Gastro-esophageal junction cancers: what is the best minimally invasive approach?
Introduction
Esophageal cancer is the sixth most-common cause of death from cancer worldwide with over 450,000 new cases annually (1). In Northern and Western Europe, and the USA the predominant histologic subtype of esophageal cancer is an adenocarcinoma and the prevalence of this subtype is increasing rapidly in these countries, particularly for males (2). Risk factors for developing an adenocarcinoma are symptomatic gastro-esophageal reflux disease (GERD), Helicobacter pylori infection, Barrett’s esophagus, obesity and a combination of alcohol and smoking (3). Mainly, adenocarcinomas develop in the distal third of the esophagus, in the esophagogastric junction (EGJ) and gastric cardia. The main symptoms of patients with esophageal cancer are dysphagia, weight loss, pain in the stomach or symptoms of anemia (4). When patients present with symptoms, a variety of diagnostic instruments are available to assess the location and spread of the tumor and to check for local or distant metastases. To assess the precise location of the tumor and to confirm the diagnosis, an endoscopy with biopsy has to be performed. For the treatment of EGJ tumors in particular it is important to assess the spread of the tumor into the esophagus and the gastric cardia. Lymphatic dissemination and the possibility of distant metastases are further investigated by endoscopic ultrasound (EUS), computed tomography scan (CT-scan) of the neck, thorax and abdomen, ultrasound of the neck, and a PET-CT-scan. If local tumor ingrowth and/or distant metastases are suspected, biopsies can confirm this. Esophageal carcinomas are staged according to the 7th edition of the American Joint Committee on Cancer staging system (5). This classification predicts the overall survival (OS) rates per stage groupings for adenocarcinomas and squamous cell carcinomas separately. It shows a risk-adjusted 5-year OS ranging from 15% to 85% for adenocarcinomas and ranging from 15% to 75% for squamous cell carcinomas, depending on the stage group. Moreover, if tumors are staged with T1a or lower it should be treated by endomucosal resection and above stage T1a patients will undergo an esophageal or gastric resection. For the surgical classification of EGJ adenocarcinomas, the Siewert classification is used despite its limitations. This classification divides tumors in type I–III based on anatomical criteria (6):
- Type I: adenocarcinoma of the distal esophagus with the center located within 1 to 5 cm above the anatomic EGJ.
- Type II: true cardia carcinoma infiltrating from 1 centimeter on the side of the esophagus up to 2 cm below the GEJ in the stomach.
- Type III: subcardial gastric carcinoma with the tumor center between 2–5 cm below the GEJ.
Important is the definition of the EGJ or Z line and the gastric cardia. The histological and endoscopic definition of the EGJ may differ. This EGJ may be defined as the histological transition from the squamous epithelium of the esophagus to the gastric columnar epithelium or as the upper margin of the longitudinal folds of the stomach. It is clear that this transition does not occur exactly in the anatomical transition between the esophagus and stomach. Additionally, it is important to describe the level of the diaphragm in relation to the Z line, in patients with a type 2 hernia, these levels differ, and this may also influence the surgical approach. The word cardia, derived from the ancient Thracian city in the Gallipoli peninsula (narrowing between the Aegean and the Marmara sea) means for surgeons the area around the opening of the esophagus into the stomach. It is clear that these landmarks are not reliable with diseases such as cancer or hiatal hernias involving the EGJ. The UICC TNM 2016 has changed the definition of the EGJ cancers in relation with the Classification of 2009. Nowadays, a tumor the epicenter of which is within 2 cm of the EGJ and also extends into the esophagus is classified and staged using the esophageal scheme. Cancers involving the EGJ whose epicenter is within the proximal 2 cm of the cardia (Siewert I/II) are to be staged as esophageal. Cancers with the epicenter more than 2 cm distal from the EGJ will be staged using the gastric cancer TNM staging system even if the EGJ is involved (7).
