Is a 5-mm diameter an appropriate cut-off value for the diagnosis of atypical adenomatous hyperplasia and adenocarcinoma in situ on chest computed tomography and pathological examination?
Introduction
The International Multidisciplinary Classification of Lung Adenocarcinoma was proposed by the International Association for the Study of Lung Cancer, the American Thoracic Society, and the European Respiratory Society in February, 2011 (1). Under this classification, both atypical adenomatous hyperplasia (AAH) and adenocarcinoma in situ (AIS) are classified as preinvasive lesions. On imaging, AAH and most cases of AIS appear as pure ground-glass nodules (pGGNs). AAH is a precancerous lesion that can lead to lung adenocarcinoma. About one-third of all AAH cases progress to AIS and then to minimally invasive adenocarcinoma (MIA) and finally to invasive adenocarcinoma (2). Since the International Multidisciplinary Classification of Lung Adenocarcinoma was introduced in clinical practice, few studies have investigated whether lesion size, density, and other morphological characteristics can aid in the detection of AIS and AAH (3-5). Moreover, the impact of a collapsed lung after surgical excision on the accuracy of pGGN evaluation has not been researched. In the present study, we analyzed 80 preinvasive lesions in 76 patients by using thin-section computed tomography (TSCT) and pathological examination in an attempt to improve the diagnostic accuracy of preinvasive lung lesions and provide valuable information for clinical treatment.
Methods
Patients
We retrospectively analyzed the data of 76 patients with 80 pathologically confirmed preinvasive lesions that appeared as pGGNs on imaging in our hospital between January 2013 and January 2016. All patients underwent spiral CT scanning at least once before the surgery. We compared patient and imaging characteristics between those with AAH and those with AIS. This retrospective study was approved by our institutional review board, which waived informed consent.
CT scanning
CT scans were performed using a Siemens Somatom Definitions AS+ 128-row and a 64-row multi-slice CT device (Germany). With the patient in a supine position, scanning was performed from the apex pulmonis until the disappearance of both costophrenic angles. Patients were taught deep inspiration and breath holding before the examination. Scanning was performed during deep inspiration, with no breath holding. The scanning parameters were as follows: tube voltage, 120 kV; automatic modulation of tube current; matrix, 512×512; collected thickness, 1.0 mm; pitch, 1.0; and conventional slice thickness, 2.0 mm. The results were constructed with a standard algorithm. 1-mm targeting scan was performed in partial lesions. The morphology of the lesion and its relationship with adjacent structures were evaluated in the sagittal and coronal plane.
CT evaluation
The CT images were independently reviewed by two experienced radiologists with more than 5 years of diagnostic experience in chest CT scans. The final conclusion was reached by consensus. The following imaging characteristics were evaluated: number of lung nodules, classification of nodules (solid, pGGN, or partially solid nodule), nodule diameter, internal characteristics (density, uniformity, mean CT value, vacuole sign, and air bronchogram sign), and peripheral characteristics (clear rim, regularity, lobulation, spiculation, and pleural indentation sign). The nodular diameter was the average value of the longest maximum cross-section of the lesion measured by the two radiologists. The region of interest to measure the CT value of the lesion covered the whole nodule through the maximum cross-section and avoided blood vessels and bronchi. The measurement was performed three times each by the two radiologists, and the average CT value was used. The vacuole sign is a gassy, lucent shadow measuring <5 mm in diameter within a lesion; it may be single or multiple. A spiculated nodule is one with thin, short, straight lines arranged radially around the nodular rim.
Pathological diagnostic criteria
On gross pathological examination, the maximum diameter of the lesion was measured using a ruler. The measurement was repeated three times, and the average value was calculated. AAH is a small (usually ≤0.5 cm), limited, mild-to-moderate atypical proliferation of type II alveolar epithelial cells and/or Clara cells in the alveolar wall or the respiratory bronchiolar wall. AIS is a small (≤3 cm), localized adenocarcinoma that grows along preexisting alveolar structures (lepidic growth) and lacks stromal, vascular, or pleural invasion.
