Evaluation of surgical approaches to anatomical segmentectomies: the transition to minimal invasive surgery improves hospital outcomes
Introduction
Anatomical segmentectomy is considered a reasonable option in the management of lung nodules, especially in patients with reduced preoperative respiratory reserve or when diagnostic doubt persists. Despite requiring more technical precision and anatomical awareness in comparison to other resection types, segmentectomy can result in reduced post-operative complications (1,2) whilst preserving equivalent oncological outcomes (3,4).
Operative trends in the modern era of thoracic surgery have moved towards minimally invasive approaches, with video-assisted thoracoscopic surgery (VATS) techniques being adopted gradually. VATS resection, which can be performed via multiple or single ports, has superior short-term postoperative morbidity (5), however remains technically demanding especially for those new to the method. We have transitioned towards VATS as our preferred way of approach, moving towards a uniportal technique universally by all surgeons in our unit. Single port access VATS pulmonary procedures were initially described by Rocco et al. in 2004 (6) and further developed by Gonzalez-Rivas et al. (7) who performed the first single port VATS lobectomy. This approach may have some potential benefits over the traditional multiport technique including ergonomics and a more direct view (8), and early reports indeed reveal them to be at least equivalent (9,10).
We aimed to evaluate this transition of surgical approaches to anatomical segmentectomies in our tertiary thoracic surgical unit over a three-year time period and compare peri- and post-operative outcomes.
Methods
This work was approved and presented to the Audit and Clinical Governance Group at the Cardiothoracic Surgical unit at Sheffield Teaching Hospitals NHS Trust, UK. All patients who underwent anatomical segmentectomy between January 2013 and December 2015 in our unit were identified using a prospectively maintained departmental database. Data was collected on patient characteristics including age, preoperative lung function and co-existing morbidity. Details on operative approach and surgical technique were taken from individual operation notes, and histopathology reports were reviewed for specifics on quality of resection margins and extent of lymph node dissection. Postoperative outcomes in terms of length of stay and short-term complications were also evaluated. Thoracotomy after VATS intraoperative biopsy was considered as a conversion for the purposes of the study. The patients identified were divided into three groups according to their year of operation: group A (January to December 2013), group B (January to December 2014) and group C (January to December 2015), as well as according to surgical approach: thoracotomy or VATS. Patients undergoing wedge resections were not included in this study. We ensured that all segmentectomies included involved division of all three elements: arterial, venous and bronchial.
Anatomical segmentectomy was performed via an open (thoracotomy) or VATS approach involving one or more ports. Under general anaesthesia with single-lung ventilation, the patient was placed in the lateral decubitus position. Once the pleural cavity was entered, the segmental pulmonary arterial and venous branches were identified, isolated and ligated by hand or with the use of an endostapler. The segmental bronchus was then located and confirmed via an inflation test prior to division. The parenchymal segmental plane was also identified at the time of inflation and subsequently divided using endostaplers. Lymph node dissection was performed and a single 28F chest drain was inserted into the pleural cavity, prior to closure.
All patients operated on in our unit after May 2014 followed an enhanced recovery program postoperatively. This involved regional analgesic methods introduced at the time of surgery, specifically intraoperative intercostal blockade with local anaesthesia and insertion of a paravertebral catheter supplying a continuous infusion of bupivacaine 0.5% lasting at least 24 hours duration following surgery. At the discretion of the anaesthetist, selected patients were provided with patient controlled analgesia intravenously or via an epidural depending on the surgical approach and patient’s preoperative state. Patients were encouraged to convert to oral analgesia alternatives at 24 hours postoperatively. Chest drains were removed if no air leak was documented for six or more hours and with drainage of less than 300 mL/day as per departmental protocol.
The measured outcomes were clinical: mortality, complications, duration of intercostal drainage, hospital readmission and prolonged air leak (over seven days); use of resources: hospital stay, need for Intensive Care Unit admission; and oncological: complete R0 excision, lymph node stations explored and pathological nodal stage. Pain outcomes included the use of epidural, paravertebral and intravenous opioids; pain scores at pre-determined times day 1 and 2, median pain scores at day 1 and 2, and maximum pain scores on day 1 and 2.
Statistical analysis was undertaken using SPSS (version 11). Results were expressed as median (range) or number (percentage) where appropriate, unless otherwise stated. Chi squared (with exact test when indicated) and Wilcoxon’s rank tests were used for qualitative and quantitative data respectively. P values of less than 0.05 were considered statistically significant throughout.
