Pregabalin as an analgesic option for patients undergoing thoracotomy: cost analysis of pregabalin versus epidural analgesia for post-thoracotomy pain relief
Introduction
Thoracic epidural analgesia is one of the most effective methods for controlling post-thoracotomy pain (1) and is common in major thoracic surgery. Epidural analgesia provides superior pain relief compared with systemic opioids (2,3), allows for early extubation, and may partially restore respiratory function (4). However, epidural analgesia may not always be ideal because of associated adverse effects such as hypotension, urinary retention, pulmonary complications, and nausea (5), and because of procedural complications including nerve injury, epidural haematoma, and epidural abscess (6). Another concern is that epidural analgesia is not suitable for patients receiving anticoagulants because of the risk of bleeding complication (7). This makes it difficult to use epidural analgesia in older patients because these patients are more likely to receive anticoagulant therapy for comorbid cardiac complications. In older patients, the risks of traumatic puncture may also increase because of the reduced epidural space or spinal degeneration (7). For these patients, a simpler and safer analgesic option is required.
One possible alternative analgesic option to manage postoperative pain is pregabalin. Pregabalin binds to the alpha-2-delta subunit of voltage-gated calcium channels at presynaptic nerve terminals and thereby suppresses the release of neurotransmitters, resulting in anticonvulsant, analgesic, and anxiolytic effects (8-10). Pregabalin is indicated for the management of neuropathic pain, and is used for various neuropathic pain conditions (11). Perioperative administration of pregabalin is also effective for postoperative pain (12-14); therefore, we expected that pregabalin could be used as a replacement for epidural analgesia for post-thoracotomy pain relief.
We previously conducted a randomized controlled trial (RCT) to evaluate the effects of pregabalin on acute post-thoracotomy pain (15). Results showed that patients receiving pregabalin reported significantly lower pain and sleep interference rates at 1, 3, and 5 postoperative days than did those receiving epidural analgesia. Adverse effects in the pregabalin group included dry mouth, dizziness, constipation, nausea, and headache; however, the number of each adverse event was similar or significantly lower compared with the epidural analgesia group. The efficacy and safety of pregabalin suggested that it could be a useful analgesic option for patients undergoing thoracotomy.
Because pregabalin is an oral medication, pain control with pregabalin would be simpler than with epidural analgesia. Using pregabalin may therefore contribute to lower medical costs. However, no economic benefit would be gained if, for example, more analgesic medications were needed perioperatively to compensate for the absence of epidural analgesia. To our knowledge, no studies have evaluated the economic aspects of pregabalin compared with the commonly used epidural analgesia. Therefore, we assessed the economic impact of pregabalin in patients undergoing thoracotomy in this analysis, comparing thoracotomy-related medical costs between patients receiving pregabalin vs. those receiving epidural analgesia for post-thoracotomy pain relief, using data from our previous RCT.
Methods
We retrospectively collected and compared medical cost data for patients undergoing thoracotomy and receiving either pregabalin or epidural analgesia in our previous RCT. This analysis was approved by the Institutional Ethics Review Board at Teikyo University (Teikyo University Review Board 15-158-2).
Study population
The methods of the RCT were published previously (15). Briefly, we recruited patients aged 20–80 years undergoing thoracotomy at Teikyo University Hospital if they met all the inclusion criteria and none of the exclusion criteria. Patients who received antidepressants, anticonvulsants, or opioids prior to surgery, and patients who had difficulty receiving epidural analgesia (e.g., patients with spinal deformity and patients treated with anticoagulants) were excluded.
Eligible patients who provided written informed consent were randomly allocated either to the epidural analgesia group or the pregabalin group. The epidural analgesia group received 0.3% ropivacaine hydrochloride intraoperatively. For post-thoracotomy analgesia, this group received 0.2% ropivacaine hydrochloride and 1 µg/mL fentanyl via patient-controlled analgesia, beginning postoperatively and continuing for 48 hours. The pregabalin group received 75 mg pregabalin twice daily from the date of surgery until the 5th postoperative day. In both groups, patients received 200 mg celecoxib from the date of surgery until the 5th postoperative day, in addition to the study treatment. Surgery was performed under general anesthesia with intraoperative anesthetics administered based on the anesthesiologist’s discretion. For surgical techniques, a muscle-sparing thoracotomy is commonly performed using a chest retractor at the 4th or 5th intercostal space where an 8- to 10-cm skin incision is made from the lateral to the anterior axillary line to preserve the latissimus dorsi and the serratus anterior muscles. Patients were followed until the 5th postoperative day. Of 96 randomized patients, 6 patients were excluded for the following reasons: protocol violation (1 patient), consent withdrawal (1 patient), change in the surgical procedures (2 patients), and cancelled surgery (2 patients). The remaining 90 patients’ data (45 patients in each group) were analyzed in the RCT. In this analysis, we next compared the medical costs for all 90 patients, for whom data regarding patients’ backgrounds (e.g., diagnosis, type of resection, and location of incision) were drawn from the RCT data.
