Lung inflammatory response syndrome after cardiac-operations and treatment of lornoxicam
Introduction
The year of 1953 consisted as a milestone in the development and evolution of cardiothoracic surgery through the establishment of the machine of extracorporeal circulation in the surgery of heart. For the first time, Gibbon (1) performed the first successful convergence of atrial Septal Defect in human in Jefferson Medical School of Philadelphia.
In 1954, Lillehei and Varco (2) applied in University of Minnesota, the “Technique of Cross Circulation” in infants and children, for the treatment of Congenital Heart Disease. For the provision of arterial blood they used a simple pump and an oxygenator in the lungs of an adult. With this technique, 45 surgeries took place with good results. Shortly thereafter Dewall et al. (3) manufactured the first bubble oxygenator.
In 1955, Kirklin et al. (4) in Mayo Clinic, began to use systematically the oxygenator and Gibbon’s Extracorporeal Circulation by which eight patients with Congenital Heart Disease were treated. Since then, the development and evolution of modern cardiothoracic starts.
The majority of patients survive after Extracorporeal Circulation without any clinically apparent deleterious effects. However, disturbances exist in various degrees sometimes, which indicate the harmful effects of cardiopulmonary bypass (CPB) in the body. The disorders are the following:
- Compulsive tendency for intra and extra tissue hemorrhages;
- Inflammatory response which is characterized by an increase in the permeability of the capillaries, increase in interstitial fluid, leukocytosis and fever;
- Renal impairment;
- Peripheral vasoconstriction with hemodynamic and metabolic disorders;
- Injuries (broken) of red blood cells resulting in hemoglobinemia, hemoglobinuria and anemia.
These disorders are subscribed as postperfusion syndrome or postpump syndrome.
The causes implicated in causing these complications is the exposure of blood to non-biological area (circuit tubing and oxygenator of extracorporeal circulation), damage from ischemia and reperfusion, endotoxemia and surgical wounds.
Several factors during extracorporeal circulation either mechanical dependent (exposure of blood to non-biological area) or mechanical independent (surgical wounds, ischemia and reperfusion, alteration in body temperature, release of endotoxins) have been shown to trigger the inflammatory reaction of the body. The complement activation, the release of cytokines, the leukocyte activation and accumulation as well as the production of several “mediators” such as oxygen free radicals, metabolites of arachidonic acid, platelet activating factors (PAF), nitric acid, and endothelin (5) are part of the process that leads to tissue damage and organ dysfunction.
These disorders can involve clinical manifestations ranging from the emergence of Systemic Inflammatory Response System-SIRS) to (Multiple Organ Dysfunction Syndrome-MODS). The severity of these conditions evidenced by the fact that people who have been under SIRS, life expectancy is reduced and the likelihood of death is increased even after the lapse of five years from the appearance of the syndrome. Warltier in their reviewing article, report that the impact of MODS after extracorporeal circulation amounts to 11%, with 41% mortality in the patients of this group.
American College of Chest Physicians and Society of Critical Care Medicine Consensus Conference Committee in 1991, established the criteria of a new classification of these statements depending on existing clinical findings (Table 1). The majority of literature reports focus on left ventricular dysfunction, giving little importance to the damaging effects of the inflammatory response of the lungs. However, lungs suffer often from the influence of inflammatory reaction which occurs in the form of pulmonary infiltrates and respiratory dysfunction, whereas at the cellular level is characteristic the activation of mast cells and macrophages, cell population is responsible for protecting the alveoli and vroncholion by microorganisms and particulate. Royston et al. (6) have shown in their study that after extracorporeal circulation is observed a disturbance of lung function in the first three postoperative days, while the disorders in gas exchange last for about a week.

Full table
The oxicams consist a new category, non-steroidal anti-inflammatory drugs (NSAIDs) which belong to the group of enolic acids. In these belong piroxicam, meloxicam, and tenoxicam whose chemical structure different to a ring only. Other medicine of this category is lornoxicam, cinnoxicam, sudoxicam as well as some piroxicam’s derivatives such as: apiroxicam, droxicam, pivoxicam, which have been created to reduce undesirable effects of gastrointestinal system.
The oxicams consist active drugs for the treatment of arthritis and the postoperative inflammation (7). The last decade, some other actions of oxicams have been discovered such as chemoprophylaxis in various neoplastic conditions (8-10) and skin protection in dermatitis caused by ultraviolet (11-14). Furthermore, they present remarkable antioxidant activity.
The majority of oxicams are related compounds produced by replacing the ester molecules with other mineral substances. Accordingly share of the parent molecule are replaced with other departments similar to solid-electrical characteristics, designed to improve the pharmacological actions the pharmacokinetics and other properties such as the greater resistance of oxicams and their enzymatic deconstruction. The configuration of the piroxicam’s molecule is different because of the existence of an incomplete hydrogen bond between the alkaline OH of the group of enolic acids and aminocarbonyl oxygen. These hydroxyl radicals are proved to have increased intermolecular proton transfer when the molecule is in the excited state (excited state intramolecular proton transfer, ESIPT) and they are transformed into their keto form.