Statistical analysis
Data were analyzed using SPSS v16.0 software. Differences in the diameters measured on TSCT and gross pathological examination between AAH and AIS were statistically analyzed using the Wilcoxon paired test. Significant results were then analyzed using a receiver operating characteristic (ROC) curve to determine the best cut-off value and measure the area under the curve (AUC). Differences in mean CT value and ages between the AAH and AIS groups were statistically analyzed using the independent-samples t-test. Characteristics such as lobular structure were analyzed using the Pearson χ2 test. When the sample number was less than 40 or the theoretical frequency was 1 or more but less than 5, a correction formula was applied. When the sample number was less than 40 or the theoretical frequency was less than 1, the Fisher exact test was applied. P<0.05 was considered to indicate significant differences.
Results
General characteristics
Of the 76 patients, 31 had AAH (33 pGGNs; 9 men and 22 women; mean age, 53.2 years; range, 33–73 years). The AAH lesions were detected on routine examination or examination for other diseases in 22 patients (Figure 1). In two patients, the lesions were first detected during a postoperative review for lung cancer. In seven patients, the lesions were detected during investigation for dyspnea and cough. The remaining 45 patients had AIS (47 pGGNs; 17 men and 28 women; mean age, 53.8 years; range, 19–77 years) (Figure 2). The lesions were detected on routine examination or examination for other diseases in 33 patients, on postoperative review of lung cancer in 3 patients, and on investigation for dyspnea and cough in 9 patients.
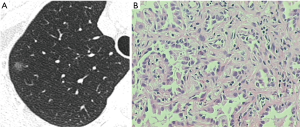
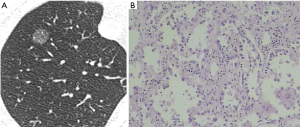
Both AAH and AIS were common in middle-aged and elderly people, especially women (Table 1). No statistical differences were found in age and sex between the AAH and AIS groups (P>0.05).

Full table
Internal and peripheral characteristics of AAH and AIS
Among the 31 AAH patients, 29 had a single lesion, while two had multiple lesions. The mean CT value of the 33 pGGNs was −607.07±87.67 HU. There were 7 cases of uniform density (21.2%), 32 cases of clear rim (97%), 27 cases of regular shape (81.8%), 3 cases of lobulation (9.1%), 5 cases of spiculation (15.2%), and 3 cases of pleural indentation (9.1%). Among the 45 AIS patients, 33 had a single lesion, while 12 had multiple lesions. The mean CT value of the 47 lesions was −592.46±106.67 HU. There were 12 cases of uniform density (25.5%), 40 cases of clear rim (85.1%), 34 cases of regular shape (72.3%), 9 cases of vacuole sign (19.1%), 10 cases of lobulation (21.3%), 4 cases of spiculation (8.5%), 7 cases of pleural indentation sign (14.9%), and 3 cases of air bronchogram sign (6.4%). There was a statistically significant difference in TSCT diameter and vacuole sign between the AAH and AIS groups (P<0.001 and P=0.021, respectively). Both the vacuole sign and the air bronchogram sign were only observed in the AIS group (Table 2).
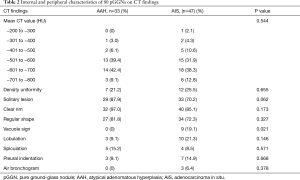
Full table
Comparison of nodular diameter between AAH and AIS
On TSCT, the average diameter of AAH lesions (6.0±1.64 mm; range, 3.0–9.7 mm) was significantly lower than that of AIS lesions (8.7±3.16 mm; range, 3.5–19.2 mm; P<0.001). On gross pathological examination too, the average diameter of AAH lesions (4.6±1.99 mm; range, 2.0–10.0 mm) was significantly lower than that of AIS lesions (6.8±2.06 mm; range, 4.0–14.0 mm; P<0.001; Table 3).

