeding. The lobectomy should be followed by systemic lymph node dissection. The dead spot-free coverage and certain amplification of the surgical field in VATS allow further thorough lymph node dissection compared with traditional open surgery. However, obviously enlarged lymph nodes often indicate the presence of metastases, which are associated with a high risk of capsule rupture and potential tumor seeding during thoracoscopic resection. Therefore, we do not recommend this surgical technique for patients with known significant enlargement of hilar or mediastinal lymph nodes, or as confirmed by preoperative PET-CT or CT. At present, however, complete thoracoscopic surgery is only available in large hospitals in China, mainly due to the strict demands for highly experienced surgeons. The major difficulty in managing rupture of blood vessels is also a primary cause of conversion to thoracotomy. The unidirectional procedure of VATS lobectomy has shaken off the shackles of traditional process, making it easier to perform the lobectomy under thoracoscopy in a smooth and simple way. The unidirectional thoracoscopic lobectomy is performed within the hilar soft tissue structure following a single direction without entering the lung parenchyma, or tearing or cutting the lung tissue, so that little injury is brought to the parenchyma. At the final step of fissure resection, any bronchial and pulmonary vascular interference has been removed, making it simple to cutting the tissue with a linear stapler, which significantly reduces the risk of postoperative air leaks. In particular, direct resection with the stapler is possible for patients with a poorly developed fissure, in which separation of the pulmonary artery is not necessary. This can significantly reduce blood loss and surgical time, and lower the difficulty of operation. The unidirectional thoracoscopic lobectomy also reduces lung injury by avoiding repeated flipping of the lung tissue.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Liu LX, Che GW, Pu Q, et al. Single-direction VATS lobectomy. Chinese journal of thoracic and cardiovascular surgery 2008;24:156-8.
- Villamizar NR, Darrabie MD, Burfeind WR, et al. Thoracoscopic lobectomy is associated with lower morbidity compared with thoracotomy. J Thorac Cardiovasc Surg 2009;138:419-25. [PubMed]
- Flores RM, Park BJ, Dycoco J, et al. Lobectomy by video-assisted thoracic surgery (VATS) versus thoracotomy for lung cancer. J Thorac Cardiovasc Surg 2009;138:11-8. [PubMed]
- Yan TD, Black D, Bannon PG, et al. Systematic review and meta-analysis of randomized and nonrandomized trials on safety and efficacy of video-assisted thoracic surgery lobectomy for early-stage non-small-cell lung cancer. J Clin Oncol 2009;27:2553-62. [PubMed]