Comparison of survival between lung cancer patients receiving single or multiple-incision thoracoscopic surgery
Introduction
Lung cancer is the leading cause of cancer death worldwide (1,2). Although long-term survival remains poor for patients with metastatic disease, complete surgical resection is potentially curative for patients with early-stage lung cancer. Thoracoscopic surgery for lung cancer has been demonstrated to result in faster postoperative recovery and fewer postoperative complications than traditional open surgery (3-7).
Although thoracoscopic surgery is recommended for the treatment of early-stage lung cancer, the surgical procedures such as the number of chest wall incisions have yet to be standardized. Theoretically, fewer chest wall incisions should result in less chest trauma, postoperative pain and inflammation. Previous case series have shown that thoracoscopic resection for lung cancer performed via a single incision is a safe and technically feasible procedure resulting in satisfactory short-term perioperative outcomes (8-15). Furthermore, other studies have shown that short-term outcome after resection via a single incision is similar to that after multiple-incision thoracoscopic surgery (16-20). However, to the best of our knowledge, no studies have investigated whether there are differences in long-term survival between the two thoracoscopic techniques. Therefore, we conducted a propensity-matched analysis to compare long-term overall survival and disease-free survival between patients with lung cancer who received single-incision thoracoscopic surgery and those who underwent resection via a multiple-incision approach.
Methods
This study was conducted at the Koo Foundation Sun Yat-Sen Cancer Center. Thoracoscopic anatomic resections have been performed at the center for the past 11 years, beginning with three-port thoracoscopy in 2005, a two-port technique in 2007 and a single-port approach in 2009. The protocols for performing single-incision and multiple-incision thoracoscopic lobectomy and segmentectomy used at the center are discussed in detail in our previous publications (10,11,16,17). This study was approved by the institutional review board of the Koo Foundation Sun Yat-Sen Cancer Center (KFSYSCC-IRB No. 20150623A).
In this study, we reviewed data on all patients who underwent lobectomy at our institution during the period 2000–2014. Surgical indications for lung cancer at our hospital include clinical T1–3 stage cancer with biopsy proven N0–1 disease or single-station N2 disease without evidence of distant metastasis as recommended by the National Comprehensive Cancer Network guidelines for non-small cell lung cancer (21). Lobectomy is the standard surgical resection for lung cancer although some selected patients with poor pulmonary function or other major comorbidities that contraindicate lobectomy are treated with sublobar resection. Intentional segmentectomy is always performed in patients with peripherally located cT1N0M0 lung cancers smaller than 2 cm in diameter.
We identified a total of 819 patients who received surgical resection for lung cancer during the study period. Exclusion criteria included: (I) multiple lung cancer (n=50); (II) adenocarcinoma in situ or stage IV disease (n=39); (III) scheduled thoracotomy (n=181); (IV) conversion to thoracotomy during thoracoscopic surgery (n=2); (V) thoracoscopic surgery via a subxiphoid incision (n=13); and (VI) in-hospital death within 30 days of surgery (n=2). A total of 532 patients fulfilled the inclusion criteria and were included for analysis. The patients were divided into a single-incision group and a multiple-incision group and propensity score matching was performed to eliminate differences in basic demographics between the two groups.
The clinical characteristics evaluated in this study included age, gender, comorbidity score, cell type, tumor size, pathologic stage, operative procedure, tumor location, operative time, intraoperative blood loss, retrieved numbers of lymph node, length of hospital stay, disease status and survival rate. Surgical mortality was defined as death within 30 days after the operation or during the same hospitalization. Comorbidity score was calculated based on the following comorbidities: chronic obstructive pulmonary disease, diabetes mellitus, tuberculosis or other cancer. A score of 0 indicated that the patient did not have any of those conditions; a score of 1 indicated that the patient had one of those conditions; and a score of 2 indicated that the patient had at least two of those conditions. Preoperative staging and radiologic work-up included history taking, physical examination, computed tomographic imaging of the chest and upper abdomen, and positron emission tomography-computed tomography. Tumors were staged according to the American Joint Committee on Cancer (AJCC) tumor-node-metastasis (TNM) staging system (AJCC staging manual, 7th edition).