An esophageal resection has always been the main curative treatment of esophageal cancer. Since the promising results of a randomized controlled trial that introduced neoadjuvant chemoradiotherapy (nCRTx) as an important additional treatment to surgery for esophageal cancer, survival rates have been improved importantly, with the increase in the 5-year OS rate from 33% to 47% (8,9). However, esophagectomies are still associated with high morbidity and mortality rates. To reduce the morbidity and to increase the quality of life (QoL), a minimally invasive esophagectomy approach (MIE) was introduced in the early nineties. Looking for evidence, the outcomes of the TIME trial showed advantages of MIE when compared to open esophagectomy (OE), such as a decreasing incidence of postoperative pulmonary infections, a shorter length of hospital stay and better QoL scores, indicating an improved patient recovery (10). Concerning oncological safety, no differences were found in OS and disease-free survival after 1- and 3-year follow-up, with a better QoL of physical components at 1- and 3-year follow-up (11,12). Therefore, MIE is currently considered to be a safe surgical procedure and the majority of patients with a resectable esophageal or EGJ-tumor should be operated by a minimally invasive approach. In other countries patients are perioperatively treated with chemotherapy according to the results of the MAGIC trial (13). Recently it has been investigated that both treatments are equally effective in terms of oncologic outcome [radical resection rates, lymphadenectomy, patterns of recurrent disease, and (disease-free) survival]. However, neoadjuvant chemoradiotherapy is associated with a considerably lower level of toxicity, and nCRT could therefore be the preferred neoadjuvant regimen (14). However, these are data from a retrospective study, no results from RCT’s are available yet.
Staging of adenocarcinomas of the EGJ
Siewert classification may be considered practical, but at the same time many EGJ tumors may be difficult to classify. Endoscopy, endosonography and CT scan are the diagnostic tools that we dispose to stage them. Parry et al. has studied the subject using a prospective database of 266 adenocarcinomas located in the EGJ. The overall accuracy for tumor localization was better for the combination of endoscopy and endosonography that for the CT scan. Especially difficult was to determine the exact location for type II. The nodal status, specifically the upper mediastinal was diagnosed better with the CT scan. Interesting is, that in only 3% of the patients the type of surgery that was planned preoperatively changed due to other intraoperative findings (15). Also in the data from the HIVEX trial we see this discrepancy in preoperative (endoscopy/EUS) and postoperative (histopathology) location of the tumor. Based on the preoperative endoscopic examination, 40 of 220 patients (18%) were classified with a type II tumor. Based on the postoperative pathologic examination of the resection specimen (gold standard), 115 of 205 patients (56%) were classified with a type II tumor (16). In only 3 patients, the operative plan changed from an esophageal resection to a total gastrectomy because of tumor location. An additional remark has to be made: in the time of the HIVEX trial a CT scan was not part of the initial staging yet, and only a conventional X-ray of the thorax and an ultrasound of the abdomen was performed. Additionally, no neoadjuvant therapy was administered or minimally invasive therapy was performed during this trial.
Treatment possibilities for EGJ tumors
The discussion about neoadjuvant or perioperative treatment for EGJ cancer has already been described in the introduction. In daily practice, most multidisciplinary teams will recommend neoadjuvant chemotherapy for EGJ tumors with Siewert type II and III, whereas they will choose for Chemoradiotherapy (nCRTx) for Siewert type I tumors, or, differently described, nCRT for patients with limited tumor ingrowth in the stomach (8,9,14). In the CROSS trial, only patients with 2 or less than 2 cm ingrowth in the stomach were included (8). After neoadjuvant therapy, there are three main surgical approaches for resection of EGJ tumors: the transthoracic esophagectomy (the 2-staged Ivor-Lewis esophagectomy or 3-staged McKeown esophagectomy), the transhiatal esophagectomy (THE) or the total gastrectomy. The decision for the surgical approach is based on the surgeon’s discretion, since there is no clear evidence about the best surgical approach in terms of morbidity and oncological outcomes yet. Generally, a laparoscopic gastrectomy is performed for Siewert type III. For type II, a MIE Ivor-Lewis procedure is the main choice (although some prefer a cervical anastomosis) and, as an alternative procedure a laparoscopic total gastrectomy with a high esophagogastrostomy anastomosis using the Orvil Circular Stapler [®Medtronic Inc., Minneapolis, MN, USA (17)] can be performed. Some surgeons indicate a laparoscopic THE with an anastomosis in the cervical area and in the case of extensive growth of the tumor along the lesser curvature an open esophageal and gastric resection followed by a colon interposition is indicated. The same MIE is indicated for the Siewert type I tumor.
Preparation for operation
Along with the use of neoadjuvant treatment, patients have to be optimally prepared for operation. This includes improvements of the general condition by optimal nutrition, and physiotherapy. Moreover, concerning the operative planning, it is important to study the radiotherapy charts (radiation field) to see if the proximal esophagus and mediastinum or parts of the stomach have been exposed to radiotherapy. The study of Goense shows increased complications with increased radiation of the gastric fundus, whereas the study by Koëter shows that the incidence of severe complications was associated with a high superior mediastinal planning target volume border (18,19). If the intrathoracic anastomosis is the preferred location of reconstruction, then the anastomosis should be created in a non-radiated area in order to reduce the leakage rate.
