Videoscope-assisted cardiac surgery
Introduction
Videoscope-assisted cardiac surgery (VACS) is a platform that provides less incisional trauma but equivalent corrective procedures for cardiac lesion as conventional open approach. It is one of the various techniques of minimally-invasive cardiac surgery (MICS) or minimal access cardiac surgery (MACS) (1) that minimizes incision size in order to lower morbidity while preserving quality in carefully selected patients (1). Compared with direct vision in minimal access approach (2), videoscope may offer smaller incisions, brighter illumination, larger images, and easier recording and broadcasting, whereas it may require more learning curve for eye-hand coordination. Three-dimensional imaging is only available in da Vinci robotic system and thoracoscope can only offer two-dimensional imaging that is a drawback relatively to direct vision. Almost all minimal access cardiac operations are videoscopic per se or have a videoscope-assisted version. Not repeating the abundant literature that covers the issues in MACS, such as comparisons to the conventional approach, peripheral cannulation, cardiopulmonary bypass management, etc., here we would like to focus on the issues that are more specific to videoscope-assisted approach, both robotic (3,4) or thoracoscopic (5), and on the procedures that we are more experienced in our own institution: endoscopic saphenous vein and radial artery harvesting (6-12), robotic left internal mammary artery (LIMA) takedown (13), robotic-assisted coronary artery bypass, mitral valve operations (4,5), aortic valve operations, and various intra-cardiac operations. We will cover the topics that include patient selection and preparation, technical concerns and issues, and some relevant issues. We welcome both novice or expert audience and this article may not be perfectly comprehensive but will serve as decent reminders and highlights.
General considerations
Ironically, intending to lower surgical morbidity, VACS is seldom performed in high-risk patients in which full sternotomy possesses insignificant risk comparing to surgical mortality. Only in elective low-risk but symptomatic patients, it matters more for smaller wounds with uncompromised surgical outcomes when the goal of surgery is to improve life quality. VACS approach further lowers surgical risks in properly selected low-risk patients. We estimate the surgical risk of our cardiac surgery patients by calculating EuroScore II (14,15). VACS patients usually fit low-risk profile that includes: elective, no comorbidity, and good myocardial contractility. With clinical feasibility, patient’s cosmetic needs and financial affordability will be next determinants of selecting VACS approach. Taiwanese have government-managed universal health insurance and patients have higher co-payments for VACS that costs higher than full sternotomy approach—robotic is even higher than thoracoscopic. Thus patient’s socio-economic status cannot be ignored for hospital cost management.
Once VACS approach has been chosen and agreed by the patient and the surgeon, we begin preoperative workup for risk assessment and surgical planning, for example, lung function test for selective one-lung ventilation, ankle-brachial index for peripheral artery integrity for peripheral femoral cannulation, chest non-contrast CT for aortic calcification and thoracic anatomy overview (16,17), confirmatory echocardiography for global contractility, regional wall motion, valve competency, and relevant structural and functional assessment, and other workups are like conventional cardiac operation such as carotid Doppler, laboratory panel, etc. If we identify excessive risk, either for VACS or overall cardiac procedure, we will re-evaluate the operation, or seek solutions accordingly.
As mentioned earlier, VACS is only a platform or vehicle to the target. Procedure-specific issues are paramount and the surgeon must have full expertise of the surgical planning and corrective procedure of the cardiac lesions as in conventional open approach. For example, is the coronary revascularization to be totally surgical or hybrid with cardiologists? What are the coronary bypass targets? What is the repair strategy for mitral annulus, leaflets, chordae, papillary muscle, and left ventricle? Also, perfusion strategy must be error-free for on-pump cases. For example, we must select the best cannulation method, cannulae sizes, venous drainage vacuum, etc. Furthermore, like conventional cardiac operations, hemostasis is also very important and it is often more difficult due to minimally invasive approach. Port bleeding is the most common reason for postoperative re-exploration in minimally invasive cardiac surgery.
Postoperative management is almost the same as the conventional cardiac surgery. One of the reminders is that the clinical alertness should not be lowered because of the minimal invasiveness or the relatively low-risk profile. Delayed management of cardiac tamponade in a routine MIDCAB can lead to mortality. Another issue specific to VACS is the pain control, especially in robotic cases or rib-spreading mini-thoracotomy cases. Chest wall pain will impair the pulmonary recovery and may cause pneumonia in fragile cases. Anesthesiologists must be involved in the pain-control issue and we may use controlled-release local infiltrative analgesics or intravenous patient-controlled analgesia.