Concerning about action of oxicams in the gastric mucosal, Villegas et al. (15) proved experimentally that meloxicam causes the same damages in the gastric mucosal as piroxicam. Furthermore the writers bolster that the mechanism of action of these substances in the gastric mucosal comprare to other NSAIDs such as indomethacin and diclofenac, is different. Specifically oxicams have a harmful affection on gastric mucosal via the oxidation of xanthine in neutrophils while indomethacin and diclofenacaffect on gastric mucosal via the metabolism of glutathione (16,17).
Although the NSAIDs show a harmful effect on gastric mucosal, however there is a rating among these harmful effects. Lugardon et al. (18) compared oxicams with coxibs, ibuprofen and diclofenac, and concluded that oxicams present lower intensity devastating injuries in gastric mucosal. Regarding the absorption of oxicams in the digestive system: oxicams connected to the gastric environment with a circular oligosaccharide (β-cyclodextrin or β-cd) which protects them. Follows, in the intestinal environment these connection is disrupted and oxicams cross the intestinal barrier to incoming blood incoming to blood (19).
Cardiopulmonary bypass and operating principles
With the term of CPB or extracorporeal circulation, we mean the BYPASS heart function bypass heart function and lungs, with the help of a special machine (Figure 1).
The mainly part of a contemporary CPB is:
- Supply pump arterial blood;
- Oxygenator;
- Pumps for suction and return of blood from the surgical field to oxygenator (coronary aspirates);
- Pump to extract (venting) the left ventricle;
- Special pumps for granting of cardiologic;
- Device for causing systematic hypothermia and rewarming of patient (heat exchanger).
The binding of the patient with CPB is made with the help of circuit tubing which was inserted the oxygenator and arterial blood filter.
Venous drainage
Patient’s venous blood whisk by gravity through the endocardial catheters, the vena cava or right atrium or rarest from the femoral vein, the jugular or the iliac vein entering the oxygenator. The venous return hampered when there are obstacles in the venous line (contracture tube or trapping air within it). In such cases it is possible, as long as the operation of the blood pump continues, to empty fully the oxygenator and large amount of air to be promoted through the patient’s arterial system.
Arterial supply
The arterial blood from the oxygenator, is advanced through special catheter in the ascending aorta or rarely in peripheral arteries such as the femoral artery, the anonyma, the flanks, and the axillary artery. The size of the arterial catheter determined from the body surface area of patient and the projected flow of CPB. The developed pressure in the arterial line of CPB is proportional to the supplied blood flow and inversely proportional to the inner diameter of the arterial catheter.
Venous cisterns
Represent an area of collecting blood from the surgical field which dissipates with coronary suction. Blood from hear cistern channeled towards to the oxygenator. Now with the use of modern membrane oxygenators, this cistern is embodied in the oxygenator.
Oxygenator
The oxygenator is a part of extracorporeal circulation in which oxygenation of arterial blood is achieved. It has not yet achieved full functional substitution of the lungs from the oxygenators (e.g., neutralization of substances or enzymes is not achieved in the oxygenator). Major drawback remains the relatively small in relation to lung, surface provided for gas exchange (Table 2).

Full table
There are two main types of oxygenators in clinical practice:
- Bubble oxygenators, where the oxygenation of the blood achieved by the direct contact with the delivered oxygen. Major drawback is the wound of cellular blood elements;
- Membrane oxygenators, in which a thin-permeable gas membrane is inserted between blood and provided oxygen. These oxygenators are more similar to human lungs because they carry interface of porous materials between blood and gas. These oxygenators have replaced the bubble oxygenators because of their fewer complications (Table 3).
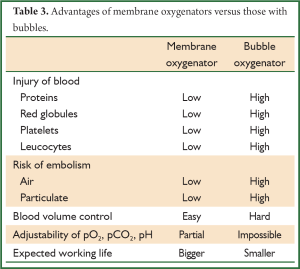
Full table
Lung’s comparative data of both types oxygenizenators.
Bubble oxygenators, where the oxygenation of the blood achieved by the direct contact with the delivered oxygen. Major drawback is the wound of cellular blood elements;
Heat exchanger
This is an additional device of extracorporeal circulation, which has the capacity of cooling-reheating the patient’s blood in the various cardiac surgeries. This device is filled with distilled water, which is cooled to the appropriate temperature depending on the planned intervention. Cool water circulates through piping systems which surround the bottom of oxygenator. As a result, the blood passes through the oxygenator and cooled. The opposite process is followed during the phase of reheating patient, which shouldn’t be rapid. Prohibit differences in temperature between water-blood of the patient greater than 12-14 °C.
Pumps
In most cases a roller pump is used, which promotes the oxygenated blood from the oxygenator in the ascending aorta or femoral artery. The blood pump is consisted of a swivel arm, variable length, the ends of which find two identical rotating cylinders. The angle between the cylinders is 180° so that one of them acts as a valve. Between the outer surface of the space in which the swivel arm is rotated, and the cylinders, the tube of arterial supply is inserted. The cylinders during rotation, do not obstruct fully the tube, succeeding in this way both consistent absorption of blood to the oxygenator, and the continuous promotion to the arterial system of the patient. With these pumps, it is possible to provide blood from 0.1 to 9.9 L/min, depending on the rate of pump speed and the internal diameter of the arterial line.