Surgical techniques
Two-staged minimally invasive Ivor-Lewis procedure
The Ivor-Lewis esophagectomy with intrathoracic anastomosis is a perfect operation for many infracarinal esophageal cancers, but has a high difficulty grade due to the creation of the intrathoracic anastomosis. The patient is intubated by selective intubation, which is only used for the anastomotic phase during thoracoscopy. The operation commences with a laparoscopy with extensive celiac trunk type D2 lymphadenectomy, gastric dissection along greater curvature with preservation of gastroepiploic vessels, creation of a gastric conduit by staplers and hiatal dissection, followed by right thoracoscopy in prone position of the patient, including dissection and mobilization of the esophagus, a mediastinal lymphadenectomy and division of the esophagus in the area between the carina and the azygos vein.
The gastric tube and the esophageal specimen are pulled into the thorax through the hiatus, followed by creation of an intrathoracic anastomosis. Although there are different types of intrathoracic anastomoses, no evidence posits one type of anastomosis as superior to another. As options, we can create a manual anastomosis or an end-to-side anastomosis using a conventional circular stapler (21, 25 or 29 mm) after a purse string suture on the esophageal stump or we can use a prepared Orvil device (®Medtronic Inc. Minneapolis, MN, USA). Additionally, the side-to-side anastomosis can be performed using a linear stapler, closing the anterior defect by a transversal suture using conventional suture material or the prepared V-loc Wound Closure™ [®Medtronic Inc. Minneapolis, MN, USA (20)]. Furthermore, the robot-assisted anastomosis (RAMIE) is increasingly used thereby permitting a high manual anastomosis in the apex of the thorax because of the ergonomy obtained by the robot (21).
Initiating the formation of a stapled anastomosis, a small thoracotomy is necessary to position the circular stapler into the gastric tube, whilst this is not required if a manual or linear stapler anastomosis is created. However, at the end of the procedure the specimen needs to be retrieved through a small thoracotomy incision anyway.
Concerning the type of intubation needed during the anastomosis phase, only a single-lumen intubation with two-lung ventilation (no collapse of the right lung is necessary) is required for the manual, RAMIE and the linear anastomosis. When performing a circular stapled anastomosis, a collapse of the right lung during anastomosis is essential, either by (I) selective intubation (one-lung ventilation); by (II) placing a Fogarty balloon catheter in the right bronchus to be inflated (during the anastomotic phase); or by (III) applying to the wound a protection device with a glove or a gel cap system along with maintaining a thoracic insufflation at 7–8 mmHg.
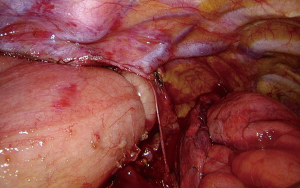
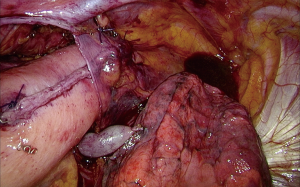
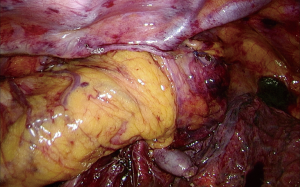
General principles for the anastomosis have to be assured, such as good vascular irrigation, no tension on the anastomosis and a watertight anastomosis. To get better outcomes of the anastomosis in which the esophagus is not covered by peritoneum, a new technique has been developed at our department. This new technique contains an anastomosis that is covered by a pleural flap followed by a wrap of omentum around the anastomosis (the ‘Flap and Wrap Technique’) and might be considered as an important improvement (Figures 1-3).
Performing a so called “Flap and Wrap” technique, a single stich is used to fix the created gastric tube behind the pleural flap (Figure 2). Therefore, the weight of the gastric tube is shifted to the pleural flap while this flap covers the gastric tube to prevent traction on the anastomosis and to protect it against the negative pressure in the thorax. The final part of the “Flap and Wrap” technique consists of wrapping the omentum around the anastomosis to ensure that it is fully covered, so that the anastomosis is completely contained with the omentum behind the pleura (Figure 3).
Three-staged minimally invasive McKeown procedure
The three-staged minimally invasive McKeown esophagectomy with cervical anastomosis is preferably used if there is a high mediastinal lymphadenopathy, or if intrathoracic anastomosis cannot be performed if tumor growth in proximal direction is too extended and the proximal residual esophagus is too short for an intrathoracic anastomosis.
