Reversible pulmonary hypertension with unclear etiology associated with suspected viral infection
Introduction
Pulmonary hypertension (PH) is defined by a mean pulmonary artery pressure (PAPm) ≥25 mmHg at rest as assessed by right heart catheterization (RHC) (1). Clinical classification of PH includes five groups: pulmonary arterial hypertension (PAH), PH due to left heart disease, PH due to lung diseases and/or hypoxemia, chronic thromboembolic PH, and PH with unclear and/or multifactorial mechanism. PH is categorized according to clinical presentation, pathological findings, hemodynamic significances, and treatment strategy (2). It is important to identify PH etiology as several etiologies, as shown, can be progressive and fatal if untreated. This case describes a young woman who presented with acute onset dyspnea due to right heart failure and anemia. Diagnostic tests for the secondary causes were negative, including congenital intra-cardiac shunts, left-sided heart diseases, lung diseases, including hypoxemia and pulmonary thromboembolism, collagen vascular disease, HIV infection, and hyperthyroidism. She did not take any drugs or toxins that could lead to PH. The possible etiologies of PH in our case were idiopathic PAH (Class I) or unclear multifocal mechanisms (Class 5). Anemia was the initial finding of pancytopenia that later occurred in this patient due to mild splenomegaly, which was suspected to be caused by a viral illness. After correction of anemia and supportive treatment, PH and right side heart failure were also improved. Therefore, we report a case of a reversible PH due to possibly unclear multifactorial mechanisms associated with a viral infection. We also report a review of the literature.
Case presentation
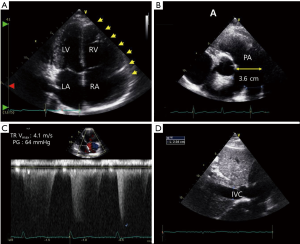
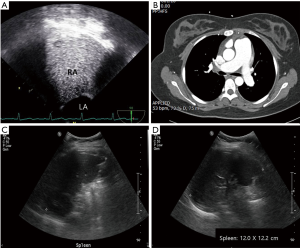
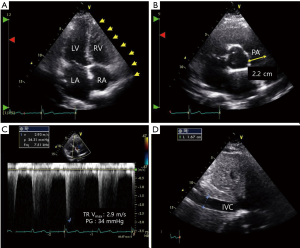
A 21-year-old previously healthy woman presented with recent onset of dyspnea on exertion and intermittent palpitation. She had upper respiratory infection (URI) 2 weeks before the onset of symptoms. She was a student and slightly overweight. She denied exposure to any drugs including illicit drugs, alcohol, or tobacco. She did not have weight loss or weight gain. Physical examination revealed a blood pressure of 100/60 mmHg and tachycardia of 112 beats/min. Her appearance was relatively normal, but anemic conjunctivae were noted. On auscultation, clear breath sounds were audible in both whole lung field, and regular heart beats without a murmur. There was no palpable mass or tenderness on abdominal examination. Twelve-lead electrocardiography showed sinus tachycardia and non-specific ST-segment/T-wave changes. Laboratory findings revealed severe anemia (level of hemoglobin: 4.9 g/dL), but normal white blood cells (5,070/µL, segment neutrophils: 59%) and platelet count (179,000/µL). Anemia study revealed iron deficiency anemia (ferritin: 2.4 ng/mL, TIBC: 464 µg/dL) maybe due to menorrhagia, but gynecologic examination did not reveal any significant structural abnormalities. Brain natriuretic peptide (BNP) level was elevated (299 pg/mL). Arterial blood gas analysis revealed respiratory alkalosis (pH: 7.48, PaCO2: 30 mmHg, PaO2: 79 mmHg, BE: −1.0 mmol/L, HCO3: 22, SaO2: 96%), which was apparently associated with tachypnea. Chest roentgenogram showed mild cardiomegaly (CT ratio: 0.55) without any lung problem. We performed transthoracic echocardiography (TTE), because we suspected she suffered from high output heart failure associated with severe anemia (3). TTE showed enlarged right atrium (RA), and right ventricle (RV), and dilated pulmonary artery (PA) (Figure 1). Pressure gradient (PG) between RA and RV was 63 mmHg calculated by tricuspid regurgitation (TR) peat velocity and estimated PA pressure (PAP) was calculated as 78 mmHg by adding estimated RA pressure of 15 mmHg (Figure 1) (4), which is indicative of severe PH. However, left atrial and ventricular (LA and LV) sizes were normal, and both systolic and diastolic functions were also normal (EF: 65%, E/E’: 4). Transesophageal echocardiography (TEE) was performed to evaluate intra-cardiac shunt, but no evidence of intra-cardiac shunt was confirmed with agitates saline (Figure 2A). Chronic thromboembolic PH and other PA obstructions were ruled out by chest computed tomography (Figure 2B). Later, pancytopenia occurred unexpectedly (white blood cells: 3,470/µL, Hb: 8.2 g/dL, platelets: 83,000/µL) based on follow up laboratory findings after RBC transfusion with iron replacement. We suspected pancytopenia was caused by splenomegaly associated with a viral illness, as she had URI 2 weeks ago. Abdominal sonography showed mild splenomegaly (Figure 2C,D). Supportive treatment with maintenance of iron supplements, resolved her dyspnea and intermittent palpitation and complete blood cell profile was completely recovered after 2 months (white blood cells: 6,140/µL, Hb: 13.8 g/dL, platelets: 211,000/µL). Follow-up TTE showed normalized RA, RV, and PA with decreased estimated PAP (39 mmHg) (Figure 3).