Robotic videoscope- and thoracoscope-assisted operations
VACS included robotic and thoracoscopic approaches and each has special considerations. Because of the market dominance, here our discussion on robotic surgery is limited to the da Vinci HD Si system by Intuitive Surgical, Inc. only. Logistic issues are not addressed here but they are essential for the back-stage operating room management.
Robotic videoscope provides three-dimension vision for the console-side surgeon and the surgical cart has robotic arms and instruments that offer supreme dexterity duplicating surgeon’s hands movements. Patient-side surgeon or assistant is very important and must have expertise in both robotic and thoracoscopic techniques. There are lots of robotic arm maneuvers and troubleshooting, knot-pusher tying, suture management, and object hand-overs. We must also get used to the dependency of visual clues that replace tactile feedback that is absent in the current robotic system, preventing excessive forces to the objects in visual field. There are also some basic robotic rules to follow as emphasized in the boot camp training such as the avoidance of moving robotic instruments outside visual field, “sweet spot” issues, etc.
Thoracoscopic approach offers equivalent results without bigger incision wounds for experienced minimally invasive surgeons. Its weaknesses include non-stereo two-dimensional images and less dexterity comparing to robotic counterpart that can be overcome in the learning curves. Its strengths include lower costs, tactile feedbacks, and less dependency on the assistant, as compared with robotic approach; the operating surgeon works mostly at the patient-side like conventional operations. Just as general abdominal and thoracic surgeon’s laparoscopic and thoracoscopic operations, there are a variety of selections of scopes, instruments, and accessories, and the operating surgeon must pick the optimally customized specifications of each item. For example, in our institute we prefer Delacroix-Chevalier minimally invasive surgery instruments set and our custom-made thoracoscope is 17 cm long, 30-degree, and using 5-mm ports.
After the general considerations, let us move on to the procedure-specific parts that we will share our experience below.
Coronary artery revascularization
Endoscopic vein and radial artery harvesting
There have been debates for intima integrity and long-term graft patency for comparing endoscopic and open vein harvesting (7,9-12). With techniques and equipment advancement, endoscopic vein harvesting has become a mature method and most concerns are experience and learning curves. With well-trained and experienced staff, the conduit quality is comparable to open method and not compromised as reported evidences (7,12). Our experience has shown satisfactory and encouraging results for both saphenous vein (8) and radial artery (6). Endoscopic vein harvesting has been our default practice if the veins fit and the patient can afford. Technical aspects have been covered elsewhere (8,11) and one of the key points of success is the preoperative vein echo mapping for endoscopic feasibility. We mark the vein paths and take the open method if the vein diameters fall outside the range of 2 to 4 mm.
Robotic left internal mammary artery (LIMA) takedown
Robotic surgery is an advanced version of videoscopic approach. Robotic coronary revascularization, assisted or total, has been reviewed in previous literature (13,18-20) and robotic internal mammary artery takedown has been considered an essential step (21). Detailed technical issues, including set-ups, are available in “da Vinci® Beating Heart Coronary Revascularization Procedure Guide”, a booklet offered by the vendor. As the most expensive among all methods, robotic LIMA takedown may offer easier learning curves than other minimal invasive methods and it offers the possibility of taking bilateral internal mammary arteries (13). Surgeons can choose preferred instruments such as 30-degree camera, right permanent cautery spatula, and left black diamond micro forceps, and take either skeletonized or pedicle method. With proper patient positioning and optimally placed ports, the surgical views and maneuvers should be satisfactory without the blockade of left shoulder. Timely troubleshooting of the robotic arms by the patient-side assistant is very valuable for the success of the procedure (Figure 1).
Robotic-assisted CABG and TECAB (3,4)
With LIMA and/or RIMA and other conduits ready, the coronary revascularization can proceed in a variety of ways, such as off-pump, beating on-pump, arrest on-pump, hand anastomosis, or robotic anastomosis, depending on learning curves or surgeon’s expertise (3,19,20,22). We may have MIDCAB, robotic-assisted CABG, or TECAB (18). In the arrest on pump approach, the aortic occlusion method must be chosen (4). Coronary anastomosis may be done with prolene, U-clips, or anastomotic connectors such as C-Port Flex A (Cardica, Redwood, CA, USA), depending on availability and surgeon’s preference. In minimally invasive CABG, we cannot forget the role of hybrid CABG, a mode of teamwork that takes both strengths: surgeons bypass left anterior descending artery and cardiologists open left circumflex and right coronary arteries. During cardiologist’s coronary intervention, immediate postoperative angiography can be done for the revascularized coronary artery for quality assurance in addition to surgeon’s transit time flow probes.