Japanese healthcare payment system
In Japan, medical fees for acute in-patient care are reimbursed based on a combination of two systems: the diagnosis procedure combination/per-diem payment system (DPC/PDPS) and a fee-for-service payment system (16).
Costs calculated based on the DPC/PDPS include fees for basic hospitalization, laboratory tests, diagnostic imaging, medication, injections, and treatment/procedures priced under 10,000 Japanese Yen (¥). For these items, a flat-rate per-diem fee has been defined for each DPC group, into which patients are classified according to diagnosis, procedure, and comorbidity/complication. Costs are calculated by multiplying the per-diem fee by the length of hospital stay and the hospital coefficient. The per-diem fees for each DPC group are set for three stages based on the length of hospital stay, with the higher per-diem fee associated with the earlier stage (17). For items not covered by the DPC/PDPS, costs are calculated on a fee-for-service basis according to national health insurance (NHI) points. These items include medical management, surgery, anesthesia, radiation therapy, and treatment/procedures ≥¥10,000 (17).
Cost data collection
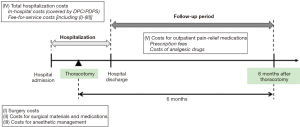
We evaluated the following five costs: (I) surgery costs; (II) costs for surgical materials and medications; (III) costs for anesthetic management; (IV) total hospitalization costs; and (V) costs for outpatient pain-relief medications (Figure 1).
Costs were collected from the health insurance claims data, which were calculated using the reimbursement prices based on the NHI points (1 point = ¥10). Costs were expressed in ¥ (1 US$ = ¥102 as of June 30, 2016).
Cost definitions
Surgery costs include the technical fees for the surgery, which vary according to the surgical procedure. These costs do not include technical fees for epidural analgesia performed intraoperatively.
Costs for surgical materials and medications include the costs of the surgical items used for thoracotomy. Costs of catheters, needles, and local anesthetic agents used for epidural analgesia are included in this category.
Costs for anesthetic management are the technical fees for anesthesia. Anesthetic management fees are calculated based on the duration of the general anesthesia and epidural analgesia (if applicable). A base cost is applied for the first two hours of general anesthesia, and an extra charge is added for every 30 minutes thereafter. When epidural analgesia is used in addition to general anesthesia, costs for the concurrent use of epidural analgesia, which also comprises the base cost and extra charges per 30 minutes after the first two hours, are added.
Total hospitalization costs correspond to the total amount of a claim. These costs comprise the in-hospital costs calculated based on the DPC/PDPS and costs calculated on a fee-for-service basis, which include surgery costs, costs for surgical materials and medications, and costs for anesthetic management.
Costs for outpatient pain-relief medications are the sum of the prescription fees and the costs of analgesic drugs from hospital discharge to 6-months after thoracotomy. Analgesic drugs include pregabalin, celecoxib, diclofenac sodium, loxoprofen sodium hydrate, acetaminophen, and tramadol hydrochloride/acetaminophen. Prescription fees are calculated by multiplying the unit price for the prescription by the total number of prescriptions of analgesic drugs during the study timeline. Costs of analgesic drugs are calculated by multiplying the NHI drug price (Table 1) by the total number of prescribed doses during the study timeline. Data for the total number of prescriptions and the total number of prescribed doses of each drug were collected from electronic medical records. Outpatient prescription was performed in the same manner as ordinary care, in which the selection of analgesic drugs was left to the physician’s judgement rather than according to a specific treatment policy or criterion for drug selection. Analgesic drugs were prescribed solely to treat post-thoracotomy pain. No patient had received these drugs for other treatment purposes (e.g., treatment of low back pain) before the surgery. The 6-month follow-up was chosen as longer than a 2-month postoperative period to include medication costs for possible chronic post-thoracotomy pain, which is defined as pain that persists for at least 2 months following thoracotomy (18).