The newest form of arterial pump is centrifugal pump, in which the advancement of bloods done by swirling a plastic cone (Biomedicus pump). This pump cause to some extent pulsate flow conditions. The trait of this pump is the small degree of induced hemolysis in comparison with conventional pumps.
Despite the streaming blood supply there is the pulsatory flow which is similar to the arterial circulation in humans. Experiments have shown that pulsatory flow presents some advantages when extracorporeal circulation is extended. When extracorporeal circulation lasts 1-4 hours the pulsatory flow is not superior to streaming blood supply. Comparatively the advantages and disadvantages are presented to the Table 4.

Full table
For more efficient drainage of returned blood to the heart-through bronchial movement, during extracorporeal circulation, vent probe is used. The entrance of this catheter can be done through the apex of the left ventricle, the right upper pulmonary vein, the flap left atrium, pulmonary artery, or even through the ascending aorta.
The placement of vent via apex of the left ventricle is simple and effective, is accompanied with low complication rate, but they rarely hazardous for the life of the patient [e.g., burrowing branch of the left anterior descending aorta, air inlet into the left ventricle, postoperative bleeding from the catheter entrance, formation of small aneurysms input area in the longer] (20). This technique today, has almost abandoned. The placement of vent through the right upper pulmonary vein is also simple and outperforms the previous method because the wound infarction is avoided. However, this technique is related to some complications: postoperative bleeding from the entry point, significant degree of stenosis of the pulmonary veins and challenge venous-stasis in the upper and middle lobe, through the wall operation of the left atrium and left ventricle and failure of mitral valve (21). The decompression of the left ventricle with the vent’s placement in the pulmonary artery currently consists endearing technique which essentially is not accompanied by these complications (20,21).
Filters trapping micro particles
It has been approved that during extracorporeal circulation, aerial microemboli formed, which released from oxygenator into the arterial line of the extracorporeal circulation. The transmission of these vaccines within the arterial system of the patient will have devastating consequences. For this reason inserted into the arterial limb of the machine special filter trapping air bubbles, in order to prevent this complication. Such particles and aerial micro emboli preexist in the circuit tubing of extracorporeal circulation before the start of CPB which are created during filling circuit. Furthermore the mass production of gassy pistons can be remarked from the sharp increase the supply of blood pumping. As a result the use of filters with small pores in the arterial providing of extracorporeal circulation, protect to some extent various organs from micro emboli during irrigate.
Nowadays almost all cardiac surgery centers (>97%) filters are used in the arterial line of extracorporeal circulation. By clinical and laboratory studies found that the filters of 40 μm do not cause increased hemolysis when placed in the arterial line. Platelets, leucocytes and levels of fibrin in the blood, vary during Extracorporeal Circulation, regardless of the interference filters (22). But there are studies (23) that argue that these filters cause destruction of proteins and cell accumulation on blood components.
On the protective effect of filtering in brain function, experimental researches of Taylor et al. (24), indicated raise in the special enzyme CPK-BB in the cerebrospinal fluid, in cases not involving the arterial line filters. Clinical work of Winer et al. showed that the neurological effects in patients to whom were used filters of 40 μm, decreased more when filters with smaller pores of Nylon 25 μm were used, which are disadvantageous because they cause hemolysis.
Circuit tubing
The various parts of the circuit of extracorporeal circulation connected by tubes made of polyvinyl chloride and polycarbonate binder. The piping system is made of PVC which is flexible, biocompatible, non-toxic, inert, smooth, waterproof, durable, and clear, resists contracture and can be sterilized.
From the mid 90’s are in widespread clinical application systems of piping and membrane oxygenators coated with heparin (25,26). The use of these systems was made with the prospect of impairment administered heparin and reduction of thromboembolic events. However there is no convincing evidence that plated circuit with heparin require less heparin for the systematic heparinization of the patient, nor that it reduces the bleeding events of thromboembolism. The one which was shown by the studies is that heparinized cells reduce the concentration of C3a and C5b-9 (27) but without affecting the inflammatory reaction of the body that trigger from extracorporeal circulation (28).
Infarction protection
The protection of infraction during extracorporeal circulation particularly during myocardial ischemia is a prerequisite for the outgrowth (rare) or damage limitation of myocardial cells. Occasionally have implemented several protection measures of infarction such as systematic hypothermia, the application of a continuous or periodic irrigation of coronary artery blood directly from the circuit of extracorporeal circulation and other techniques. Every technique has its disadvantages which eventually reflect the deficient protection of infarction in the ischemic phase and causing myocardial damage ranging from edema and myocardial cell necrosis of these. O’Connor et al. (29). studied 8,641 patients who underwent coronary bypass and they reported overall mortality 4.48%. The majority of deaths (65%) attributed to operatively heart failure. The intra operative myocardial infarction was a dominant factor of poor postoperative course, pointing out that persistent effort must be made to improve existing protection techniques (30).