Pitfall scenarios and suggestions
Here we show some near-miss scenarios and provide some proposed troubleshooting.
Shock develops upon inserting the first port trocar with core
The patient develops sudden decrease of systolic blood pressure from 110 to 70 mmHg right upon the insertion of the camera trocar port with core. Electrocardiogram shows sinus rhythm and pulse oximetry shows 99%. Instantly we inflate both lungs, release intra-thoracic CO2, and anesthesiologists begin fast fluid intravenous infusion. The blood pressure is still low. How to troubleshoot?
Trans-esophageal echocardiography (TEE)!
TEE finds pericardial effusion with cardiac tamponade! Our port trocar with core injures epicardium and leads to bleeding. Then we make a mini-thoracotomy just as big as we need for robotic-assisted CABG and use minimally invasive long instruments to drain the tamponade and fix the problem. The hemodynamics is restored. We proceed to place ports, dock, and finish the robotic-assisted CABG.
After LIMA takedown, ventricular fibrillation attacks during pericardial opening
With LIMA mobilized and ready, we proceed to open the pericardium with micro-forceps and cautery spatula. While we cauterize the pericardium, suddenly pulseless ventricular tachycardia attacks! Defibrillator shock paddles are non-sterilized and in the closet! Intra-thoracic defibrillator shock paddle are sterilized but too big to enter the mini-thoracotomy.
External defibrillator pads are already in place!
We immediately deliver defibrillator shocks and convert the rhythm back to sinus. Anesthesiologists begin some lidocaine and amiodarone intravenous infusion. Operation proceeds. For all MICS or VACS, we routinely place external defibrillator pads in case of pericardiotomy-related ventricular arrhythmia that mandates electric defibrillation. Unprepared access for electric defibrillator will lead to disaster.
Progressive hypotension and hypoxia develop during robotic LIMA takedown
During robotic LIMA takedown, anesthesiologists report dropping blood pressure to below 90 mmHg and O2 saturation down to 90%. From the videoscope, there is no bleeding and the left lung is not injured.
CO2 over-inflation!
Robotic LIMA takedown needs artificial left pneumothorax with CO2. The patient has to tolerate both one-lung ventilation and left CO2 pneumothorax. In fragile patients, hypoxia may develop from inadequate lung reserve by one-lung ventilation and have hypotension from relative “tension pneumothorax”. The optimal CO2 intra-thoracic pressure may vary in different settings. We may adjust or lower the CO2 pressure first if hypotension develops. We may also take a break and anesthesiologists can ventilate both lungs for a while. The ventilation and pressure must be addressed for robotic LIMA takedown.
Mitral valve operations
Mitral valve procedures are among the most successful applications of VACS. There have been plenty of recent literature on the topic, either robotic (4,23-27) or thoracoscopic (28-31), and there are coverage of perfusion and cardioplegia strategy (30,32-34) and anesthesia planning (35). There are also discussions on detailed technical issues (36-40). Readers are encouraged to seek these excellent references.
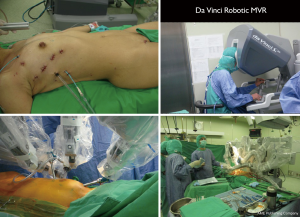
In our institute, our mitral VACS has been performed since 2000, with the methodology has been described elsewhere (28,33). In brief, either robotic (Figure 2) or thoracoscopic (Figure 3), we use double-lumen endotracheal tube, right-up 30-degree supine, right mini-thoracotomy, femoral arterial and venous cannulation, transcutaneous pigtail adenosine-induction cardioplegia delivery (41), HTK cardioplegia as default for robotic (34) or difficult cases, Chitwood transthoracic aortic cross-clamps, and Delacroix-Chevalier tripod atrial lifter with CO2 insufflation line.
