Thoracoscopic primary repair with mediastinal drainage is a viable option for patients with Boerhaave’s syndrome
Introduction
Spontaneous esophageal rupture was first reported by Boerhaave in 1724 (Boerhaave 1724) and typically occurs in conjunction with vomiting, retching, or swallowing a large food bolus. Any delay between perforation and treatment leads to severe mediastinitis and represents a life-threatening condition. Patient mortality following rupture is relatively high, especially with late diagnosis and treatment (1). Conventional surgical repair is recommended within 24 h of initial onset (2-5). However, some reports recommend surgery regardless of the time interval since onset (6-8). Thoracoscopic surgery has been introduced as a minimally invasive surgical approach for repairing a ruptured esophagus. Various surgical procedures, conservative to esophagectomy, have been utilized in clinical practice. These methods include open thoracotomy, thoracoscopic surgery, or the addition of laparotomy for reinforcement, using the greater omentum flap. However, a standard surgical procedure has not been established. Herein, we report our experience with thoracoscopic surgery and primary suture of the ruptured esophagus via a thoracoscopy or laparotomy.
Methods
Between November 2002 and May 2014, 11 patients with Boerhaave’s syndrome presented to our department and were treated with thoracoscopic mediastinal drainage. Another 2 patients were treated during the study period using other methods and were excluded from analysis: one was treated with mediastinal drainage via an open thoracotomy because of a tension pneumothorax following the introduction of general anesthesia, and the other was treated with thoracoscopic drainage and thoracoscopic esophagectomy because of a long tear in the esophagus. Medical records of the included patients were retrospectively reviewed. Six of the 11 patients were treated using a left thoracoscopic primary suture and mediastinal drainage (group A), and the remaining 5 patients were treated using a primary suture with omentopexy via a laparotomy followed by left thoracoscopic mediastinal drainage (group B). Patients in group A underwent surgery between 2002 and 2014, and patients in group B underwent surgery between 2009 and 2014. Seven surgical operators have contributed to their operations of group A or B in this period. The surgical procedure of group A or B was selected with consideration of each surgeon’s usual preference. All patients of the group B were also selected to undergo through laparoscopy instead of laparotomy with consideration of those surgeons’ usual preference. This choice was not influenced by radiological findings or clinical status. Preoperative signs, time interval between perforation and surgery, and treatment outcomes were compared between groups.
Perioperative treatment
Computed tomography (CT) scans were evaluated for the presence of pneumothorax, pneumomediastinum, or leakage into the pleural or mediastinum cavity. Contrast esophagography was performed in all patients, and confirmed extravasation to the outside of the esophagus. All patients initially received hydration and broad-spectrum antibiotics in the emergency room or intensive care unit (ICU), and were admitted to the ICU after surgery. Two patients in group B received enteral nutrition via tube jejunostomy, which was placed during the operation. Total parenteral nutrition was administered for all other patients until resumption of oral intake. Unless a leak was identified on contrast esophagography performed on postoperative day 7, the esophageal rupture was considered healed, and an oral diet was initiated.
Surgical procedure
Group A
General anesthesia was administered under differential lung ventilation, using a double-lumen endobronchial tube. The patient was placed in the standard right lateral decubitus position and was fixed in place using surgical beanbags. Six ports were inserted in the third and fifth intercostal space (ICS) on the anterior axillary line, fifth ICS on the middle axillary line, and fifth and seventh ICS on the posterior axillary line. The surgeon stood on the patient’s posterior side, with the assistant and camera operator on the patient’s anterior side. Monitors were placed on the patient’s anterior and posterior sides. Thus, the assistant and camera operator could see an upside-down image of the surgeon’s viewpoint. The mediastinal pleura were incised. Then, the widening thoracic cavity and mediastinum were irrigated and drained, followed by primary suture of the esophageal tear. Debridement and excision of necrotic and inflamed tissue was performed if necessary. Suturing of the tear consisted of an entire layer or a 2-layer suturing of the mucosal and muscularis layers with absorbable thread. Three drain catheters were inserted, with one catheter placed close to the esophageal suture line, and others placed along the anterior and dorsal chest wall.