The duration of ischemia after it is developed irreversible damage belly is not precisely defined but is around 30 min. The observation that the endokardio and hypertrophic ventricles are more vulnerable to ischemic injury, led to conclusion that the disproportion between energy supply and myocardial oxygen requirements and the myocardial cells demands in energy and oxygen is the mainly mechanism for the post ischemic heart operation. So, the first efforts to protect infraction turned to searching for ways to reduce the demands for myocardial oxygen.
Final measure of protection infarction
Localized cooling infarction
The localized cooling of infraction as a method of reducing energy requirements was introduced clinically by the late 50 s by Norman Shumway. Hypothermia reduces the energy requirements of infraction restricts as was proven by Sonnenblick, the oxygen consumption of infraction, it improves cardiac voltage, heart rate and contractility of the myocardium. By decreasing the above parameters oxygen consumption from infarction drops to a level which was defined as “basic”. Despite the absence of contraction the myocyte requires oxygen to satisfy the basic functions which further reduces the temperature drop. Buckberg et al. showed that normothermia pause (37 °C) reduces the Cardiac oxygen demands by 90% (1 mL O2/100 gr/min). In the 22 °C the oxygen demands are reduced to 0.3 mL/100 G/min with heart paused. In this temperature the reduction of myocardial oxygen consumption touches essentially a “flat”.
With these studies hypothermia was established as an effective method protection of infarction during cardiac ischemia, but it soon became clear that only hypothermia was not sufficient for intraoperative protection.
Cold crystalloid cardioplegia
The application of cold crystalloid cardioplegia improved significantly the cardio protection in heart surgeries. There are two types of crystalloid cardioplegic solutions, depending on their constitution:
- Intracellular solutions whose constitution is similar to those of intracellular fluid and is characterized from the absence or the low concentration sodium and calcium;
- Extracellular solutions whose constitution is similar to those of extracellular fluid and is characterized from high concentrations sodium, calcium and magnesium.
The granting of crystalloid cardioplegia happens after the blockage (occlusion) of the ascending aorta either at the root of the aorta (ante grade cardioplegia) or through the coronary sinus (retrograde cardioplegia). Initially grants a quantity of 800-1,000 mL cold (4 °C) cardioplegic solution and follows short iterative administrations (300-500 mL) of 20 min usually, or even smaller, if heart displays electrical activity.
Although there are many controversies regarding the ideal cardioplegic solution, however cold crystalloid cardioplegia is used exclusively in many cardiac surgery centers. In many studies, it is reported that the preoperative frequency is less than 4% and surgical mortality less than 2%. Furthermore, in other studies is reported that this type of cardioplegia reduced the postoperative levels of CPK-MB, while postoperative indicator (CI) was independent from the time of blockage of aorta.
Cold blood cardioplegia
Another of cardioplegia is cold blood cardioplegia which is applied in the majority of cardiac surgery centers of USA and Europe. It is consisted of the blood which is taken directly from the circuit of the extracorporeal circulation and is enriched with crystalloid solution which contain electrolytes (mainly potassium and magnesium) in several concentrations. Cold blood cardioplegia’s pH amounts to 7.8 and concentration of potassium to 30 mmol/L. Before granting cardioplegic solution, the solution is cooled with exchanger so that the temperature ranges from 12-4 °C. The ratio between blood and crystalloid solution various from 8:1, 4:1, 2:1, and it depends from the patient’s hematocrit. In some cases, “miniplegia” is used which is a blood mixture with a minimum amount of crystalloid solution (66:1), although no substantial superiority of cardioplegia was found with the aforementioned.
The providing of cool blood cardioplegia is done either orthodoxy to the root of the aorta or directly in coronary hydrants or retrogradly through the coronary sinus.
The major advantage of this cardioplegia compare to crystalloid is:
- The possibility of periodic oxygen delivery to ischemic myocardium and as a result the maintenance its aerobic metabolism to some extent. As a result the production of lactic acid is reduced while simultaneously high energy phosphates are marinating in some levels (31);
- The possibility of clearing the free radicals of oxygen from red blood cells of cardioplegia with the help of antioxidants with the consequent reduction of oxidative stress (32).
The antioxidant activity of red blood cells tried to enhance some researchers by adding substances into the cardioplegic solution. Viten-Johansen et al. and Menasche et al. (33) studying the efficacy of blood cardioplegia added antioxidants such as allopurinol and desferoxamine and observed improvement in post ischemic functionality of ventricles. However, the addition of such substances did not receive widespread clinical application.