Results
A total of 86 patients [56 females and 30 males, median age 70 years (range, 43 to 83 years); median FEV1 of 78% predicted (range, 41% to 126%) and median TLCO of 59% predicted (range, 18% to 122%)] underwent anatomical segmentectomy during the study period with a similar number of procedures performed each year (27, 28 and 31 cases respectively). There were no significant differences in patient characteristics between the three groups (Table 1).

Full table
A notable transition in surgical approach was observed over the course of the study period (Figure 1), with higher proportions of cases completed via single port as the study progressed. Four surgeons formed the team (with some variability during the years). Two of these surgeons originally performed multiple port VATS procedures with gradual conversion to a single-port technique during the study period, whilst the other two were single-port trained VATS surgeons.
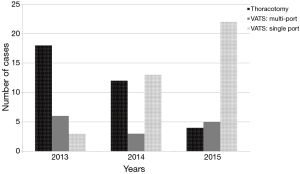
Thirty-four operations were completed by thoracotomy and 52 (60%) performed by VATS. Of these 73% were via single port and comprised the majority of cases by 2015. There were four conversions to thoracotomy (7%) due to technical difficulties or lack of progress, and five cases proceeding to planned thoracotomy after a VATS wedge and positive intraoperative frozen section. These nine conversions occurred in four cases after multiport VATS (22%) and in five after uniportal VATS (11%) (P=0.4).
The operations were performed in all lobes. Middle lobectomies were not included in this series. The most common segmentectomies performed were apical segmentectomies equally distributed in both lower lobes, and upper trisegmentectomies of the left upper lobe, 30% each of the total. Other segmentectomies included lingulectomies, and basal segmentectomies of the lower lobes.
Primary indications for segmentectomy were the need to preserve lung parenchyma in cancer cases or for biopsy of a small PET-positive nodule of unknown histology. Overall there were 70 (81%) cases of primary lung cancer, 5 (6%) were metastases from other organs and 11 (13%) were confirmed to be benign disease. Of the primary lung cancer, 61% were adenocarcinoma, 22% squamous carcinoma and 4% neuroendocrine tumours. The proportion of primary lung malignancies was not significantly different between the three groups (74%, 96%, 77% respectively). Similarly, there were no differences in the pathological stages, completeness of resection or lymphatic exploration of the malignant cases in the time periods (Table 2). The use of segmentectomy comprised around 15% of the lung cancer cases performed in our unit during the time period.

Full table
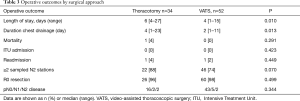
Intraoperatively we did not encounter any haemorrhagic events and had only one in-hospital death in the open group over the study period. This was due to pneumonia and respiratory failure. Most common complications were prolonged air leak greater than seven days (17%), lower respiratory tract infection (7%), atrial fibrillation (3%) and wound infection (3%). Complications and readmissions rates (2.2%) were similar between the groups. Patients had a significantly shorter hospital stay in 2015 with a median length of stay of 4 days (range, 1–15 days), versus a median stay of 6 days (range, 3–27 days) in previous years (P=0.01). Duration of chest drainage also reduced over time, with a median two days duration of chest drain in situ in 2015 (range, 1–24 days) (P=0.01). The proportion of prolonged air leaks over seven days also decreased significantly after the first year of the study (P=0.001) (Table 2).
Unsurprisingly these results per year were also visible when groups were compared by surgical approach, open thoracotomy versus VATS, as described in Table 3.
Full table
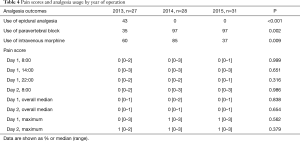
Bedside observations were monitored at regular intervals by nursing staff and included a recording of pain using a Four-scale Verbal Pain Score ranging from zero to three (0= no pain, 3= severe pain). As the use of an epidural catheter was eliminated from clinical practice after 2013 there was an initial increase in the use of intravenous analgesia. This usage was reduced in the final year and local audits of analgesia practice have continued to show this. Despite the reduction in pre-emptive analgesia prescription, we have maintained excellent pain control as reported by patients. Pain scores at determined times (at rest and after mobilisation), absolute pain scores and median pain scores over the postoperative periods were similar between the groups, with median pain scores of 0–1 achieved near universally (Table 4).