We could like to do mitral VACS in the most cost-effective way. For example, we prefer Chitwood transthoracic aortic cross-clamps to endo-aortic balloon, in addition to cost advantage, clinical benefits have been demonstrated in a recent comparative study (42). There have been encouraging reports of mitral VACS without aortic occlusion (43,44) that we may try in selected cases, but we still believe trans-thoracic aortic cross-clamps with myocardial arrest provide optimal myocardial protection and operative exposure. With experience and confidence obtained over the years, we also perform mitral VACS in less ideal patients and get satisfactory results, like the recent reports for the patients with left ventricle dysfunction (45,46).
Since the rules and methods of mitral valve repair per se are similar in either conventional sternotomy or VACS approaches (36-39,47), and the readers must also have expertise in the techniques, we would not repeat them in the following discussions that are just highlighted reminders and we welcome further incoming comments and advices.
Pitfall scenarios and suggestions (5,28)
During peripheral cannulation, the cannula fails to advance properly
Using Seldinger technique, with our guidewire in place and we have dilated the vascular entry, we insert the cannula through the wire, but it gets stuck and cannot advance.
Peripheral cannulation with proper pump perfusion is vital in mitral VACS. We may take open or puncture techniques for artery and vein cannulation and each has strengths and weaknesses. Sometimes we use graft interposition for femoral artery. Whenever we get access into femoral vein and upward, we always ask anesthesiologist to check the TEE for the guidewire position that is best in right atrium. With the properly placed guidewire, the cannula should advance smoothly. If not, we have several solutions. First, we may downsize the cannula. Second, we can repeat dilators to open fascia entry further. Third, we can use alternative cannulation site while ask the assistant to compress the failed puncture site firmly.
With arterial and venous cannulae ready, the heart-lung machine pump cannot achieve adequate flows
With artery and vein cannulae placed properly at groin, we find the cannulae fluctuate vigorously and the pump flow cannot reach 3 liters/min and up, with adequate intravascular volume.
Our solutions to the scenario include the following. We can add venous drain vacuum (Maquet VAVD Controller, Maquet Cardiopulmonary AG, Hirrlingen, Germany) to facilitate venous drainage. Also, we can place an additional venous cannula at right internal jugular vein. Of course, cannula sizes must be big enough to fit patient’s body size.
Persistent or progressive acidosis develops during cardiopulmonary bypass
During the mitral VACS, everything seems going smoothly. But our perfusionists have found metabolic acidosis persists even with the correction by using sodium bicarbonate.
This scenario should be reported to the operating surgeon as early as possible and timely troubleshooting is mandatory. It may reflect sub-optimal perfusion, either cannula-related or unrelated. Is mean arterial pressure optimal? Is there any unexpected blood loss? Is there any distension for peritoneal or retroperitoneal space? Are all cannulae in good positions? Is there any kinking of venous cannula inside right atrium or vena cava that is retracted up for left atrial opening? We may ask the anesthesiologists to use TEE to explore additional clues. We must keep alert and continue troubleshooting until the event improves or is resolved.
After mitral valve repaired and atrial lifter released, left atrial opening is too floppy to close
Following a saline jet test, the repaired mitral valve is competent and we proceed to remove the left atrial lifter and close the left atriotomy. However, from the camera, left atrial wall dropped and we cannot see the edges for placing stitches. The assistant also sees the problem on the screen, but it is almost impossible to place an additional grasper to help.
Like conventional open operations, good retractions are vital for VACS exposure. Left atrial midway traction suture can solve the problem. We can place a traction suture in left mid-atrium and pull it out through the entry of left atrial lifter when opening left atrium. At the end of mitral valve repair, we remove the left atrial lifter but the traction suture still holds the left atrium open. Without any additional assistance, we can close the left atriotomy with ease.
Pigtail catheter for cardioplegia and deairing (33)
Even with limited exposure and controlled budgets, we have found cardioplegia delivery and deairing venting can be done easily with our inexpensive 8-French pigtail catheter. The technical details are covered elsewhere (41). In brief, it is placed transcutaneously under videoscope vision into ascending aorta, can be used to deliver cardioplegia solution and remove air bubbles during deairing, and is removed after pledgetted purse-string suture. When it is removed from aorta, we keep it inside thorax as an additional pericardial drain. Along with adenosine induction and Chitwood trans-thoracic aortic-cross clamps, we can achieve rapid myocardial arrest optimal myocardial protection without expensive endovascular devices for aortic occlusion or cardioplegia delivery.