Warm blood cardioplegia
The delay in the metabolic and functional recovery after myocardial ischemia phase is probably due to the action of suppressing myocardial products enzymes because of low temperature caused by cold cardioplegic pause. In 1982, Rosencranz et al. (34) showed that the blood cardioplegic cessation improves metabolic and functional recovery of the myocardium. An experimental study of Teoh et al. (35) demonstrated that a dose of hot (37 °C) blood cardioplegia (hot shot) just before the removal of AOC had a favorable effect on metabolic recovery of my heart, while it maintained the electro pause. The authors argue that normothermic dose leads to faster restoration of the enzyme activity of mitochondria and aerobic metabolism resulting to faster reconstruction of the ATP. These studies laid the initial groundwork for the implementation of normothermic blood cardioplegia.
Interest is the work of Lichtenstein et al. (36) who applied normothermic continuous blood cardioplegia during the phase of myocardial ischemia in patients who were submitted into coronary bypass in order to enhance further the metabolism of myocardium during the intraoperative and immediate postoperative period. Comparing these patients with a group of patients treated with cold blood cardioplegia, researchers found that patients of normothermic group had showed lower rate of per operative myocardial infarction and IABP. In another study with 2,000 patients who underwent coronary artery bypass, compared the effects of continuous administration normothermic blood cardioplegia versus intermittent cold blood cardioplegia. Although there were no differences in perioperative mortality and incidence of perioperative myocardial infarction, patients of normothermic group showed lower incidence of low cardiac output syndrome.
Enrichment cardioplegic solutions with energy substrates
Undoubtedly with the use of blood cardioplegia was achieved on the one hand the requirements of reducing myocardial oxygen and also better control of oxidative “stress”. Further improvement of these metabolic processes could be achieved by enriching the cardioplegic solution with various substances, aimed at the highest rate of reconstitution ATP.
Occasionally different energy substrates were used such as adenosine, amino acids (glutamate, aspartate and ornithine, L-arginine) insulin, L-carnitine and scavengers of free radicals of oxygen. Although these substances are shown to improve the metabolism of myocardial cells during the ischemic period and the contractility of myocardial fibber during the phase of reperfusion, however, they do not receive widespread clinical application.
Key factors releasing inflammatory reaction
Supplement
The complement system comprises a set of 30 plasma proteins with potent vocative anaphylatoxins the C3a, C4a, C5a and the final product toxic C5b-9 (37).
The complement is activated by three pathways, but the EC involved classical and the alternative pathway mainly (38). Complement’s activation during EC is accomplished in three phases:
- From blood contact with no biological surfaces (38-41);
- The administration of protamine and the complex formation of protamine-heparin (38-42);
- During reperfusion after the ischemic period of the heart (43).
The final toxic product of the above phases is the production of C5b-9, which causes the pores in the cell membrane of erythrocytes and destruction of intracellular oedema. In Macrophages C5b-9 induces the production of arachidonic acid metabolites (A2 thromboxanes, leukotrienes), while in neutrophilst the production of oxygen free radicals (40).
In normal situations, the human body has factors (factors H and I, complement receptor 1-CD35, membrane cofactor protein-CD46 and decay accelerating factor) that inhibit the action of complement C5b-9, but these are destroyed during the EC (44).
Complement activation has been implicated in a number of years for the increased mortality after EC (45,46) but nevertheless studies are investigating the involvement in the development of acute lung injury (47).
The Song Wan and colleagues (5) in their retrospectively article that elevated levels of C3a are observed during EC. These levels remain heightened extracorporeal circulation and the level of increase is associated with the period of CPB, but not with the length of the membrane oxygenator. The same authors reported that postoperative morbidity is associated with the supplement and that there is no relationship between complement activation and acute lung injury.
The Cugno M and colleagues (48) found increased production of bradykinin (a derivative of prostaglandin, a potent vasodilator which causes increased capillary permeability) during EC, yielding the increase in non catabolism of the substance in the lungs, which are excluded from the bloodstream. Also, the same authors state that there is a direct correlation of the levels of bradykinin in the recovery time of the pulmonary circulation.
Neutrophils
The Brown GM and colleagues (49) studied the participation of neutrophils in the inflammatory response of the lungs. Polymorph nuclear neutrophils promote detrimental effects on the microcirculation and tissue in one of three mechanisms:
- Cells become stickier by the expression of adhesion molecules or reducing their deformation capacity;
- Neutrophils can be stimulated resulting the secretion of cytotoxic agents, out of which oxygen radicals and proteolytic enzymes are most often involved. These two processes interact with the remaining rows of white blood cells;
- It is possible that the lung damage is the result of lung reperfusion with oxygenated blood, after a period of ischemia.
The Markos J and partners (50) interpreted the mechanism of in causing lung lesions and they studied the passage of neutrophil in the pulmonary circulation. It was noted that just before the release of the EC and with conducted hyperventilation of the lungs, isolation of the cells is caused in the pulmonary vascular bed which was described as “isolation region of neutrophils”. Similar action has the application of positive end-expiratory pressure (PEEP) from the ventilator.
The Gebb SA and collaborators (51) in their experimental study in dogs-tried to discover isolating areas in leucocytes using a video-microscopy system for high resolution. They found that the leucocytes move smoothly (rolling) along the artery walls when it is almost spherical. Their passage through the capillary network causes a change in their shape, they become elongated, and many of them remain and many of them remain elongated after entry to venules. Despite the change of shape, 46% of leukocytes moving smoothly, while 54% is fixed at some point in their course.