Full table
Discussion
The advantages of VATS over open surgery have been shown in a number of studies evaluating results following lobectomy (11,12). However reasonably sized series of uniportal segmentectomy are sparingly reported (13-16). Proponents of the technique suggest that by entering just one intercostal space via a four cm incision, single port VATS can provide the viewing angle experienced with open surgery with minimal morbidity to the patient (17).
Our results found that the transition to single port VATS is associated with a shorter hospital stay and chest tube duration without compromise in postoperative complications or early oncological metrics. This is in keeping with other studies comparing VATS to open segmentectomies. Atkins et al. (18) found a significantly decreased length of stay of 4.3±3.0 days following VATS segmentectomy. The same was true for Schuchert et al. (19) whose analysis of 225 consecutive segmentectomies also found a reduced incidence of pulmonary complications in the VATS group. Shorter chest tube duration following VATS segmentectomy has likewise been reported (2).
As described in the results section, the improvements we have observed over time in terms of hospital stay and use of analgesia are also seen when comparing thoracotomy and VATS.
VATS segmentectomy is now more frequently utilised in the management of lung cancer resection (19-23), although is often reserved for patients with poor operability (24). Studies thus far have shown equivalent oncological outcomes to open segmentectomy with no significant differences in locoregional recurrence or overall survival (25). A recent report by Ghaly et al. (23) retrospectively compared 91 cases of VATS segmentectomy for stage 1 non-small cell lung cancer with 102 cases performed via thoracotomy over a 13-year time period. They found no clear differences in the quality of resection margins or mediastinal nodal stations sampled between the two groups, although noted a greater number of hilar lymph node sampling via VATS. In our series we found more extensive hilar dissection in the open group, but not when only patients with primary lung cancer are compared.
In addition to the reduced length of hospital stay, we have also found a decline in analgesic requirements without compromising patients’ pain control. By monitoring pain scores at rest and after mobilisation, recording maximum pain scores and complete median scores, we believe that our assessment is the most completely reported in this field to date.
Our report describes a limited series of cases in terms of anatomical sublobar resections and lacks long-term follow-up to be able to assess oncological outcomes. However, we believe that it provides a good insight and evaluation of a changing service from open to uniportal VATS surgery and can be of benefit to other institutions.
Our increasing adoption of minimally invasive techniques over the study period, suggests that as their experience developed, our surgeons were able to offer single port segmentectomy to more patients, hence overtaking open surgery as the primary approach to anatomical sublobar resection in our unit. We conclude that the transition to single port VATS segmentectomies is both feasible and safe and can maintain or improve short-term postoperative outcomes with a reduction in the use of hospital resources.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This work was approved and presented to the Audit and Clinical Governance Group at the Cardiothoracic Surgical unit at Sheffield Teaching Hospitals NHS Trust, UK.
References
- Kilic A, Schuchert MJ, Pettiford BL, et al. Anatomic segmentectomy for stage I non-small cell lung cancer in the elderly. Ann Thorac Surg 2009;87:1662-6; discussion 1667-8.
- Leshnower BG, Miller DL, Fernandez FG, et al. Video-assisted thoracoscopic surgery segmentectomy: a safe and effective procedure. Ann Thorac Surg 2010;89:1571-6. [Crossref] [PubMed]
- Schuchert MJ, Pettiford BL, Keeley S, et al. Anatomic segmentectomy in the treatment of stage I non-small cell lung cancer. Ann Thorac Surg 2007;84:926-32; discussion 932-3. [Crossref] [PubMed]
- Shapiro M, Weiser TS, Wisnivesky JP, et al. Thoracoscopic segmentectomy compares favorably with thoracoscopic lobectomy for patients with small stage I lung cancer. J Thorac Cardiovasc Surg 2009;137:1388-93. [Crossref] [PubMed]
- Paul S, Altorki NK, Sheng S, et al. Thoracoscopic lobectomy is associated with lower morbidity than open lobectomy: a propensity-matched analysis from the STS database. J Thorac Cardiovasc Surg 2010;139:366-78. [Crossref] [PubMed]
- Rocco G, Martin-Ucar A, Passera E. Uniportal VATS wedge pulmonary resections. Ann Thorac Surg 2004;77:726-8. [Crossref] [PubMed]
- Gonzalez-Rivas D, de la Torre M, Fernandez R, et al. Single-port video-assisted thoracoscopic left upper lobectomy. Interact Cardiovasc Thorac Surg 2011;13:539-41. [Crossref] [PubMed]
- Bertolaccini L, Rocco G, Viti A, et al. Geometrical characteristics of uniportal VATS. J Thorac Dis 2013;5 Suppl 3:S214-6. [PubMed]
- Perna V, Carvajal AF, Torrecilla JA, et al. Uniportal video-assisted thoracoscopic lobectomy versus other video-assisted thoracoscopic lobectomy techniques: a randomized study. Eur J Cardiothorac Surg 2016;50:411-5. [Crossref] [PubMed]
- Shen Y, Wang H, Feng M, et al. Single- versus multiple-port thoracoscopic lobectomy for lung cancer: a propensity-matched studydagger. Eur J Cardiothorac Surg 2016;49 Suppl 1:i48-53. [PubMed]
- Cao C, Manganas C, Ang SC, et al. A meta-analysis of unmatched and matched patients comparing video-assisted thoracoscopic lobectomy and conventional open lobectomy. Ann Cardiothorac Surg 2012;1:16-23. [PubMed]
- Whitson BA, Groth SS, Duval SJ, et al. Surgery for early-stage non-small cell lung cancer: a systematic review of the video-assisted thoracoscopic surgery versus thoracotomy approaches to lobectomy. Ann Thorac Surg 2008;86:2008-16; discussion 2016-8.