Aortic valve operations and other intra-cardiac operations
In contrary to mitral valve procedures, aortic valve procedures often can be done with minimally invasive non-endoscopic approach (48-51). We have done it with endoscope by anterior/axillary approach whereas mostly we do aortic valve replacement under direct vision via right mini-parasternotomy approach. We may share our endoscopic methodology in the future.
With our VACS platform, we can perform other intra-cardiac operations, such as Ebstein’s anomaly, tricuspid valve operations, ventricular septal defect, atrial septal defect, Maze procedures, and many combined cardiac operations, of course, in optimized and selected patients. One of the reminders is the bi-caval cannulation and occlusion for opening right atrium. With femoral vein cannula in inferior vena cava, right internal jugular vein cannula can be placed in superior vena cava, and both cavae must be clamped when right atrium is opened.
Specific complications
When VACS meets most outcomes and quality measurements such as mortality, coronary patency, valve competency, major morbidity such as stroke, ventilator dependency, bleeding re-exploration, sepsis, pneumonia, shock, cardiac events, hemolysis, etc., we address some minor complications specific to VACS here: groin morbidity, and compressive neuropathy.
Groin complications are related to peripheral cannulation and include seroma, and wound infection. They may prolong the hospital stay and affect patient’s satisfaction. There are several solutions. We should expose the femoral vessels with appropriate dissections, not too much. We may place artery and vein cannulae at different sides to minimize the local exploration. Proper hemostasis and decannulation techniques that avoid excessive bleeding also reduce the incidence. Adequate local wound compression or good surgical closure is also essential for prevention. But once a seroma or infection develops, it is advisable to do surgical debridement and place a drain, as well as local compression.
Compressive neuropathy is related to inadequate cushions or improper pressure in patient positioning, such as brachial plexus injury, ulnar nerve palsy, peroneal nerve palsy. Care must be taken during patient positioning for VACS. Excessive pressure to any body parts should be avoided, or adequate cushion must be applied. Its prevention is easier than treatment. Once it develops, neurological clinic and rehabilitation may help but the recovery process is lengthy. Sometimes, walking or forearm movement is impaired and the life quality is considerably affected.
Conclusions
With appropriate hardware set-up, staff training, patient selection, and surgeon’s learning curves, VACS can be performed for most cardiac procedures, such coronary artery bypass and mitral valve repair. Teamwork approach, including anesthesiologist, is essential for timely troubleshooting. We must ensure safety and provide quality to the target patients of VACS.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Raja SG, Amrani M. Past, present, and future of minimal access cardiac surgery. J Thorac Dis 2013;5:S629. [PubMed]
- Ward AF, Grossi EA, Galloway AC. Minimally invasive mitral surgery through right mini-thoracotomy under direct vision. J Thorac Dis 2013;5:S673-9. [PubMed]
- Canale LS, Mick S, Mihaljevic T, et al. Robotically assisted totally endoscopic coronary artery bypass surgery. J Thorac Dis 2013;5:S641-9. [PubMed]
- Mandal K, Alwair H, Nifong WL, et al. Robotically assisted minimally invasive mitral valve surgery. J Thorac Dis 2013;5:S694-703. [PubMed]
- Chirichilli I, D’Ascoli R, Rose D, et al. Port access (Thru-Port System) video-assisted mitral valve surgery. J Thorac Dis 2013;5:S680-5. [PubMed]
- Chiu KM, Li SJ, Chen JS, et al. Endoscopic radial artery harvest for coronary artery bypass surgery. J Formos Med Assoc 2006;105:384-9. [PubMed]