By using the same technology shown that the recirculation leukocytes develop rough connections without rolling or moving slowly, with the endothelial cells of the respiratory membrane -process which contributes to the accumulation of neutrophils. It appears that the reduction of the deformability of cells in the pulmonary microcirculation is the root cause that promotes delay or trapping of neutrophils, thereby creating formations adhesion between neutrophils and endothelial cells.
Of course it should be noted that the neutrophils are not easily lose their shape (which leads to trapping in capillaries), but if this occurs and the trapped cells are isolated then they are activated and they contribute to the pathogenesis of ischemic disease and lung injury (52). The EC reduces the ability to change the shape of red and white blood cells, thus increasing their adherence (53). Maximum reduction observed one day after the surgery. Generally, the change in cell shape depends on:
- The relationship surface-volume of the cell;
- The elastic and viscous properties of the cell membrane and cytoplasm. With regard to the red cells and neutrophils, these cells during EC modify the elastic properties and the degree of modification associated with the duration of the EC.
Leukocytes are also found to be involved in the early stages of cerebral damage which sometimes follows the EC because infiltrate the region and causing ischemic cerebral oedema (54).
Monocytes
Since 1995 already Naldini A and colleagues (55) postulated that the immunosuppression observed during and after EC associated with significant decrease of the proliferation of activated monocytes in the peripheral blood. Monocytes produce and release many mediators of the inflammatory response during acute inflammation. The mechanism of initial activation during EC is not known, but it appears that four factors implicated for this: complement C5a, platelet factor 4-PF4, thrombin and bradykinin, which are created by the contact of the blood to non-biological surface (56-58).
Monocytes produce IL-1b, TNFa, interleukin-8 (IL-8), IL-1a, interleukin-6 (IL-6), IL-10, e-proteins of extracellular amorphous substance (matrix proteins), interferon, prostaglandins, products of lipooxygennasis, myeloperoxidase and nitric oxide (NO) (56,58). Chemokines such as MCP-1, MCP-4 (monocyte chemo tactic protein), MIP-a, MIP-b (monocyte inhibitor protein) and RANTES (regulated upon activation normal T-cell expressed and presumably secreted) are produced by monocytes and simultaneously activate them. Also monocytes exhibit a large number of surface receptors for the binding of C5a, CD11b/CD18, IL-1, CD11c/CD18 and LTB4. Monocytes externalize β1, β2, α1β4 which are associated with endothelial cell receptor ICAM-1, ICAM-2, VCAM-1 to allow the movement in response to chemo tactic stimulation.
Endothelial cells
The primary factors that activate endothelial cells during EC is thrombin, the C5a, as well as IL-1b and TNFa These factors promote crack exteriorization of P-selectin and then the E-selectin, that trigger the initial stages of neutrophil adhesion and monocytes.
Both cytokines, IL-1b and TNFa, cause the externalization of ICAM-1 and VCAM-1 and which bind neutrophils and monocytes to endothelium and promote trafficking of leukocytes into the extra vascular space. Also these cytokines act on endothelial cells and promote the production of chemotactic proteins such as IL-8, IL-6, PAF, and MCP-1 as well as production and prostakiklinis NO. Additionally endothelial cells produce endoithlini-1 and inactive other vocative agents s histamine, norepinephrine and vradikinini (59).
Endothelin is the most potent vasoconstrictor that has been found, which is involved in regulating blood pressure and cardiac output . Elevated endothelin levels have been found in patients with congenital or rheumatic heart diseases and have been associated with pulmonary hypertension. The kidneys are about ten times more sensitive to the effects of endothelin in relation to the other organs. The maximum increase in plasma endothelin which is observed at the end of the EC, is related directly to the time duration, but has a negative correlation with the volume of urine during the EC. Possibly the elevated levels of endothelin are related to renal impairment following the EC.
The release of endothelin-1 can lead to a high degree of cardiac muscle ischemia although is not released into by the endothelium of the coronary arteries or myocardium. It is a fact that the lungs are an important part in the production and clearance of endothelin-1.
The vasoconstrictive properties of endothelin may play a role in poor perfusion of colon, resulting in the release of endotoxin in circulation, which in turn may stimulate the production of endothelin. In the post-ischemic hematose heart, the vasoconstrictor property of endothelin-1 causes the release of thromboxane A2, while regional induced vasoconstriction of small pulmonary veins is a significant case with consequent causing pulmonary edema (5). In addition, endothelial dysfunction may lead to hyperplasia of medial vascular and consequently the narrowing of the graft at the site of anastomosis, and progressive atherosclerosis. The vasoconstrictive properties of endothelin can be reversed by nitroglycerin.