Discussion
Our patient had a reversible PH with unknown etiology associated with pancytopenia caused by splenomegaly in the clinical setting of viral infection. There were some case reports of reversible PH in association with hyperthyroidism (5,6) and vitamin C deficiency (7). Adverse cardiovascular effects of hyperthyroidism are well established. High output biventricular heart failure is common (8), but also isolated right heart failure with variable degrees of TR and PH is less frequently reported. However, our patient showed normal thyroid function and she did not have any signs of vitamin C deficiency. She was a previously healthy young student with slightly overweight who had regular, heathy diet. Intra-cardiac shunt and other possible adult congenital heart diseases were excluded by TEE and agitated saline. Even though we did not perform lung perfusion scan, chronic thromboembolic PH and other PA obstructions could be excluded by chest computed tomography and normal arterial blood gas analysis showed normal arterial oxygen. In addition, connective tissue disease or HIV infection were not possible in this case. PH is defined as an increase in PAPm 25 mmHg at rest assessed by RHC, and pulmonary capillary wedge pressure (PCWP) is needed to classify pre-capillary or post-capillary PH (1). In the present case, the patient was a young women and it was a major limitation that she refused RHC because of fear of the invasive test. However, Doppler echocardiographic measurement of systolic PAP using TR peak velocity, estimated RA pressure, and modified Bernoulli equation in the absence of pulmonary flow obstruction is a reliable method (9). In addition, if LV systolic or diastolic dysfunction, left atrial enlargement, E/E; ratio >15, or valvular disease exist, post capillary PH must be suspected (9). In this case, TTE showed normal left cardiac size and function (EF: 65%, E/E’: 4). Therefore, PH due to other causes could be excluded for our patient.
The patient with splenomegaly gives a diagnostic challenge to a wide variety of clinicians, because conditions associated with splenomegaly are extensive (10). Splenomegaly can be defined as increased size of the spleen on the ultrasound exam (11). Craniocaudal length is most often used to measure splenic size, and its upper normal limit varies from 11 to 14 cm (12). Her spleen size measured by ultrasound was 12 cm, which means borderline enlarged spleen when considering her short height (147 cm). The spleen was not palpable on physical examination but clinical presentation (history of URI, initially anemia, and later pancytopenia) was the clue for splenomegaly. Her symptoms were improved and she recovered from pancytopenia. We did not perform bone marrow biopsy. Some viral infections such as HIV, human herpesvirus-8, hepatitis B and C, are associated with angioproliferation and the development of PH (13). Serum viral marker tests were performed to examine if any of above mentioned viral infections were present in this patient, but the results were all negative.
Our patient had very unique presentation of reversible PH with unknown etiology associated with acute viral infection and transient pancytopenia. Here we are reporting our clinical findings and reviewing the literatures.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Galiè N, Humbert M, Vachiery JL, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Heart J 2016;37:67-119. [Crossref] [PubMed]
- Simonneau G, Galiè N, Rubin LJ, et al. Clinical classification of pulmonary hypertension. J Am Coll Cardiol 2004;43:5S-12S. [Crossref] [PubMed]
- Anand IS, Florea VG. High output cardiac failure. Curr Treat Options Cardiovasc Med 2001;3:151-9. [Crossref] [PubMed]
- Parasuraman S, Walker S, Loudon BL, et al. Assessment of pulmonary artery pressure by echocardiography-A comprehensive review. Int J Cardiol Heart Vasc 2016;12:45-51. [Crossref] [PubMed]
- Lozano HF, Sharma CN. Reversible pulmonary hypertension, tricuspid regurgitation and right-sided heart failure associated with hyperthyroidism: case report and review of the literature. Cardiol Rev 2004;12:299-305. [Crossref] [PubMed]
- Ismail HM. Reversible pulmonary hypertension and isolated right-sided heart failure associated with hyperthyroidism. J Gen Intern Med 2007;22:148-50. [Crossref] [PubMed]
- Kupari M, Rapola J. Reversible pulmonary hypertension associated with vitamin C deficiency. Chest 2012;142:225-7. [Crossref] [PubMed]
- Forfar JC, Muir AL, Sawer SA, et al. Abnormal left ventricular function in hyperthyroidism: evidence for a possible reversible cardiomyopathy. N Engl J Med 1982;307:1165-70. [Crossref] [PubMed]
- Bossone E, D'Andrea A, D'Alto M, et al. Echocardiography in pulmonary arterial hypertension: from diagnosis to prognosis. J Am Soc Echocardiogr 2013;26:1-14. [Crossref] [PubMed]
- Osler W. Discussion on splenic enlargements other than leukaemic. Brit Med J 1908;2:1151-8.
- Lamb PM, Lund A, Kanagasabay RR, et al. Spleen size: how well do linear ultrasound measurements correlate with threedimensional CT volume assessments? Br J Radiol 2002;75:573-7. [Crossref] [PubMed]
- Peddu P, Shah M, Sidhu PS. Splenic abnormalities: a comparative review of ultrasound, microbubble-enhanced ultrasound and computed tomography. Clin Radiol 2004;59:777-92. [Crossref] [PubMed]
- Cool CD, Voelkel NF, Bull T. Viral infection and pulmonary hypertension: is there an association? Expert Rev Respir Med 2011;5:207-16. [Crossref] [PubMed]