Group B
The ruptured lower esophagus was sutured and re-enforced using an omentum patch placed via an open laparotomy performed with the patient in the supine position. Then, the patient was placed in the right decubitus position, and the thoracic cavity and mediastinum were irrigated with physiological saline. The number and placement of the inserted ports and operator’s standing position were the same as those for group A.
Statistics
All analyses were implemented using JMP Pro version 11 software (SPSS, Inc., Chicago, IL). Group comparisons were performed using the Mann-Whitney or chi-square test, followed by a two-tailed Fisher test for the variables of interest. Group comparisons were performed using repeated measures of analysis of variance (ANOVA).
Ethical statement
Given the retrospective nature of the non-intervention study, review by the local ethics committee of Tohoku University Hospital was not required.
Results
The study included 10 male and 1 female (in group B) patients, with an overall mean age of 59.9 years. Patient characteristics and the time interval between symptom onset and surgery are presented in Table 1. All parameters were similar between the two surgical groups. The range of time between symptom onset and surgery was 5–45 h for group A and 8–47 h for group B, with median times of 8.5 h and 10 h, respectively. In all patients, the perforation was located in the lower third of the esophagus. The mean length of the esophageal perforation was 3.1 cm in group A and 4.5 cm in group B. Although the mean duration of the surgery was shorter for group A (190.5 min) than for group B (249.4 min), this difference was not statistically significant.
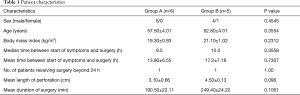
Full table
The general preoperative health conditions of the patients as reflected by hemoglobin, C-reactive protein, systolic blood pressure, heart rate, oxygen saturation, and body temperature, were also similar between the two surgical groups (Table 2). Although it trended upward on postoperative days 3 and 7 for group B, no significant group difference existed (Table 3).
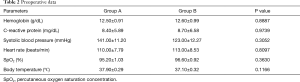
Full table
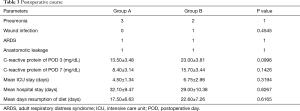
Full table
Postoperative complications occurred in 4 patients in group A and in 3 patients of group B. Suture line leakage occurred in one patient from each group and was successfully managed with a conservative approach, without any additional surgical intervention. One patient in group A with minor leakage revealed on contrast esophagography was managed using antibiotics and resumption of oral food was delayed an additional 7 days until the disappearance of the leakage along the suture line. Another patient of group B with thoracic abscess due to leakage along the suture line was well-managed by a thoracic tube placed during surgery, and his esophagothoracic fistula was closed by the postoperative day 34.
Pneumonia occurred in 50% of the patients of group A and in 40% of those in group B (Table 3). There were no significant differences in postoperative pulmonary complications, ICU stay, days until resumption of a solid diet, or the length of hospital stay (Table 3). There were no deaths in either group during the acute phase. One patient was transferred to another hospital for the purpose of rehabilitation; all other patients were discharged home. The mean hospital stay was 32 days (range, 17–75 days) and 29 days (range, 11–68 days) in groups A and B, respectively. At discharge, all patients were on a normal diet without dysphagia. No cases required re-surgery or re-hospitalization in relation to the treatment for this disease.
Discussion
Spontaneous esophageal rupture (Boerhaave’s syndrome) is a medical emergency that can cause life-threatening conditions including mediastinitis, pleuritis, and sepsis. Acute mortality has been reported in approximately 10% of cases when treated within 48 h, and in 25–40% of cases treated beyond 48 h (9). This disease requires early diagnosis and should be followed by prompt and appropriate management.
Boerhaave’s syndrome can be successfully treated using a conservative approach such as continuous thoracic drainage, administration of antibiotics, and hyperalimentation (9,10); however, non-surgical management requires longer hospital stays than successful surgical management (4,5,9). Furthermore, successful closure using endoscopic ligation with a snare loop, over-the-endoscope clips (11,12), or endoscopic sponge therapy (13) have been reported. However, only a small number of such reports exist, and larger studies are required to clarify the indications for these endoscopic treatments. Schweigert et al. (14) reported that endoscopic stent insertion offers no advantage in terms of morbidity, ICU, or length of hospital stay. Therefore, a non-operative approach should be considered in well-selected cases.