Platelets
Platelets are activated primarily by thrombin and of plasma epinephrine, vasopressin and the PAF. The connection to the collagen causes the release of thromboxane A2. Because of the strong reaction to wound and extracorporeal circuits, the “contribution” to the inflammatory response is complex with the release of eicosanoids, IL-1b, serotonin, IL-8, MCP-1, MCP-4, MIP-a, MIP RANTES-b and a granules. Circulating neutrophils and monocytes during EC are associated with platelet aggregation by means of P-selectin that is secreted by activated platelets.
The PAF is a phosholipid which is synthesized in platelets, vascular endothelial cells and possibly in the myocardium. The PAF receptors may be present not only on platelets and neutrophils, monocytes and endothelial cells. The PAF plays a complex role in myocardial injury by reperfusion ischemia. It has been found to cause damage to the myocardium and hemodynamic burden and research studies have shown its role as mediator in myocardial and lung injury following the EC. Inhibitors may reduce morbidity after the EC (5).
Other mediators of inflammation
Cytokines
Cytokines are peptides with a molecular weight of 5,000 to 70,000 that act on local targets to promote cell growth, apoptosis, or chemo taxis. Cytokines are produced from blood cells or tissue and are released in the blood or the extra vascular space. The collaborative action between the cytokines and the target cells enhances the inflammatory response to attract leukocytes to the site of injury and the additional release of cytokines. The lungs act as a major source of pro-and anti-inflammatory cytokines (60,61).
During EC have confirmed different levels pre-and anti-inflammatory cytokines, and has found that the length of time the EC and time of AOC affect the levels of cytokines.
Controversial is the issue of detection of IL-1 in peripheral blood during and immediately after the EC. The Butler J and associates (62) and Cavaillon JM and collaborators (63) support non-detection in the peripheral blood of IL-1 at the duration of the EC whereas by contrast, the Steinberg JB and colleagues (64) found an increase of (64) IL-1 in the plasma, with a maximum value of three hours after the EC.
The main anti-inflammatory cytokine is IL-10, which is produced from the T2 cells, monocytes, tumor B cells and Kupffer cells. The IL-10 acts buffers in blood monocytes by stimulating receptor antagonist IL-1 and suppressing the synthesis of IL-8, IL-6 by T cells (65,66) The IL-10 also inhibits the synthesis of pro-inflammatory cytokines by monocytes and macrophages but its role during the EC remains undetermined (67). Mtairag and associates found that IL-10 causes a decrease in the adhesion of monocytes to endothelial cells.
Wan et al. (68) demonstrated that IL-10, TNF-a and IL-6 are produced mainly in the myocardium rather lungs. Other inflammatory cytokines are IL-1ra (interleukin-1 receptor antagonist), are TNFsr 1 and 2 (tumor necrosis factor, TNF soluble receptors 1 and 2) and transforming growth factor b (transforming growth factor-b, TGF-b). It appears that the clinical prognosis after EC is determined by the balance of pro-inflammatory and anti-inflammatory cytokines.
The a2-macroglobulin or a2-M is a widely-produced protein which binds to the IL-6, IL-8, TNF-a, IL-1b. The precise role in the regulation of cytokines is unknown but argues that participates in the clearance of cytokines from plasma and extracellular fluid.
Positive correlation was found between the group of thrombin-antithrombin III (thrombine antithrombin III complex or TAT) and S-100B, TAT and sP-selectin, FDP and sE-selectin, assuming the increase in thrombin production during or after the EC in patients which have brain damage, causing the extension of the lesion.
Interleukin-6 (IL-6)
The IL-6 is a cytokine with many biological properties. Regulates immunological viral body’s response to acute inflammation. Studies have demonstrated that regulates the synthesis of factors: fibrinogen, haptoglobin, serum amy-loid-P (SAP) and C-reactive protein (CRP). IL-6 is produced in the acute phase of inflammation of the T cells from the endothelial cells and monocytes.
IL-6 is increased during and immediately after the EC and displays gradually decline to pre-surgery levels 3-5 days later. Postoperative plasma concentration appears to be highest in complicated cases.
Increased IL-6 is pointed in normothermia and hypothermia and in cases where they are used heparin zed circuits EC. IL-6 does not contribute to cell damage or to hemodynamic instability, so it is considered to be an indicator only “mediator” of the inflammatory response. In a study of pediatric patients who underwent cardiac surgery it was observed that elevated IL-6 levels in serum was accompanied by increased postoperative mortality, while elevated levels of IL-6 only in bronchoalveolar lavage fluid were accompanied by increased postoperative morbidity.
An important finding was that the levels concluding that the lungs consume rather than produce these mediators during reperfusion. The main difference between the two institutions was that the heart was exposed to complete ischemia and topical hypothermia, while the lungs were almost normothermic and possibly partially hematose through the bronchial circulation. The authors conclude that the global myocardial ischemia during the exclusion of the aorta, not the EC itself appears to be a critical factor in heart cytokine release.
Interleukin-8 (IL-8)
IL-8 is a factor that is released by the stimulation of neutrophils. It is the primary mediator for the concentration of neutrophils in the lungs after the EC and it appears in the blood is after 24 hours of EC (69). The IL-8 correlates with impaired mobility infarction and blood levels increases more the longer the left ventricular dysfunction (70).