This procedure is started through a right thoracoscopy in prone position with a single-lumen tracheal intubation, followed by upper laparoscopy and left cervical incision. To maintain a partial collapse of the right lung during thoracoscopy, the thoracic cavity must be insufflated with carbon dioxide at 6–8 mmHg. During thoracoscopy the esophagus is dissected and a mediastinal lymphadenectomy is performed as comparable to the previous described in the Ivor Lewis esophagectomy section.
After thoracoscopy, the patient is repositioned in French position to perform a laparoscopy with lymphadenectomy and formation of the gastric conduit as comparable with the Ivor-Lewis procedure. No Kocher maneuver or intervention to the pylorus is performed. Subsequently, a cervical anastomosis can be created starting with left cervical incision to decrease the risk of bilateral recurrent laryngeal nerve injury. The esophagus is divided and the gastric tube is pulled into the cervical region via the prevertebral route. A cervical end-to-end anastomosis is created manually. The advantages of a cervical anastomosis compared to an intrathoracic anastomosis (Ivor-Lewis procedure) are presumed better clinical management of leakages (e.g., by bedside opening of the cervical wound) and a larger proximal resection margin.
THE
This procedure is performed by laparoscopy and left cervical incision. Starting with transhiatal dissection of the esophagus (and tumor) from the pericardial sac and aortic planes up to the carina, it is followed by an extended D2 abdominal lymphadenectomy and gastric dissection. After dissection of the cervical esophagus, a small-assisting-protected laparotomy is performed to retrieve the whole specimen by stripping. Extracorporeal creation of gastric tube and resection of the specimen is then followed by pulling the gastric tube into the cervical wound where the anastomosis is made. Due to the transhiatal approach, the mediastinal lymphadenectomy is limited (16). Details of the surgical techniques have been published elsewhere (22).
Total gastrectomy
A laparoscopic total gastrectomy is performed through a laparoscopy in the upper abdomen, usually using 4 trocars and a separate incision for the Nathanson liver retractor or 5 trocars. A formal D2 lymphadenectomy is performed and the duodenum is divided by a stapler. The distal esophagus is mobilized and both pleura’s may or may not be opened, depending on the tumor. Intrathoracic, only the distal paraesophageal lymph nodes can be resected. After transection of the vagal nerves and the esophagus above the level of the tumor, an end-side esophagojejunostomy is created, usually using the Orvil (®Medtronic Inc. Minneapolis, MN, USA), since a purse string suture in the confined space of the narrow hiatus so high intrathoracic is usually not possible. It is advocated to send the resection specimen for frozen section before the anastomosis is created.
Evidence for surgical techniques
A prospective database of 266 consecutive patients with surgical resectable EGJ adenocarcinomas is analyzed by Parry et al. (22). Twenty-five % of patients had a Siewert I, 66% had type II tumor and 5% a type III tumor. In total, 86% were treated with esophagectomy and 14% with gastrectomy. Overall 5 years survival was 38% (41% of patients did not receive any neoadjuvant therapy). In type II patients the type of operation did not significantly influence OS on multivariate analysis. A positive circumferential resection margin (CRM) at the site of the esophagus was more common with gastrectomy than with esophagectomy (29% vs. 11%; P=0.025). No significant differences in morbidity, mortality or disease recurrence were found. In patients with type II, upper mediastinal nodal involvement (subcarinal, paratracheal, and aortopulmonary window) was found in 11% of the patients. In 34% of patients treated with esophagectomy, paraesophageal lymph nodes metastases were harvested compared with 5% of patients treated with gastrectomy. They conclude that in patients with a type II EGJ adenocarcinoma, a positive CRM was more common with gastrectomy. Esophagectomy provides for a more complete paraesophageal lymphadenectomy. Furthermore, the high prevalence of mediastinal nodal involvement indicates that a full lymphadenectomy of these stations should be considered.
In the era before the systematic use of neoadjuvant therapy, the Dutch HIVEX-trial compared the transhiatal approach versus the transthoracic approach for esophageal carcinoma (16). The trial revealed no differences in survival rates between the two approaches for EGJ tumors type II, whereas for type I the survival rates after transthoracic esophagectomy (TTE) were higher. In order to update this comparison in the current era of nCRTx and MIE, a comparable trial should be performed. The so-called IVORY-trial is currently in preparation and will compare minimally invasive transhiatal versus the transthoracic approach after nCRTx for distal and EGJ tumors type I and II according to the Siewert classification. The advantages of the transthoracic approach are an extensive esophageal dissection, a more complete mediastinal lymphadenectomy and possibly a better anastomosis. Because the thoracic anastomosis is more distally created than the cervical anastomosis, it is possible to perform a more extended gastric resection that in those type II tumors will help to achieve free resections margins. Moreover, the gastric tube may be shorter, but is better vascularized and consequently this may result in less morbidity, especially with less anastomotic leakages. It seems that Siewert type I tumors will metastasize to the paratracheal lymph nodes in 10% of the patients after neoadjuvant therapy, whilst this is less than 2% for type II tumors after neoadjuvant therapy (16). The role and extension of mediastinal lymphadenectomy is still controversial after the use of the nCRTx according to the CROSS-study (23).