In the present study, a patient in group A who underwent surgery after 45 h experienced acute respiratory distress syndrome and was discharged on postoperative day 34. However, a patient of group B who underwent surgery after 48 h experienced an uneventful postoperative course and was discharged on postoperative day 15. Surgery within 24 h of onset is traditionally and generally recommended (2-5), since any delay in management promotes tissue necrosis and edema and prevents successful esophageal suturing (15). However, similar mortality rates have been reported for patients who underwent surgery in less than or greater than 24 h after onset (7,8,15). Furthermore, some reports of successful surgery after 48 h exist (6,8,16). Therefore, primary esophageal repair for patients with this disease should be considered as a surgical alternative based on operative findings and the patient’s status, rather than the time frame alone.
Widening the mediastinum for drainage and suturing the esophageal tear have generally been performed via thoracotomy. Recently, thoracoscopic drainage of the mediastinum or thoracic cavity while suturing the tear has been reported. Cho et al. (6) reported that for 7 patients treated with thoracoscopic surgery and 8 patients treated with open thoracotomy, mean hospital stay and frequency of postoperative complications did not differ between the two groups. The time required for thoracoscopic surgery is less than that for open surgery (6), and is expected to cause less surgical trauma and pain, thus likely prompting better postoperative recovery (17).
It has been reported that persistent leakage, if present, is significantly decreased by reinforcement using a gastric patch or fibrin glue (18). However, these patients underwent open thoracotomy, and there are few studies comparing the usefulness of reinforcement with thoracoscopic surgery. Yamashita et al. (19) reported the successful treatment of two patients for whom thoracoscopic suturing was used to close an esophageal tear and reinforce the greater omentum; however, an omental flap was formed via laparotomy followed by thoracoscopic irrigation of the thoracic and mediastinal cavity and required re-thoracotomy repaired with omentopexy. Performing omentopexy after the thoracoscopic procedure required an additional operative position to minimize the prolongation of the operation and anesthesia times. On the other hand, additional laparotomy might cause further surgical stress. We did not reinforce the suture of the esophagus in group A; however, reinforcement using fibrin glue may be favorable. This procedure is easy to perform and is considered to be noninvasive (18).
There are some published case reports that mention good results following laparotomy or laparoscopy with primary suture (20,21). Surgical procedures, such as those used in group B in the present study could be selected as an alternative given the good surgical outcomes we experienced following this procedure. As a hand operation might not reach the upper end of the tear when the upper end is high in the mediastinum or in the middle and upper esophagus, it is important to diagnose the preoperative localization of the lesion. Suitable operative procedures should be selected with consideration for the length and location of the tear.
Each surgical procedure has its own advantages. Therefore, both procedures should be considered as suitable when selecting the approach used to treat Boerhaave’s syndrome with regard to patient condition, status of the esophageal tear, and the surgeon’s skills. Limitation of this study includes that small series as the data is insufficient to make changes in clinical practice. However, in the present study, patient baseline characteristics and surgical outcomes were similar for both surgical procedures, with results no worse than that which has been previously published. Although further studies are required in the future to establish the indications and efficacy of this therapeutic approach, thoracoscopic esophageal repair, as well as suturing via laparotomy, is a good surgical alternative for patients with Boerhaave’s syndrome.
Acknowledgements
Funding: This work was partially supported by JSPS KAKENHI Grant Numbers JP15K1548.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Given the retrospective nature of the non-intervention study, review by the local ethics committee of Tohoku University Hospital was not required.