Full table
Comparison of nodular diameters measured using TSCT and gross pathology
The nodular diameter measured using TSCT was significantly greater than that measured on gross pathological examination in the case of both AAH (6.0±1.64 vs. 4.6±1.99 mm) and AIS (8.7±3.16 vs. 6.8±2.06 mm; P<0.001). This difference was also maintained in the overall cohort of all 80 preinvasive lesions (7.5±2.88 vs. 5.9±2.31 mm; P<0.001; Table 4).

Full table
ROC curve analysis of AAH and AIS diameters measured using TSCT and gross pathology
ROC curve analysis indicated that the AUC of nodular diameter as measured on TSCT was 0.792 (95% confidence interval: 0.693–0.891). The best diagnostic cut-off value correlated with the highest Youden index, and was 6.99 mm (sensitivity, 76.6%; specificity, 78.8%; Figure 3).

The AUC of nodular diameter as measured on gross pathological examination was 0.794 (95% confidence interval: 0.689–0.899). The best diagnostic cut-off value correlated with the highest Youden index, and was 4.5 mm (sensitivity, 93.6%; specificity, 60.6%; Figure 4).
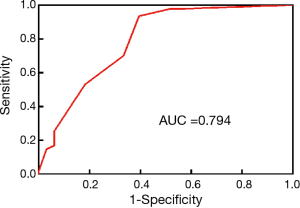
Discussion
With the wide application of spiral CT, improved awareness of medical examinations among the public, and an increase in air pollution, the detection rate of pulmonary nodules, especially GGNs, has been steadily increasing. These GGNs, were single or multiple, or accompanied with pulmonary solid nodules (mostly lung cancer). Most of the nodules were small, with a diameter of <10 mm. The management of these GGNs, including early diagnosis, treatment, long-term follow-up, psychological factors, radiation dose, cost, and other patient problems, often encountered in clinical practice, must be explored.
The Fleischner Society (6) recommends that pGGNs with a diameter of <5 mm do not need CT follow-up, while those with a diameter of >5 mm require annual review for 3 years. However, other reports indicate that most continuously existing pGGNs with a diameter of <5 mm are AAH; in fact, at least one-third of pGGNs gradually increase in size and/or density during follow-up (2). Furthermore, most postoperative specimens are pathologically confirmed as AIS, MIA, or invasive adenocarcinoma (3,7-10). These reports are consistent with our clinical findings. Therefore, regular follow-up of pGGNs is necessary. Early detection and treatment can greatly improve prognosis. In our study, the preinvasive lesions were mostly located around lung cancer, and were mostly treated using surgical resection at the same time.
The imaging and pathological characteristics of AAH and AIS have been illustrated in the International Multidisciplinary Classification of Lung Adenocarcinoma published in 2011 (1): these lesions appear as small pGGNs (AAH diameter, usually ≤5 mm; AIS diameter, ≤3.0 cm). In our study, both AAH and AIS appeared as pGGNs, which is consistent with previous reports (7,8). In our study, lesion size was an independent factor for the diagnosis of malignant lesions, with larger lesions having a greater chance of being malignant; this is consistent with the findings of previous studies (9,10). In our study, the difference in AAH diameter as measured using TSCT and gross pathology was 1.4 mm, while the difference for AIS was 1.7 mm. The reason for this difference may be that the postoperative collapsed lung affects the accuracy of the measurement, resulting in a larger diameter on TSCT. The best cut-off value of nodular diameter was 6.99 mm on TSCT and 4.5 mm on gross pathology, both of which differ from the 5-mm diameter recommended in the International Multidisciplinary Classification of Lung Adenocarcinoma. Therefore, the impact of imaging and gross pathological findings should take into consideration during the diagnosis of AAH and AIS.
In our study, the vacuole sign was only seen in the AIS group. The vacuole sign is a gassy, lucent shadow with a diameter of <5 mm, which is common in early lung adenocarcinoma. Thus, the vacuole sign can aid in the differentiation between AAH and AIS.