Currently, after esophagectomy for cancer both cervical and intrathoracic esophagogastric anastomosis are used. Although a cervical anastomosis seems to be the best option for proximal and mid-esophageal tumors, yet a cervical anastomosis is followed by a higher frequency of anastomotic leakages—probably due to the worse perfusion at the top of the gastric tube (24). Additionally, after MIE with cervical anastomoses for distal or EGJ tumors, patients do have more complaints of dysphagia, dumping and regurgitation (25). This might be attributable to the higher incidence of strictures in these patients (26,27). A recent study showed that there seems to be a trend to create more Ivor-Lewis esophageal resections for EGJ-tumors than McKeown esophagectomies, involving in the period 2007 to 2014 an increase from 15% to 46% of intrathoracic anastomosis (28). In contrast, in the same period the incidence of three-stage McKeown esophagectomy decreased from 85% into 54% in the same study. To reach consensus about this ongoing topic the multicenter ICAN-trial has been launched to compare the short-term outcomes of transthoracic resections between patients with a cervical anastomosis (McKeown procedure) versus patients with an intrathoracic anastomosis (Ivor-Lewis procedure). The first patients have been recently included (29).
Concerning the surgical techniques, another important improvement with the aim to decrease the anastomotic leakage rate is the covering of the intrathoracic anastomosis by wrapping the omentum around it. A systematic review not only showed a significant decrease in the anastomotic leakages rate, but also in the length of hospital stay (30).
Another point of discussion is the position of patients, lateral or in prone position during Ivor-Lewis or McKeown esophagectomy. Initially, the lateral decubitus position with selective intubation and ventilation of one lung was preferred. However, the introduction of the prone position by Cuschieri in 1994 described many advantages of this position over the lateral decubitus position, such as no necessity for a complete lung block and a better visualization of the esophageal area (31). A recently published systematic review from Markar et al., suggests that the prone position is associated with less pulmonary complications, less blood loss, and a higher number of resected lymph nodes (32). It must be noted that there was some evidence of heterogeneity for the analysis of pulmonary complications and blood loss in this review.
Thus, not only the proper approach or the extension of lymphadenectomy, but also the ideal thoracoscopic position of the patient will be important items for future studies in patients who undergo MIE.
Authors’ own experience and type of anastomosis
In the Netherlands in the last years, most esophageal resections have been centralized in centers performing more than 20 esophageal resections yearly. Along with the increased use of minimally invasive surgery, the morbidity and mortality rates in patients have since decreased (10,33,34). Moreover, neoadjuvant therapies such as neoadjuvant chemoradiotherapy and chemotherapy are now extensively used. Data from the National Dutch Register (DUCA) show that the use of the thoracic esophagectomy increased from 47% in 2011 to 74% in 2015; that the implementation of total MIE increased from 32% to 72%; and that neoadjuvant therapy was administered in 91% of the registered 846 patients in 2015 (34).
As distal esophageal and EGJ adenocarcinomas are more than 80% of all esophageal cancers in the Netherlands, our department has increased the use of MIE Ivor-Lewis approach—a comparable development found at the other upper GI centers in The Netherlands. The first multicenter study about MIE Ivor-Lewis esophagectomy included more than 282 patients from six centers, performing different types of anastomoses, and showed a leakage in more than 15% of the patients with a 30-days mortality of 2.1% (35). This high-leakage rate obliged the surgeons to analyze these numbers, the learning curve and the cause of this rate. The result of this analysis in our department produced a change in the used anastomosis technique, resulting in the “Flap and Wrap” technique as described in the intrathoracic anastomosis paragraph of this chapter. This anastomosis technique is increasingly used in our Department, rising from 24.2% of the total esophageal resections in 2014 up to 72% of the resections in 2016. With this anastomosis anastomotic leakage rate has dropped to less than 5% for intrathoracic anastomosis with the “Flap and Wrap” technique, whereas leakages are still found in 20% of the patients with a cervical anastomosis without “Flap and Wrap” technique (unpublished data). Moreover, an important decrease of overall morbidity, reoperation rate, readmissions to the ICU and in ICU length of stay have been found (LOS). It seems that a lower incidence of the morbidity rates is associated to a higher frequency of transthoracic resections.