References
- Chang CH, Lin PJ, Chang JP, et al. One-stage operation for treatment after delayed diagnosis of thoracic esophageal perforation. Ann Thorac Surg 1992;53:617-20. [Crossref] [PubMed]
- Jones WG 2nd, Ginsberg RJ. Esophageal perforation: a continuing challenge. Ann Thorac Surg 1992;53:534-43. [Crossref] [PubMed]
- Okten I, Cangir AK, Ozdemir N, et al. Management of esophageal perforation. Surg Today 2001;31:36-9. [Crossref] [PubMed]
- Whyte RI. Boerhaave's syndrome. N Engl J Med 2001;344:139. [PubMed]
- Wright CD, Mathisen DJ, Wain JC, et al. Reinforced primary repair of thoracic esophageal perforation. Ann Thorac Surg 1995;60:245-8; discussion 8-9. [Crossref] [PubMed]
- Cho JS, Kim YD, Kim JW, et al. Thoracoscopic primary esophageal repair in patients with Boerhaave's syndrome. Ann Thorac Surg 2011;91:1552-5. [Crossref] [PubMed]
- Cho S, Jheon S, Ryu KM, et al. Primary esophageal repair in Boerhaave's syndrome. Dis Esophagus 2008;21:660-3. [Crossref] [PubMed]
- Jougon J, Mc Bride T, Delcambre F, et al. Primary esophageal repair for Boerhaave's syndrome whatever the free interval between perforation and treatment. Eur J Cardiothorac Surg 2004;25:475-9. [Crossref] [PubMed]
- Morimoto Y, Mukai T. Spontaneous esophageal rupture treated by conservative therapy. Jpn J Thorac Cardiovasc Surg 2000;48:473-5. [Crossref] [PubMed]
- Sato T, Obinata I, Takahashi S, et al. Spontaneous esophageal rupture successfully treated by conservative therapy: report of two cases. Surg Today 2002;32:421-5. [Crossref] [PubMed]
- Kuwabara J, Watanabe Y, Kojima Y, et al. Successful closure of spontaneous esophageal rupture (Boerhaave's syndrome) by endoscopic ligation with snare loops. SpringerPlus 2016;5:921. [Crossref] [PubMed]
- Van Weyenberg SJ, Stam FJ, Marsman W. Successful endoscopic closure of spontaneous esophageal rupture (Boerhaave syndrome). Gastrointest Endosc 2014;80:162. [Crossref] [PubMed]
- Scharl M, Stanek N, Kroger A, et al. Successful treatment of a proximal esophageal rupture with a luminal sponge. Endoscopy 2015;47 Suppl 1 UCTN:E293-4.
- Schweigert M, Beattie R, Solymosi N, et al. Endoscopic stent insertion versus primary operative management for spontaneous rupture of the esophagus (Boerhaave syndrome): an international study comparing the outcome. Am Surg 2013;79:634-40. [PubMed]
- Teh E, Edwards J, Duffy J, et al. Boerhaave's syndrome: a review of management and outcome. Interact Cardiovasc Thorac Surg 2007;6:640-3. [Crossref] [PubMed]
- Ando H, Shitara Y, Hagiwara K, et al. Successful surgical treatment of a spontaneous rupture of the esophagus diagnosed two days after onset. Case Rep Gastroenterol 2012;6:260-5. [Crossref] [PubMed]
- Haveman JW, Nieuwenhuijs VB, Kobold JP, et al. Adequate debridement and drainage of the mediastinum using open thoracotomy or video-assisted thoracoscopic surgery for Boerhaave's syndrome. Surg Endosc 2011;25:2492-7. [Crossref] [PubMed]
- Sulpice L, Dileon S, Rayar M, et al. Conservative surgical management of Boerhaave's syndrome: experience of two tertiary referral centers. Int J Surg 2013;11:64-7. [Crossref] [PubMed]
- Yamashita S, Takeno S, Moroga T, et al. Successful treatment of esophageal repair with omentum for the spontaneous rupture of the esophagus (Boerhaave's syndrome). Hepatogastroenterology 2012;59:745-6. [PubMed]
- Hueting WE, van Laarhoven CJ, Gooszen HG. Surgical treatment of distal esophageal perforation not due to a malignancy: results in 11 patients, University Medical Center Utrecht, 1994-1998. Ned Tijdschr Geneeskd 2000;144:1276-9. [PubMed]
- Landen S, El Nakadi I. Minimally invasive approach to Boerhaave's syndrome: a pilot study of three cases. Surg Endosc 2002;16:1354-7. [Crossref] [PubMed]