Surgical technique
All surgeries were performed with double-lumen endotracheal tube intubation under general anesthesia. Patients were positioned in the decubitus position, and the surgeon stood at the anterior side of the patient. Thoracoscopic surgery was performed with three incisions, included 3- to 5-cm mini-thoracotomy at the anterior axillary line at the 4th or 5th intercostal space, a port at the 8th intercostal space in the mid-axillary line for thoracoscope and a 10-mm accessory incision at the tip of the scapula. The incision at the tip of the scapula was omitted for 2-incision thoracoscopic surgery. With the growing endoscopic experience, the single incision (3- to 5-cm mini-thoracotomy) was made in the 6th intercostal space at the anterior axillary line. The Alexis® wound protector (XS size) was routinely used at the mini-thoracotomy wound for all patients. The surgical techniques of the single-incision approach are also similar to those of the multiple-incision method. Pulmonary vessels and bronchus were divided individually with endoscopic staplers under thoracoscopic guidance. Mediastinal lymphadenectomy facilitated with a Harmonic scalpel (Ethicon Endo-Surgery, Inc., Cincinnati, OH, USA) and several simple traction methods (10). A 10-mm, 30-degree thoracoscopic video camera was also routinely used.
Statistical analysis
Patients who received single-incision thoracoscopic surgery were matched on propensity score at a 1:2 ratio to those who received multiple-incision thoracoscopic pulmonary resection to minimize bias due to the nonrandom allocation of treatments among patients. Propensity scores were estimated using a logistic model that included gender, age, gender, cell type, tumor size, tumor location, pathological cancer stage, and operation types as covariates.
Demographic data and clinical information were compared before and after the matching between those who underwent single-incision thoracoscopic surgery and those who received multiple-incision thoracoscopic surgery for lung cancer. The independent t-test was used to compare differences in continuous variables and the chi-square test was used for the comparison of categorical variables. Overall survival was calculated based on the time period starting on the date of surgery for lung cancer and ending with death or on April 2016. Disease-free survival was defined as the interval between the date of surgery and the date of recurrence or the last follow-up on April 2016. Kaplan-Meier survival analysis was used to compare overall survival and disease-free survival between the un-matched and matched groups. Differences in survival were determined by the log-rank test. Univariate and multivariate Cox proportional hazards regression analyses were conducted before propensity score matching. Age, gender, comorbidity score, pathologic T stage, pathologic N stage, tumor location, operation type, cell type and number of incisions were included into a multivariate regression model to investigate the impact of surgical technique on overall survival and disease-free survival. A P value <0.05 was considered to indicate statistical significance. All statistical analyses were performed with the statistical package SAS for Windows (Version 9.2, SAS, Cary, NC, USA).
Results
A total of 532 patients who were treated via single-incision or multiple-incision thoracoscopic surgery were analyzed before propensity score matching. Of them, 150 (28.2%) received single-incision thoracoscopic surgery and 382 (61.8%) were treated with multiple-incision thoracoscopic surgery. The un-matched single-incision group comprised a higher proportion of females and younger patients and had a higher rate of segmentectomy than the multiple-incision thoracoscopic group. Propensity matching resulted in 138 patients in the single-incision group and 276 patients in the multiple-incision group. The clinical characteristics of the un-matched and matched patients are presented in Table 1.
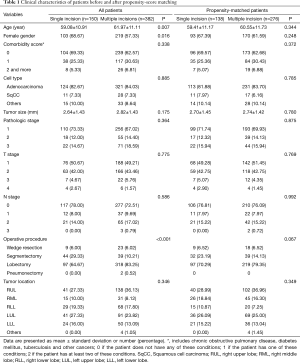
Full table
The mean follow-up time was 29.35 months in the single-incision group and 81.30 months in the multiple-incision group. The outcome variables are summarized in supplement Table S1. Analysis of the matched patients showed that that the single-incision group had a significantly shorter operative time, shorter length of hospital stay, a lower postoperative mortality rate and a lower rate of disease progression (defined by either death, local recurrence or metastasis) than the multiple-incision group.
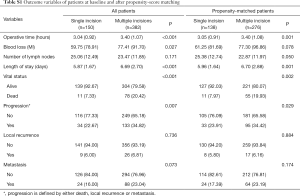
Full table
The overall survival and disease-free survival rates for unmatched and matched patients are summarized in Tables S2,S3, respectively. The 1-, 3-, and 5-year overall survival rates for matched patients in the single-incision group were 97.8%, 88.5%, and 88.5%, respectively. For matched patients in the multiple-incision group the 1-, 3-, and 5-year overall survival rates were 97.5%, 87.3%, and 78.3%, respectively. Patients treated with single-incision thoracoscopic surgery had better 5-year overall survival than those treated with multiple-incision thoracoscopic surgery (P=0.027). However, disease-free survival was similar between the two groups before and after matching (Table S3).