There is still no consensus about the ideal type of intrathoracic anastomosis. Different types of anastomoses are used, including manual, linear stapler, circular stapler and robot manual-assisted anastomoses. There is no evidence that one technique is better than the other, but general principles for anastomoses such as the need of a well-vascularized gastric tube, no tension or traction on the anastomosis and adequate patency are important factors to respect. In our experience, the flap and wrap anastomosis technique in which the anastomosis is covered behind a flap of pleura, so fixing it with stitches and having wrapped the entire anastomosis in omentum, hence accounts for a relative low leakage rate.
Outcome of published series
Data about morbidity and mortality rates following a MIE after neoadjuvant therapy for EGJ tumors specifically are scarce. The largest prospective study described the morbidity and mortality rates of a series of more than 1,000 patients in whom minimally invasive Ivor-Lewis esophagectomy and McKeown esophagectomy is performed (36). They did not find any differences in the frequency of anastomotic leakages requiring surgery (4% versus 5%, respectively). However, there were significant differences in the occurrence of vocal cord paralyses, with a higher incidence among McKeown esophagectomies than Ivor-Lewis procedures (8% versus 1%, respectively). Moreover, no difference in 30-days mortality was reported (0.9% versus 2.5%, respectively). In general, there seems to be a trend towards lower morbidity rates among MIE Ivor-Lewis resections as compared to MIE McKeown resections and THE, but results of evidence based studies comparing these different approaches are lacking (25,36).
Outcomes for minimally invasive TTE esophagectomy
Two studies that reported short-term outcomes of MIE Ivor-Lewis resections for EGJ tumors specifically are recently reported. One study reported a multicentric series of patients with a distal or a EGJ tumor of the esophagus treated by MIE Ivor Lewis and the other study compared two cohorts, MIE Ivor Lewis and MIE McKeown procedure. Straatman et al. investigated the short-term outcomes of 282 patients among 6 different European centers who underwent minimally invasive Ivor-Lewis esophagectomy for only distal and EGJ tumors and showed a morbidity rate of 44% (35). The most frequent complications were anastomotic leakages (15.2%), pulmonary complications (13.1%), and cardiac complications (4.3%). Perioperative outcomes were: a median operative time of 333 minutes, 242mL blood loss (median) and 1.8% conversions to open Ivor-Lewis esophagectomy. Radical resections (R0) were performed in 93% of the patients. Further postoperative outcomes were a median length of stay of 12 days, and a median length of ICU stay of 2 days and the 30-day morbidity was 2.1%.
The second retrospective study compared 356 patients who underwent Ivor-Lewis MIE (intrathoracic anastomosis, n=210) with patients who underwent McKeown or Transhiatal MIE (cervical anastomosis, n=146) (25). The incidence of recurrent laryngeal nerve palsy was 14.4% after a cervical anastomosis and 0% after an intrathoracic anastomosis. Dysphagia, dumping, and regurgitation were reported less frequently after creation of an intrathoracic anastomosis. Dilatation of benign strictures occurred in 43.8% of the cervical anastomoses versus in 6.2% of the intrathoracic anastomoses. If a benign stricture was identified, it was dilated for a median of 4 times in the cervical group and only once in the intrathoracic group. Anastomotic leakage for which reoperation was required occurred in 8.2% after cervical anastomosis and in 11.4% after intrathoracic anastomosis (not significant). Median ICU stay, hospital stay, in-hospital mortality, 30-day mortality, and 90-day mortality were similar between the groups (not significant). They conclude that MIE with an intrathoracic anastomosis is associated with better functional results with less dysphagia, less benign anastomotic strictures requiring fewer dilatations, and a lower incidence of recurrent laryngeal nerve palsy as compared to MIE with cervical anastomoses. Other postoperative morbidity and mortality did not differ between the groups.