Full table
We did not evaluate the costs of treating comorbidities in this analysis because we considered that these costs would not reflect differences related to the analgesic techniques. Also, because this analysis used a randomized sample from a RCT, patients should have been randomly divided into two groups regarding the distribution of comorbidities. Therefore, in theory, costs to treat comorbidities can be considered equal in the two groups. We also did not compare costs to treat pain and adverse events during hospitalization because these costs were comprehensively calculated based on the DPC/PDPS and were included in the total hospitalization costs. Exact costs for specific treatments could not be extracted from claims data; therefore, we decided to focus on outpatient prescriptions to evaluate costs for pain treatment.
Statistical analysis
Summary statistics were used to describe patients’ characteristics and costs for each group. Continuous variables were expressed as medians and ranges, and categorical variables as numbers and percentages. We compared costs between the groups using the Wilcoxon rank sum test. The proportions of six types of analgesic drugs prescribed from hospital discharge to 6-months after thoracotomy were calculated for each group. The number of doses for each drug prescribed during the study timeline were also calculated for each group. All statistical analyses were performed using SAS Release 9.4 (SAS Institute, Inc., Cary, NC, USA). The significance level was set at 0.05 on both sides.
Results
We analyzed data for all 90 patients, and patients’ characteristics in each group are summarized in Table 2. The median [range] age was 69 [31–77] years in the epidural analgesia group and 71 [35–80] years in the pregabalin group. In both groups, the majority of patients were diagnosed as having primary lung cancer. The median total length of hospital stay was 10 days in the epidural analgesia group and 11 days in the pregabalin group; however, one patient in the pregabalin group was hospitalized for an extraordinarily long period (64 days). No remarkable differences were observed between the groups for age, sex, diagnosis, type of resection, location of incision, and length of hospital stay.

Full table
Costs during hospitalization
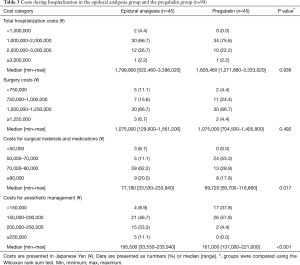
Costs incurred during hospitalization are shown in Table 3. Total hospitalization costs were similar between the two groups; for the majority of patients (66.7% in the epidural analgesia group and 75.6% in the pregabalin group), costs ranged from ¥1,000,000 to less than ¥2,000,000. Surgery costs were also similar between the groups; for the majority of patients (66.7% in both groups), costs ranged from ¥1,000,000 to less than ¥1,250,000. However, median costs for surgical materials and medications were significantly lower in the pregabalin group than in the epidural analgesia group by approximately ¥7,000 (Wilcoxon rank sum test, P=0.017). Median costs for anesthetic management were also lower in the pregabalin group than in the epidural analgesia group by ¥34,500, with significant differences between the groups (the Wilcoxon rank sum test, P<0.001).
Full table
Costs from hospital discharge to 6-months after thoracotomy
Table 4 contains a summary of the costs for outpatient pain-relief medications. Of the 90 patients, 10 patients were excluded because of loss to follow-up: 4 patients who were treated at different hospitals, 5 patients who were hospitalized for chemotherapy, and 1 patient who underwent another surgery after thoracotomy. Data from the remaining 80 patients, 40 patients for each group, were analyzed to determine outpatient costs.
Full table
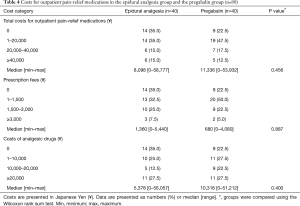
Total costs for outpatient pain-relief medications were similar between the groups; in both groups, the total costs were less than ¥20,000 for 70% of patients. Fourteen patients in the epidural analgesia group and nine patients in the pregabalin group incurred no outpatient costs. The median costs of analgesic drugs were lower in the epidural analgesia group than in the pregabalin group, but differences did not reach statistical significance (¥5,378 and ¥10,316, respectively; Wilcoxon rank sum test, P=0.400). In contrast, median prescription fees in the pregabalin group were half those in the epidural analgesia group, but the differences were not statistically significant (Wilcoxon rank sum test, P=0.887).