Full table
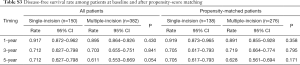
Full table
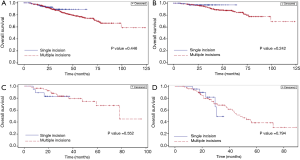
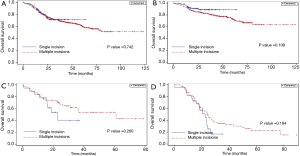
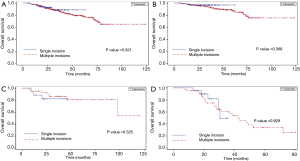
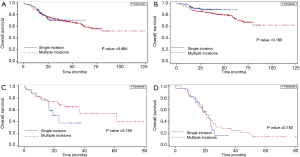
The Kaplan-Meier curves for overall survival among the 532 un-matched patients stratified by number of incisions and by pathologic stage are shown in Figure 1A. There were no significant differences in survival between the single-incision and multiple-incision groups (Figure 1A) and no significant survival differences between the two groups of patients stratified by disease stage I, II or III (Figure 1B-D). The Kaplan-Meier curves for disease-free survival among the unmatched patients stratified by number of incisions and by pathologic stage are shown in Figure 2A-D. Similarly, there were no significant differences in disease-free survival between the single-incision and multiple-incision groups (Figure 2A) and no significant survival differences between the two groups of patients stratified by disease stage (Figure 2B-D). Differences in overall survival between matched patients stratified by number of incisions and pathologic stage were also analyzed and the results are presented in Figure 3A-D. Kaplan-Meier analysis revealed that there was no significant difference in overall survival between the two groups (Figure 3A). In addition, there were no significant differences in overall survival between the two groups stratified by pathologic stage (Figure 3B-D). Disease-free survival was also similar between matched patients stratified by incision number (Figure 4A) and pathologic stage (Figure 4B-D).
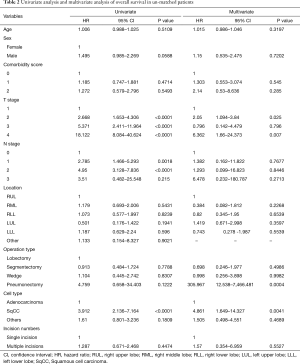
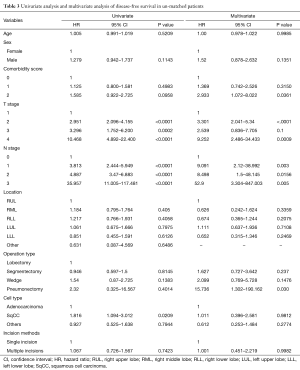
Significant predictors of overall survival among un-matched patients in the univariate analysis (pathologic T stage, pathologic N stage, and cell type) were included in a multiple logistic-regression model to identify the most important predictors of overall survival. The results revealed that pathologic T stage, pneumonectomy and cell type were independent prognostic factors. The number of incisions, however, was not a significant predictor in either the univariate or multivariate analysis (Table 2). In the univariate analysis of disease-free survival in un-matched patients, pathologic T stage, pathologic N stage, and cell type were prognostic factors. In the multivariate analysis, however, only pathologic T stage and pathologic N stage remained predictors of disease-free survival. The number of incisions was not a significant predictor of disease-free survival in the univariate or multivariate analysis (Table 3).
Full table
Full table
Discussion
This study investigated the long-term overall survival and disease-free survival of patients with lung cancer who were treated with either single-incision or multiple-incision thoracoscopic resection. Propensity matching was conducted to create groups of patients who were well-matched with respect to age, sex, cell type, comorbidity score, tumor size, pathologic stage, operative procedure and tumor location. We found that overall survival and disease-free survival were similar between the two thoracoscopic techniques.
Gonzalez-Rivas and his colleagues reported that uniportal thoracoscopic lobectomy results in favorable perioperative outcomes (9). Similarly, we also previously reported that single-incision thoracoscopic lobectomy and segmentectomy good postoperative outcomes (10). In a large series (n=1,063) of patients who underwent single-incision thoracoscopic pulmonary resection Xie et al. and demonstrated that a single-incision approach results in less post-operative wound pain, faster recovery and better cosmetic results (13). Many thoracic surgeons across the world have adopted single-incision thoracoscopic surgery as the treatment of choice for patients with lung cancer (8-20).