- Bakaeen FG. Endoscopic vein harvest for coronary artery bypass grafting is safe. J Surg Res 2013;185:522-3. [PubMed]
- Chiu KM, Chen CL, Chu SH, et al. Endoscopic harvest of saphenous vein: a lesson learned from 1,348 cases. Surg Endosc 2008;22:183-7. [PubMed]
- Deppe AC, Liakopoulos OJ, Choi YH, et al. Endoscopic vein harvesting for coronary artery bypass grafting: a systematic review with meta-analysis of 27,789 patients. J Surg Res 2013;180:114-24. [PubMed]
- Lopes RD, Hafley GE, Allen KB, et al. Endoscopic versus open vein-graft harvesting in coronary-artery bypass surgery. N Engl J Med 2009;361:235-44. [PubMed]
- Raja SG, Sarang Z. Endoscopic vein harvesting: technique, outcomes, concerns & controversies. J Thorac Dis 2013;5:S630-7. [PubMed]
- Sastry P, Rivinius R, Harvey R, et al. The influence of endoscopic vein harvesting on outcomes after coronary bypass grafting: a meta-analysis of 267,525 patients. Eur J Cardiothorac Surg 2013;44:980-9. [PubMed]
- Itagaki S, Reddy RC. Options for left internal mammary harvest in minimal access coronary surgery. J Thorac Dis 2013;5:S638-40. [PubMed]
- Nashef SA, Roques F, Sharples LD, et al. EuroSCORE II. Eur J Cardiothorac Surg 2012;41:734-44; discussion 744-5. [PubMed]
- Takkenberg JJ, Kappetein AP, Steyerberg EW. The role of EuroSCORE II in 21st century cardiac surgery practice. Eur J Cardiothorac Surg 2013;43:32-3. [PubMed]
- Manenti A, Giuliani E, Colasanto D. Computed tomography AIDS minimally invasive mitral valve surgery. Tex Heart Inst J 2013;40:211. [PubMed]
- Moodley S, Schoenhagen P, Gillinov AM, et al. Preoperative multidetector computed tomograpy angiography for planning of minimally invasive robotic mitral valve surgery: impact on decision making. J Thorac Cardiovasc Surg 2013;146:262-8.e1.
- Gao C, Yang M, Wu Y, et al. Early and midterm results of totally endoscopic coronary artery bypass grafting on the beating heart. J Thorac Cardiovasc Surg 2011;142:843-9. [PubMed]
- Srivastava S, Barrera R, Quismundo S. One hundred sixty-four consecutive beating heart totally endoscopic coronary artery bypass cases without intraoperative conversion. Ann Thorac Surg 2012;94:1463-8. [PubMed]
- Srivastava S, Gadasalli S, Agusala M, et al. Beating heart totally endoscopic coronary artery bypass. Ann Thorac Surg 2010;89:1873-9; discussion 1879-80.
- Oehlinger A, Bonaros N, Schachner T, et al. Robotic endoscopic left internal mammary artery harvesting: what have we learned after 100 cases? Ann Thorac Surg 2007;83:1030-4. [PubMed]
- Bonatti J, Schachner T, Bernecker O, et al. Robotic totally endoscopic coronary artery bypass: program development and learning curve issues. J Thorac Cardiovasc Surg 2004;127:504-10. [PubMed]
- Murphy DA, Miller JS, Langford DA. Endoscopic robotic mitral valve surgery. J Thorac Cardiovasc Surg 2007;133:1119-20; author reply 1120. [PubMed]
- Chitwood WR Jr, Rodriguez E, Chu MW, et al. Robotic mitral valve repairs in 300 patients: a single-center experience. J Thorac Cardiovasc Surg 2008;136:436-41. [PubMed]
- Nifong LW, Rodriguez E, Chitwood WR Jr. 540 consecutive robotic mitral valve repairs including concomitant atrial fibrillation cryoablation. Ann Thorac Surg 2012;94:38-42; discussion 43. [PubMed]
- Suri RM, Burkhart HM. Robotic mitral valve repair using polytetrafluoroethylene neochordal resuspension: the “known and unknown knowns”. Ann Thorac Surg 2013;96:1914-5. [PubMed]
- Ramzy D, Trento A, Cheng W, et al. Three hundred robotic-assisted mitral valve repairs: the Cedars-Sinai experience. J Thorac Cardiovasc Surg 2014;147:228-35. [PubMed]
- Chiu KM, Lin TY, Li SJ, et al. Less invasive mitral valve surgery via right minithoracotomy. J Formos Med Assoc 2006;105:715-21. [PubMed]
- Suri RM, Schaff HV, Meyer SR, et al. Thoracoscopic versus open mitral valve repair: a propensity score analysis of early outcomes. Ann Thorac Surg 2009;88:1185-90. [PubMed]