Outpatient pain-relief medications
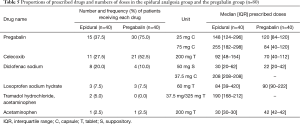
The proportions of analgesic drugs and the median doses of each drug prescribed in each group are summarized in Table 5. Among the listed six types of analgesic drugs, pregabalin made up the largest share in both groups: 37.5% (15 patients) in the epidural analgesia group and 75.0% (30 patients) in the pregabalin group. Celecoxib was the second most commonly used drug in both groups (27.5% in the epidural analgesia group and 52.5% in the pregabalin group).
Full table
Although the number of patients receiving pregabalin and/or celecoxib was higher in the pregabalin group, the median doses of pregabalin and celecoxib were both lower in the pregabalin group. For pregabalin 25 mg capsules and celecoxib 200 mg tablets, the median doses were more than 20 doses less in the pregabalin group. The epidural analgesia group received almost three times as many doses of pregabalin 75 mg capsules as the pregabalin group.
Discussion
Epidural analgesia is considered the gold standard for acute post-thoracotomy pain control (3,5,19), and physicians may assume that more analgesic medications or longer time under anesthesia would be required for patients not receiving epidural analgesia. If such extra medications are required, using pregabalin may not lead to lower medical costs. However, the economic aspects of pregabalin used for postoperative pain relief have not been fully investigated. To our knowledge, ours is the first study comparing medical costs between pregabalin and epidural analgesia as analgesic options for post-thoracotomy pain relief, and we observed that pregabalin did not result in higher costs compared with epidural analgesia.
Total hospitalization costs were similar regardless of which analgesic technique patients received. However, when examining the breakdown, the use of pregabalin as a replacement for epidural analgesia resulted in lower costs for surgical materials and medications as well as for anesthetic management by approximately ¥40,000 in total. The lower costs for surgical materials and medications reflect the non-use of items needed only for epidural analgesia such as catheters, needles, and local anesthetic agents. The lower costs for anesthetic management are attributed not only to the absence of epidural analgesia, but also to the shortened anesthesia induction time, which was shown in our previous study (15). These lower costs did not lead to lower total hospitalization costs; however, considering increasing healthcare expenditures and limited healthcare resources in Japan, it is important to restrain these fee-for-service-based costs.
Costs for outpatient pain-relief medications were similar between the groups despite the fact that the number of patients receiving pregabalin was higher in the pregabalin group. This finding is noteworthy considering the relatively higher unit prices of pregabalin (¥77.0 and ¥128.1) compared with the other drugs (¥8.1–¥105.9). These results can be explained by the number of prescribed doses. Compared with the epidural analgesia group, the pregabalin group received much smaller doses of pregabalin and celecoxib, which were the most and the second most common types of analgesic drugs in both groups. The median doses of pregabalin 75 mg capsules in the pregabalin group were almost one third the number of doses in the epidural analgesia group (84 and 255 doses, respectively). This reduced need for pain-relief medications may reinforce our previous findings that pregabalin may provide better acute post-thoracotomy pain relief than epidural analgesia, although the reduced need did not yield economic benefits, largely because of the high unit prices.
The number of older patients undergoing thoracotomy is increasing as is apparent from the growing number of older patients with lung cancer. In 2013, patients aged 70 years or older accounted for 64.6% of all patients diagnosed as having lung cancer that year (111,837 patients) (20). Among 72,734 patients dying of lung cancer in 2013, 53,529 patients (73.6%) were 70 years or older (21). One concern is that epidural analgesia is difficult to use in older patients because of the high possibility of these patients also receiving anticoagulant therapy or having reduced spinal space. Our results showed no increased economic burden if patients received pregabalin instead of epidural analgesia. Giving patients pregabalin may even save some fee-for-service surgery- or anesthesia-related costs. Along with its efficacy and safety, the economic benefits of pregabalin may make it a useful alternative, especially for older patients.