- Gonzalez-Rivas D, Mendez L, Delgado M, et al. Uniportal video-assisted thoracoscopic anatomic segmentectomy. J Thorac Dis 2013;5 Suppl 3:S226-33. [PubMed]
- Han KN, Kim HK, Lee HJ, et al. Single-port video-assisted thoracoscopic pulmonary segmentectomy: a report on 30 cases†. Eur J Cardiothorac Surg 2016;49 Suppl 1:i42-7. [PubMed]
- Shih CS, Liu CC, Liu ZY, et al. Comparing the postoperative outcomes of video-assisted thoracoscopic surgery (VATS) segmentectomy using a multi-port technique versus a single-port technique for primary lung cancer. J Thorac Dis 2016;8:S287-94. [PubMed]
- Cheng K, Zheng B, Zhang S, et al. Feasibility and learning curve of uniportal video-assisted thoracoscopic segmentectomy. J Thorac Dis 2016;8:S229-34. [PubMed]
- Gonzalez-Rivas D, Fieira E, Mendez L, et al. Single-port video-assisted thoracoscopic anatomic segmentectomy and right upper lobectomy. Eur J Cardiothorac Surg 2012;42:e169-71. [Crossref] [PubMed]
- Atkins BZ, Harpole DH Jr, Mangum JH, et al. Pulmonary segmentectomy by thoracotomy or thoracoscopy: reduced hospital length of stay with a minimally-invasive approach. Ann Thorac Surg 2007;84:1107-12; discussion 12-3. [Crossref] [PubMed]
- Schuchert MJ, Pettiford BL, Pennathur A, et al. Anatomic segmentectomy for stage I non-small-cell lung cancer: comparison of video-assisted thoracic surgery versus open approach. J Thorac Cardiovasc Surg 2009;138:1318-25.e1. [Crossref] [PubMed]
- Shiraishi T, Shirakusa T, Iwasaki A, et al. Video-assisted thoracoscopic surgery (VATS) segmentectomy for small peripheral lung cancer tumors: intermediate results. Surg Endosc 2004;18:1657-62. [PubMed]
- Martin-Ucar AE, Nakas A, Pilling JE, et al. A case-matched study of anatomical segmentectomy versus lobectomy for stage I lung cancer in high-risk patients. Eur J Cardiothorac Surg 2005;27:675-9. [Crossref] [PubMed]
- Yang CF, D'Amico TA. Thoracoscopic segmentectomy for lung cancer. Ann Thorac Surg 2012;94:668-81. [Crossref] [PubMed]
- Ghaly G, Kamel M, Nasar A, et al. Video-assisted thoracoscopic surgery is a safe and effective alternative to thoracotomy for anatomical segmentectomy in patients with clinical stage I non-small cell lung cancer. Ann Thorac Surg 2016;101:465-72. [Crossref] [PubMed]
- Lau KK, Martin-Ucar AE, Nakas A, et al. Lung cancer surgery in the breathless patient--the benefits of avoiding the gold standard. Eur J Cardiothorac Surg 2010;38:6-13. [Crossref] [PubMed]
- Yang CF, D'Amico TA. Open, thoracoscopic and robotic segmentectomy for lung cancer. Ann Cardiothorac Surg 2014;3:142-52. [PubMed]