Previous studies have demonstrated that overall survival after thoracoscopic surgery for lung cancer is equivalent to and in some cases better than that after thoracotomy (3). However, a few studies have reported on the short-term survival rates associated with single-incision thoracoscopic surgery for lung cancer. For example, Ng et al. (12) demonstrated that the overall 2-year disease-free survival rates among 150 consecutive patients who underwent single-port thoracoscopic resection for lung cancer were 96% for stage I disease and 83% for stage II or greater lung cancer. Also, Gonzalez-Rivas et al. (14) reported that the 30-month survival rate was 90% for early-stage lung cancer and 74% for advanced-stage lung cancer (>5 cm, or T3 or T4 or tumors requiring neoadjuvant chemotherapy). However, to the best of our knowledge, no studies have compared the long-term survival rates achieved with single-incision thoracoscopic surgery with those achieved with multiple-incision thoracoscopic surgery for lung cancer.
Therefore, we conducted a comparative study to investigate the influence of surgical incision numbers on long-term survival in patients with lung cancer. We found that patients in the single-incision group had better 5-year overall survival than those in the multiple-incision group before and after propensity score matching. Disease-free survival, however, was similar between the two groups before and after matching. The mortality rate was lower and the degree of disease progression was less severe in matched patients in the single-incision group than those in the multiple-incision group. This is a reasonable finding because the mean follow-up period in the single-incision group was markedly shorter (29.35 months) than that in the multiple-incision group (81.30 months) as we only began performing single-incision thoracoscopic surgery in 2009. The univariate and multivariate analyses showed that the number of chest wall incisions did not influence disease-free survival. We also found that long-term overall survival and disease-free survival were equivalent for both procedures when patients were stratified by pathologic stage. Our results, therefore, show that single-incision thoracoscopic surgery does not compromise long-term survival of patients with lung cancer.
A number of studies have directly compared short-term perioperative outcome between single-incision and multiple-incision thoracoscopic surgery for lung cancer (16-20). In two of our previous studies we demonstrated that single-incision thoracoscopic surgery did not compromise perioperative safety of patients (16,17). Mu et al. compared uniportal and triportal thoracoscopic lobectomy and sublobectomy for lung cancer and found that short-term outcomes after uniportal thoracoscopic surgery were similar to those after triportal thoracoscopic pulmonary resection (18). Furthermore, Chung et al. (19) reported that the perioperative outcomes of uniportal thoracoscopic lobectomy were similar to those of multiportal thoracoscopy, indicating that uniportal thoracoscopic lobectomy is a viable alternative to performing the procedure via multiple incisions. Yan et al. (20) conducted a review article (n=1,314) and demonstrated the similar or better short-term outcome between single-incision and multiport thoracoscopic surgery in the treatment of lung cancer. In the current study, we found that patients in the single-incision thoracoscopic group had a shorter length of hospital stay and less blood loss than those in the multiple-incision group, indicating that single-incision thoracoscopic surgery results in similar if not better perioperative outcomes.
The primary limitations of this study are its retrospective design and selection bias, although we tried to reduce the degree of bias by matching patients on propensity score. We began thoracoscopic surgery for lung cancer with three-port in 2005, a two-port technique in 2007 and a single-port approach in 2009. We didn’t perform different in the same period. We performed multiple-incision thoracoscope in the early period. Therefore, it contributed to uneven follow-up time between two groups. The mean follow-up differed markedly between the two groups because single-incision thoracoscopic surgery has been performed in our center for a shorter period of time than multiple-incision thoracoscopy. The difference in mean follow-up between the two groups, therefore, might have influenced the overall and disease-free survival results.
Conclusions
The long-term survival and disease-free survival rates achieved with single-incision thoracoscopic surgery are comparable to those achieved with multiple-incision thoracoscopic surgery for lung cancer.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by the institutional review board of the Koo Foundation Sun Yat-Sen Cancer Center (KFSYSCC-IRB No. 20150623A).