The reduced need for outpatient pain-relief medications suggests that pregabalin may lead to reduced duration of postoperative pain treatment. Because patients with more intense acute postoperative pain develop more long-term post-thoracotomy pain (22), better acute pain control with pregabalin may help reduce the likelihood of developing chronic post-thoracotomy pain, reducing pain treatment costs in the long term. Considering that chronic post-thoracotomy pain occurs in approximately 50% of patients undergoing thoracotomy (19,23), the potential benefits are substantial. However, the possible associations between pregabalin and potential long-term benefits must be examined in future studies. In the present analysis, we did not assess the numbers of prescribed doses over time; therefore, it is unknown whether doses were gradually decreased over time, or if pain treatment was finished early in the follow-up period before the development of chronic post-thoracotomy pain (e.g., by 2 months post-thoracotomy). In further research, perioperative pain treatment (e.g., the amount of morphine consumed) and the long-term clinical outcomes should be evaluated.
This study has several limitations. First, our analysis was conducted at a single institution in Japan, which may limit the generalizability of the findings to other populations. At this hospital, postoperative pain is managed primarily with epidural analgesia and non-steroidal anti-inflammatory drugs, and selecting pain-relief medications is left to physicians’ judgement. That physicians selected analgesic drugs based on their judgement rather than according to certain criteria implies that physicians tended to continue prescribing pregabalin to patients in the pregabalin group. Such treatment strategies, which are expected to vary among institutions, would largely influence patients’ outcomes and medical costs. Therefore, our results must be interpreted with care. Second, because costs were calculated based on the Japanese healthcare payment system, our results may not be applicable to other societies using different healthcare systems. However, our findings provide useful information for many hospitals in Japan. The number of hospitals adopting the same system is increasing; as of 2014, the DPC/PDPS covered 1,585 hospitals with a total of 492,206 acute care beds, which accounted for approximately 55% of the total hospital beds (894,216 beds) in general hospitals (24). Third, cost data were collected retrospectively, introducing possible selection bias. The observed cost differences may not appropriately reflect differences in analgesic techniques. For example, we did not consider room charges or costs for daily meals when comparing hospitalization costs. Although the total hospitalization costs were similar between the groups, our results may have been influenced by costs not affected by analgesic techniques. Finally, some components incorporated in the in-hospital costs under the DPC/PDPS (e.g., costs to treat comorbidities, adverse events, and pain) were not independently examined in our analysis. Although the distribution of comorbidities could be considered theoretically equal in both groups, some comorbidities may have had a large influence on costs. In-hospital pain treatment should have influenced outpatient pain treatment and would affect costs in the long term. Therefore, further studies are warranted to evaluate the longer-term economic benefits of pregabalin.
In conclusion, our analysis showed that using pregabalin as a replacement for epidural analgesia for post-thoracotomy pain relief did not change the total hospitalization costs, but reduced costs for anesthetic management and for surgical materials and medications. Although the use of pregabalin did not reduce costs for outpatient pain-relief medications, it may reduce the required amount of analgesic drugs. Our results may serve to reinforce pregabalin as a useful analgesic option, especially for patients who are unsuitable for epidural analgesia.
Acknowledgements
None.
Footnote
Conflicts of Interest: N Matsutani has received research grants from Pfizer Japan Inc. Pfizer Japan Inc. had no role in study design; collection, analysis, and interpretation of data; writing of the report; and the decision to submit the report for publication. H Yamane and T Suzuki are employees of Pfizer Japan Inc. A Murakami, Y Haga and M Kawamura have no conflicts of interest to declare.
Ethical Statement: This study was approved by the Institutional Ethics Review Board at Teikyo University (Teikyo University Review Board 15-158-2).