The 2011 International Multidisciplinary Classification of Lung Adenocarcinoma also states that AAH is the earliest preinvasive lesion of lung adenomatous carcinoma detected on TSCT and characteristically exhibits very blurred pGGNs. While the typical appearance of AIS is pGGN, few cases also appear as solid nodules or consolidation. The density of AIS is slightly higher than that of AAH (1). In our study, both AAH and AIS appeared as pGGNs with a slight difference in mean CT values (15 HU). The density of AIS was slightly higher than that of AAH. In addition, AAH did not differ from AIS in terms of average CT value, uniformity of density, shape, rim, lobulation, spiculation, pleural indentation sign, and aerated bronchus sign.
The main shortcoming of our study is the small sample size. We intend to expand more cases in the future and summarize the findings in a larger sample size.
In conclusion, most AAH and AIS lesions appeared as pGGNs. Lesion size and the vacuole sign are beneficial in the diagnosis of AAH and AIS. The vacuole sign was common in AIS. There was a slight difference in mean CT value (15 HU) between AAH and AIS. The density of AIS was slightly higher than that of AAH. The best diagnostic cut-off value of nodular diameter for differentiation between AAH and AIS was 6.99 mm on TSCT and 4.5 mm on gross pathology.
Acknowledgements
Funding: The research was supported by Open Project of State Key Laboratory of Respiratory Disease (SKLRD2016OP011) and Science and Technology Planning Project of Guangdong Province (Grant No. 2014A020212340 and 2017A040405065).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This retrospective study was approved by our institutional review board [Medical Ethical Review (MER) 2017-38], which waived informed consent.
References
- Travis WD, Brambilla E, Noguchi M, et al. International association for the study of lung cancer/american thoracic society/european respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 2011;6:244-85. [Crossref] [PubMed]
- Aoki T. Growth of pure ground-glass lung nodule detected at computed tomography. J Thorac Dis 2015;7:E326-8. [PubMed]
- Lim HJ, Ahn S, Lee KS, et al. Persistent pure ground-glass opacity lung nodules ≥ 10 mm in diameter at CT scan: histopathologic comparisons and prognostic implications. Chest 2013;144:1291-9. [Crossref] [PubMed]
- Lee CT. What do we know about ground-glass opacity nodules in the lung? Transl Lung Cancer Res 2015;4:656-9. [PubMed]
- Si MJ, Tao XF, Du GY, et al. Thin-section computed tomography-histopathologic comparisons of pulmonary focal interstitial fibrosis, atypical adenomatous hyperplasia, adenocarcinoma in situ, and minimally invasive adenocarcinoma with pure ground-glass opacity. Eur J Radiol 2016;85:1708-15. [Crossref] [PubMed]
- Naidich DP, Bankier AA, MacMahon H, et al. Recommendations for the management of subsolid pulmonary nodules detected at CT: a statement from the Fleischner Society. Radiology 2013;266:304-17. [Crossref] [PubMed]
- Liu Y, Sun H, Zhou F, et al. Imaging features of TSCT predict the classification of pulmonary preinvasive lesion, minimally and invasive adenocarcinoma presented as ground glass nodules. Lung Cancer 2017;108:192-7. [Crossref] [PubMed]
- Kitami A, Sano F, Hayashi S, et al. Correlation between histological invasiveness and the computed tomography value in pure ground-glass nodules. Surg Today 2016;46:593-8. [Crossref] [PubMed]
- Liu LH, Liu M, Wei R, et al. CT findings of persistent pure ground glass opacity: can we predict the invasiveness? Asian Pac J Cancer Prev 2015;16:1925-8. [Crossref] [PubMed]
- Zhang Y, Qiang JW, Ye JD, et al. High resolution CT in differentiating minimally invasive component in early lung adenocarcinoma. Lung Cancer 2014;84:236-41. [Crossref] [PubMed]