References
- Parkin DM, Bray F, Ferlay J, et al. Global cancer statistics 2002. CA Cancer J Clin 2005;55:74-108. [Crossref] [PubMed]
- Wang BY, Huang JY, Cheng CY, et al. Lung cancer and prognosis in taiwan: a population-based cancer registry. J Thorac Oncol 2013;8:1128-35. [Crossref] [PubMed]
- Wang BY, Huang JY, Lin CH, et al. Thoracoscopic Lobectomy Produces Long-Term Survival Similar to That with Open Lobectomy in Cases of Non-Small Cell Lung Carcinoma: A Propensity-Matched Analysis Using a Population-Based Cancer Registry. J Thorac Oncol 2016;11:1326-34. [Crossref] [PubMed]
- Onaitis MW, Petersen RP, Balderson SS, et al. Thoracoscopic lobectomy is a safe and versatile procedure: experience with 500 consecutive patients. Ann Surg 2006;244:420-5. [PubMed]
- Swanson SJ, Herndon JE 2nd, et al. Video-assisted thoracic surgery lobectomy: report of CALGB 39802--a prospective, multi-institution feasibility study. J Clin Oncol 2007;25:4993-7. [Crossref] [PubMed]
- Ohtsuka T, Nomori H, Horio H, et al. Is major pulmonary resection by video-assisted thoracic surgery an adequate procedure in clinical stage I lung cancer? Chest 2004;125:1742-6. [Crossref] [PubMed]
- Ceppa DP, Kosinski AS, Berry MF, et al. Thoracoscopic lobectomy has increasing benefit in patients with poor pulmonary function: a Society of Thoracic Surgeons Database analysis. Ann Surg 2012;256:487-93. [Crossref] [PubMed]
- Gonzalez D, de la Torre M, Paradela M, et al. Video-assisted thoracic surgery lobectomy: 3-year initial experience with 200 cases. Eur J Cardiothorac Surg 2011;40:e21-8. [Crossref] [PubMed]
- Gonzalez-Rivas D, Paradela M, Fernandez R, et al. Uniportal video-assisted thoracoscopic lobectomy: Two years of experience. Ann Thorac Surg 2013;95:426-32. [Crossref] [PubMed]
- Wang BY, Tu CC, Liu CY, et al. Single-incision thoracoscopic lobectomy and segmentectomy with radical lymph node dissection. Ann Thorac Surg 2013;96:977-82. [Crossref] [PubMed]
- Hsu PK, Lin WC, Chang YC, et al. Multiinstitutional analysis of single-port video-assisted thoracoscopic anatomical resection for primary lung cancer. Ann Thorac Surg 2015;99:1739-44. [Crossref] [PubMed]
- Ng CS, Kim HK, Wong RH, et al. Single-Port Video-Assisted Thoracoscopic Major Lung Resections: Experience with 150 Consecutive Cases. Thorac Cardiovasc Surg 2016;64:348-53. [Crossref] [PubMed]
- Xie D, Wang H, Fei K, et al. Single-port video-assisted thoracic surgery in 1063 cases: a single-institution experience. Eur J Cardiothorac Surg 2016;49 Suppl 1:i31-6. [Crossref] [PubMed]
- Gonzalez-Rivas D, Fieira E, Delgado M, et al. Is uniportal thoracoscopic surgery a feasible approach for advanced stages of non-small cell lung cancer? J Thorac Dis 2014;6:641-8. [PubMed]
- Drevet G, Ugalde Figueroa P. Uniportal video-assisted thoracoscopic surgery: safety, efficacy and learning curve during the first 250 cases in Quebec, Canada. Ann Cardiothorac Surg 2016;5:100-6. [Crossref] [PubMed]
- Wang BY, Liu CY, Hsu PK, et al. Single-incision versus multiple-incision thoracoscopic lobectomy and segmentectomy: A propensity-matched analysis. Ann Surg 2015;261:793-9. [Crossref] [PubMed]
- Liu CC, Shih CS, Pennarun N, et al. Transition from a multiport technique to a single-port technique for lung cancer surgery: is lymph node dissection inferior using the single-port technique? Eur J Cardiothorac Surg 2016;49 Suppl 1:i64-72. [PubMed]
- Mu JW, Gao SG, Xue Q, et al. A Matched Comparison Study of Uniportal Versus Triportal Thoracoscopic Lobectomy and Sublobectomy for Early-stage Nonsmall Cell Lung Cancer. Chin Med J (Engl) 2015;128:2731-5. [Crossref] [PubMed]
- Chung JH, Choi YS, Cho JH, et al. Uniportal video-assisted thoracoscopic lobectomy: an alternative to conventional thoracoscopic lobectomy in lung cancer surgery? Interact Cardiovasc Thorac Surg 2015;20:813-9. [Crossref] [PubMed]
- Yang Z, Shen Z, Zhou Q, et al. Single-incision versus multiport video-assisted thoracoscopic surgery in the treatment of lung cancer: a systematic review and meta-analysis. Acta Chir Belg 2017;21:1-9. [Crossref] [PubMed]
- Ettinger DS, Wood DE, Akerley W, et al. NCCN Guidelines Insights: Non-Small Cell Lung Cancer, Version 4.2016. J Natl Compr Canc Netw 2016;14:255-64. [Crossref] [PubMed]