References
- Yegin A, Erdogan A, Kayacan N, et al. Early postoperative pain management after thoracic surgery; pre- and postoperative versus postoperative epidural analgesia: a randomised study. Eur J Cardiothorac Surg 2003;24:420-4. [Crossref] [PubMed]
- Ochroch EA, Gottschalk A. Impact of acute pain and its management for thoracic surgical patients. Thorac Surg Clin 2005;15:105-21. [Crossref] [PubMed]
- Koehler RP, Keenan RJ. Management of postthoracotomy pain: acute and chronic. Thorac Surg Clin 2006;16:287-97. [Crossref] [PubMed]
- De Cosmo G, Aceto P, Gualtieri E, et al. Analgesia in thoracic surgery Minerva Anestesiol 2009;75:393-400. review. [PubMed]
- Ng A, Swanevelder J. Pain relief after thoracotomy: is epidural analgesia the optimal technique? Br J Anaesth 2007;98:159-62. [Crossref] [PubMed]
- Baidya DK, Khanna P, Maitra S. Analgesic efficacy and safety of thoracic paravertebral and epidural analgesia for thoracic surgery: a systematic review and meta-analysis. Interact Cardiovasc Thorac Surg 2014;18:626-35. [Crossref] [PubMed]
- Freise H, Van Aken HK. Risks and benefits of thoracic epidural anaesthesia. Br J Anaesth 2011;107:859-68. [Crossref] [PubMed]
- Ben-Menachem E. Pregabalin pharmacology and its relevance to clinical practice. Epilepsia 2004;45 Suppl 6:13-8. [Crossref] [PubMed]
- Sabatowski R, Gálvez R, Cherry DA, et al. Pregabalin reduces pain and improves sleep and mood disturbances in patients with post-herpetic neuralgia: results of a randomised, placebo-controlled clinical trial. Pain 2004;109:26-35. [Crossref] [PubMed]
- Baidya DK, Agarwal A, Khanna P, et al. Pregabalin in acute and chronic pain. J Anaesthesiol Clin Pharmacol 2011;27:307-14. [Crossref] [PubMed]
- Toth C. Pregabalin: latest safety evidence and clinical implications for the management of neuropathic pain. Ther Adv Drug Saf 2014;5:38-56. [Crossref] [PubMed]
- Agarwal A, Gautam S, Gupta D, et al. Evaluation of a single preoperative dose of pregabalin for attenuation of postoperative pain after laparoscopic cholecystectomy. Br J Anaesth 2008;101:700-4. [Crossref] [PubMed]
- Engelman E, Cateloy F. Efficacy and safety of perioperative pregabalin for post-operative pain: a meta-analysis of randomized-controlled trials. Acta Anaesthesiol Scand 2011;55:927-43. [PubMed]
- Mishriky BM, Waldron NH, Habib AS. Impact of pregabalin on acute and persistent postoperative pain: a systematic review and meta-analysis. Br J Anaesth 2015;114:10-31. [Crossref] [PubMed]
- Matsutani N, Dejima H, Nakayama T, et al. Impact of pregabalin on early phase post-thoracotomy pain compared with epidural analgesia. J Thorac Dis 2017;9:3766-73. [Crossref] [PubMed]
- Matsuda S, Ishikawa KB, Kuwabara K, et al. Development and use of the Japanese case-mix system. Eurohealth 2008;14:25-30.
- Ishii M. DRG/PPS and DPC/PDPS as Prospective Payment Systems. Japan Med Assoc J 2012;55:279-91. [PubMed]
- Merskey H, Bogduk N, editors. Classification of chronic pain: descriptions of chronic pain syndromes and definitions of pain terms. 2nd ed. Seattle: IASP Press, 1994.
- Soto RG, Fu ES. Acute pain management for patients undergoing thoracotomy. Ann Thorac Surg 2003;75:1349-57. [Crossref] [PubMed]
- Cancer Information Service, National Cancer Center, Japan [Internet]. National estimates of cancer incidence based on cancer registries in Japan (1975-2013). [cited 2017 Dec 4]. Available online: http://ganjoho.jp/en/professional/statistics/table_download.html
- Vital Statistics in Japan, tabulated by Cancer Information Service, National Cancer Center, Japan [Internet]. Cancer mortality from Vital Statistics in Japan (1958-2015). [cited 2017 Dec 4]. Available online: http://ganjoho.jp/en/professional/statistics/table_download.html
- Katz J, Jackson M, Kavanagh BP, et al. Acute pain after thoracic surgery predicts long-term post-thoracotomy pain. Clin J Pain 1996;12:50-5. [Crossref] [PubMed]
- Rogers ML, Duffy JP. Surgical aspects of chronic post-thoracotomy pain. Eur J Cardiothorac Surg 2000;18:711-6. [Crossref] [PubMed]
- Ministry of Health, Labour and Welfare [Internet]. Overview of the revision of medical fees (parts related to DPC) in 2016. D-3. [cited 2016 Jul 12]. Available online: http://www.mhlw.go.jp/stf/shingi2/0000125205.html