- Grossi EA, Loulmet DF, Schwartz CF, et al. Evolution of operative techniques and perfusion strategies for minimally invasive mitral valve repair. J Thorac Cardiovasc Surg 2012;143:S68-70. [PubMed]
- McClure RS, Athanasopoulos LV, McGurk S, et al. One thousand minimally invasive mitral valve operations: early outcomes, late outcomes, and echocardiographic follow-up. J Thorac Cardiovasc Surg 2013;145:1199-206. [PubMed]
- Murzi M, Cerillo AG, Miceli A, et al. Antegrade and retrograde arterial perfusion strategy in minimally invasive mitral-valve surgery: a propensity score analysis on 1280 patients. Eur J Cardiothorac Surg 2013;43:e167-72. [PubMed]
- Chiu KM, Chen RJ, Lin TY, et al. Cardioplegia delivery by transcutaneous pigtail catheter in minimally invasive mitral valve operations. Ann Thorac Surg 2013;95:e77-8. [PubMed]
- Patel N, DeLaney E, Turi G, et al. Custodiol HTK cardioplegia use in robotic mitral valve. J Extra Corpor Technol 2013;45:139-42. [PubMed]
- Rodrigues ES, Lynch JJ, Suri RM, et al. Robotic mitral valve repair: a review of anesthetic management of the first 200 patients. J Cardiothorac Vasc Anesth 2013; [PubMed]
- Chu MW, Gersch KA, Rodriguez E, et al. Robotic “haircut” mitral valve repair: posterior leaflet-plasty. Ann Thorac Surg 2008;85:1460-2. [PubMed]
- Smith JM, Stein H. Endoscopic placement of multiple artificial chordae with robotic assistance and nitinol clip fixation. J Thorac Cardiovasc Surg 2008;135:610-4. [PubMed]
- Suri RM, Burkhart HM, Rehfeldt KH, et al. Robotic mitral valve repair for all categories of leaflet prolapse: improving patient appeal and advancing standard of care. Mayo Clin Proc 2011;86:838-44. [PubMed]
- Suri RM, Burkhart HM, Daly RC, et al. Robotic mitral valve repair for all prolapse subsets using techniques identical to open valvuloplasty: establishing the benchmark against which percutaneous interventions should be judged. J Thorac Cardiovasc Surg 2011;142:970-9. [PubMed]
- Yozu R, Okamoto K, Kudo M, et al. New innovative instruments facilitate both direct-vision and endoscopic-assisted mini-mitral valve surgery. J Thorac Cardiovasc Surg 2012;143:S82-5. [PubMed]
- Chiu KM, Chen RJ, Lin TY, et al. Cardioplegia delivery by transcutaneous pigtail catheter in minimally invasive mitral valve operations. Ann Thorac Surg 2013;95:e77-8. [PubMed]
- Mazine A, Pellerin M, Lebon JS, et al. Minimally invasive mitral valve surgery: influence of aortic clamping technique on early outcomes. Ann Thorac Surg 2013;96:2116-22. [PubMed]
- Umakanthan R, Leacche M, Petracek MR, et al. Safety of minimally invasive mitral valve surgery without aortic cross-clamp. Ann Thorac Surg 2008;85:1544-9; discussion 1549-50. [PubMed]
- Petracek MR. Minimally invasive mitral valve surgery without aortic cross-clamping. Tex Heart Inst J 2011;38:701-2. [PubMed]
- Atluri P, Woo YJ, Goldstone AB, et al. Minimally invasive mitral valve surgery can be performed with optimal outcomes in the presence of left ventricular dysfunction. Ann Thorac Surg 2013;96:1596-601; discussion 1601-2. [PubMed]
- Santana O, Reyna J, Pineda AM, et al. Outcomes of minimally invasive mitral valve surgery in patients with an ejection fraction of 35% or less. Innovations (Phila) 2013;8:1-5. [PubMed]
- Falk V, Autschbach R, Krakor R, et al. Computer-enhanced mitral valve surgery: toward a total endoscopic procedure. Semin Thorac Cardiovasc Surg 1999;11:244-9. [PubMed]
- Raja SG, Benedetto U, Amrani M. Aortic valve replacement through J-shaped partial upper sternotomy. J Thorac Dis 2013;5:S662-8. [PubMed]
- Fenton JR, Doty JR. Minimally invasive aortic valve replacement surgery through lower half sternotomy. J Thorac Dis 2013;5:S658-61. [PubMed]
- Raja SG, Benedetto U. Minimal access aortic valve replacement via limited skin incision and complete median sternotomy. J Thorac Dis 2013;5:S654-7. [PubMed]
- Luciani GB, Lucchese G. Minimal-access median sternotomy for aortic valve replacement. J Thorac Dis 2013;5:S650-3. [PubMed]