Conclusions
The incidence of cancer of the distal esophagus and EGJ is increasing, whereby adenocarcinomas are paramount in the Western World (up to 80% of all esophageal carcinomas). The advantages of MIE as a treatment of esophageal or a EGJ cancer in comparison with OE are important improvements in the short-term outcomes, such as less blood loss, less respiratory infections, a better postoperative QoL, with similar 1- and 3-year survival rates. Tumors of the EGJ are classified by the Siewert classification, despite all its limitations. For Siewert type III a total or proximal gastrectomy is the indicated resection whereas for type II both the extended gastrectomy or the Ivor Lewis approach are the two possibilities. Importantly, a R0 radical resection should be performed. For Siewert types I and II, the Ivor-Lewis esophagectomy is an ideal operation following neoadjuvant therapy. There is an important increase in the use of this approach per year with 41% of all esophageal resections in 2015 in the Netherlands. This approach includes an intrathoracic anastomosis after laparoscopic preparation of the gastric tube and thoracoscopic esophageal resection and intrathoracic anastomosis in prone position. Other surgical options for an esophageal resection include a THE or the (transthoracic) McKeown resection, in which a cervical anastomosis is performed. The transhiatal approach has its limitations due to the incapacity to perform a mediastinal lymphadenectomy and is reserved to frail patients with a distal or EGJ tumor. In a non-randomized study, patients with intrathoracic anastomoses showed a better functional outcome than the group with cervical anastomoses along with less dysphagia, less benign strictures and lower incidence than recurrent nerve palsy. Our experience with intrathoracic anastomoses accompanied by the ‘Flap and Wrap’ technique limited the postoperative leakage rate to approximately 5% whereas the leakage rate in patients with cervical anastomoses remains up to 20%.
Additionally, a more extended lymphadenectomy is possible in transthoracic surgery compared to THE or gastrectomy, and lymph node status is still the most important predictor for survival.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, et al. Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer 2013;49:1374-403. [Crossref] [PubMed]
- Arnold M, Soerjomataram I, Ferlay J, et al. Global incidence of oesophageal cancer by histological subtype in 2012. Gut 2015;64:381-7. [Crossref] [PubMed]
- Abbas G, Krasna M. Overview of esophageal cancer. Ann Cardiothorac Surg 2017;6:131-6. [Crossref] [PubMed]
- Pennathur A, Gibson MK, Jobe BA, et al. Oesophageal carcinoma. Lancet 2013;381:400-12. [Crossref] [PubMed]
- Rice TW, Blackstone EH, Rusch VW, et al. 7th Edition of the AJCC Cancer Staging Manual: esophagus and esophagogastric junction. Ann Surg Oncol 2010;17:1721-4.
- Rüdiger Siewert J, Feith M, Werner M, et al. Adenocarcinoma of the esophagogastric junction: results of surgical therapy based on anatomical/topographic classification in 1,002 consecutive patients. Ann Surg 2000;232:353-61. [Crossref] [PubMed]
- Deng JY, Liang H. Adenocarcinoma of esophagogastric junction. Chin J Cancer Res 2014;26:362-3. [PubMed]
- Shapiro J, van Lanschot JJ, Hulshof MC, et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): Long-term results of a randomised controlled trial. Lancet Oncol 2015;16:1090-8. [Crossref] [PubMed]
- van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 2012;366:2074-84. [Crossref] [PubMed]
- Biere SS, Van Berge Henegouwen MI, Maas KW, et al. Minimally invasive versus open oesophagectomy for patients with oesophageal cancer: A multicentre, open-label, randomised controlled trial. Lancet 2012;379:1887-92. [Crossref] [PubMed]
- Maas KW, Cuesta MA, Van Berge Henegouwen MI, et al. Quality of Life and Late Complications After Minimally Invasive Compared to Open Esophagectomy: Results of a Randomized Trial. World J Surg 2015;39:1986-93. [Crossref] [PubMed]
- Straatman J, van der Wielen N, Cuesta MA, et al. Minimally Invasive Versus Open Esophageal Resection: Three-year Follow-up of the Previously Reported Randomized Controlled Trial: the TIME Trial. Ann Surg 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Cunningham D, Allum WH, Stenning SP, et al. Perioperative Chemotherapy versus Surgery Alone for Resectable Gastroesophageal Cancer. N Engl J Med 2006;355:11-20. [Crossref] [PubMed]
- Anderegg MC, van der Sluis PC, Ruurda JP, et al. Preoperative Chemoradiotherapy Versus Perioperative Chemotherapy for Patients With Resectable Esophageal or Gastroesophageal Junction Adenocarcinoma. Ann Surg Oncol 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Parry K, Haverkamp L, Bruijnen RC, et al. Staging of adenocarcinoma of the gastroesophageal junction. Eur J Surg Oncol 2016;42:400-6. [Crossref] [PubMed]
- Omloo JM, Lagarde SM, Hulscher JB, et al. Extended transthoracic resection compared with limited transhiatal resection for adenocarcinoma of the mid/distal esophagus: five-year survival of a randomized clinical trial. Ann Surg 2007;246:992-1000. [Crossref] [PubMed]
- Medtronic Inc. (2013) Orvil Circular Stapler.
- Koëter M, Kathiravetpillai N, Gooszen JA, et al. Influence of the Extent and Dose of Radiation on Complications After Neoadjuvant Chemoradiation and Subsequent Esophagectomy With Gastric Tube Reconstruction With a Cervical Anastomosis. Int J Radiat Oncol Biol Phys 2017;97:813-21. [Crossref] [PubMed]
- Goense L, van Rossum PS, Ruurda JP, et al. Radiation to the Gastric Fundus increases the risk of Anastomotic Leakage after Esophagectomy. Ann Thorac Surg 2016;102:1798-804. [Crossref] [PubMed]
- Medtronic Inc. (2015) V-loc Wound Closure.
- van Hillegersberg R, Seesing MF, Brenkman HJ, et al. Robot-assisted minimally invasive esophagectomy. Chirurg 2016;87:635-42. [Crossref] [PubMed]
- Parry K, Haverkamp L, Bruijnen RC, et al. Surgical treatment of adenocarcinomas of the gastro-esophageal junction. Ann Surg Oncol 2015;22:597-603. [Crossref] [PubMed]
- Koen Talsma A, Shapiro J, Looman CW, et al. Lymph Node Retrieval During Esophagectomy With and Without Neoadjuvant Chemoradiotherapy. Ann Surg 2014;260:786-92. [Crossref] [PubMed]
- Scheepers JJ, Veenhof XA, van der Peet DL, et al. Laparoscopic transhiatal resection for malignancies of the distal esophagus: Outcome of the first 50 resected patients. Surgery 2008;143:278-85. [Crossref] [PubMed]
- van Workum F, van der Maas J, van den Wildenberg F, et al. Improved Functional Results After Minimally Invasive Esophagectomy: Intrathoracic Versus Cervical Anastomosis. Ann Thorac Surg 2017;103:267-73. [Crossref] [PubMed]
- Biere SS, Maas KW, Cuesta MA, et al. Cervical or thoracic anastomosis after esophagectomy for cancer: A systematic review and meta-analysis. Dig Surg 2011;28:29-35. [Crossref] [PubMed]
- Markar SR, Arya S, Karthikesalingam A, et al. Technical Factors that Affect Anastomotic Integrity Following Esophagectomy: Systematic Review and Meta-analysis. Ann Surg Oncol 2013;20:4274-81. [Crossref] [PubMed]
- Haverkamp L, Seesing MF, Ruurda JP, et al. Worldwide trends in surgical techniques in the treatment of esophageal and gastroesophageal junction cancer. Dis Esophagus 2017;30:1-7. [PubMed]
- van Workum F, Bouwense SA, Luyer MD, et al. Intrathoracic versus Cervical ANastomosis after minimally invasive esophagectomy for esophageal cancer: study protocol of the ICAN randomized controlled trial. Trials 2016;17:505. [Crossref] [PubMed]
- Yuan Y, Zeng X, Hu Y, et al. Omentoplasty for oesophagogastrostomy after oesophagectomy. Cochrane Database Syst Rev 2014.CD008446. [PubMed]
- Cuschieri A. Thoracoscopic subtotal oesophagectomy. Endosc Surg Allied Technol 1994;2:21-5. [PubMed]
- Markar SR, Wiggins T, Antonowicz S, et al. Minimally invasive esophagectomy: Lateral decubitus vs. prone positioning; Systematic review and pooled analysis. Surg Oncol 2015;24:212-9. [Crossref] [PubMed]
- Wouters MW, Karim-Kos HE, le Cessie S, et al. Centralization of esophageal cancer surgery: does it improve clinical outcome? Ann Surg Oncol 2009;16:1789-98. [Crossref] [PubMed]
- Dutch Upper GI Cancer Audit 2015. 2015. Available online: https://www.dica.nl/jaarrapportage-2015/duca
- Straatman J, van der Wielen N, Nieuwenhuijzen GA, et al. Techniques and short-term outcomes for total minimally invasive Ivor Lewis esophageal resection in distal esophageal and gastroesophageal junction cancers: pooled data from six European centers. Surg Endosc 2017;31:119-26. [Crossref] [PubMed]
- Luketich JD, Pennathur A, Awais O, et al. Outcomes after Minimaly Invasive Esophagectomy: review over 1000 patients. Ann Surg 2012;256:95-103. [Crossref] [PubMed]