IL-8 has been detected in bronchoalveolar lavage in adults and pediatric patients who underwent cardiac surgery under extracorporeal circulation (71). Frering B and colleagues (72) suggest that IL-8 plays an important role in the damage caused by reperfusion where the primary role play macrophages in the alveoli. Similar conclusions reached and other writers (73,74).
Tumor necrosis factor (TNF)
The TNFa activates neutrophils and in synergy with IL-1 affects endothelial cells by activating the pre-thrombotic state. The TNFa has been implicated in the pathogenesis of sepsis and in combination with IL-1 for the complications are involved in sepsis (75).
Levels of TNFa increases at the end of EC up to two hours afterwards. This increase can be correlated with the degree of impairment of left ventricular.
The TNFa is involved in the pathophysiology of the vessel palsied syndrome (76,77) during the immediate postoperative period, in which patients experience transient low systemic vascular resistance, hypotension, reduced filling pressures of the ventricles, normal or increased cardiac output and tachycardia. The treatment of this syndrome is the vasopressin agent (norepinephrine) (78).
The TNFa may cause apoptosis which is characterized by fragmentation of DNA (79).
Wei M and associates (79) in their study showed that the activation of TNFa and IL-6 happens to the myocardium during the cardiac surgery. After the phase of ischemia-reperfusion myocardium produces soluble receptors TNFa (sTNFR-I and sTNFR-II) that compete with their membrane receptors in binding of free cytokines, acting as inhibitors of these limiting their action at the local level.
Adhesion molecules
The vascular integrins and selectins recognized as families specific receptors in leukocyte adhesion to endothelial cells. Integrins are transmembrane glycoproteins consisting of large and smaller chains. Studies have expressed the view that leukocyte adhesion to endothelial and epithelial cells becomes during neutrophil migration It must (80,81) be noted that the mechanism in neutrophil migration seems not to be released by a special stimulator and also differs in the pulmonary from the systemic circulation.
E-selectin και P-selectin
Selectins are adhesion molecules and characterized according to the carrier in which developed as E-selectin for these identified endothelial cells, as L-selectin for these identified on leukocytes and P-selectin on these identified in platelets, although the latter are detected and endothelial cells. The E-selectin is an adhesion molecule soluble and present in a variety of acute and chronic inflammatory conditions (82,83). Activation of the endothelium by cytokines or lip polysaccharide, promotes the expression of E-selectin which was followed by the ICAMs.
Expression of selectins in microcirculation varies. For example, E-selectin has a maximum concentration of 4 hours after stimulation as opposed to the vascular ICAM-1 which has a maximum concentration 24 hours after this (84,85). It is therefore obvious at least for coronary circulation that periods of hypoxia, hypothermia and activation of cytokines during and after the EP can increase local expression of selectins.
The L-selectin is a structural component of the cell membrane . Interacts with P-selectin to mediate the adhesion and “rolling” of leukocytes along the endothelium. The shear stress activates the P-selectin and forces neutrophils to develop attachments to the endothelium, resulting in the interruption of their movement and performing dialysis. The fact that L-selectin is usually expressed on the surface of cellulosic resources even at rest, may explain why no studies in humans have shown increased expression during or after the period of EC.
Oxidants
The activation of leukocytes, monocytes and macrophages results in the release of large amounts of oxygen free radicals, which act on the cytotoxic phagosomes but additionally consist cytotoxic mediators in acute inflammation in the environment outside the cells. In creating oxidants there are four enzymes: nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, superoxide dismutase, NO synthase and myeloperoxidase which produce four main products: O2-, H2O2, NO and HOCl and which create all active oxidants (86).
Of these products, NO is a direct influential factor cell death because inactivates the enzymes of glycolysis, the Krebs cycle and the electron transport chain, as well as reduces the intracellular adenosine triphosphate.
The other three products act on the membrane lipids, increasing the permeability and cause cell swelling with the effect of reducing the cardiac and pulmonary function. It is proved that CPB promotes the production of free radicals and other tissues outside the myocardium. Also other factors, such as temperature, can affect the production of free radicals. It has been found that maintaining the lung at 100 °C and 100% O2 inhalation causes more damage from oxygen free radicals in relation to temperature and 10 °C lungs inhaling room air (5).
Endotoxins
They are potent activators of the inflammatory cascade, because activate complement, neutrophils, monocytes, and other leukocytes. During and after the EC, circulating levels of endotoxin increase (87-90). Splanchnic vasoconstriction during EC may cause ischemia of the intestinal mucosa and producing bacteria, which ultimately lead to increased permeability and release of large amounts of endotoxin in circulation. Some authors have revealed a positive correlation between increasing endotoxin and duration of the EP as time of AOC, which is disputed by other authors.
There is a hypothesis that endotoxin levels after the EC related mainly to vasoconstriction over time AOC and phase of EC. Important is the finding that the pulsatory flow during the EC causes less increase in endotoxins from the continuous flow and therefore may reduce the intestinal ischemia (5).
Metalloproteinase
They are proteolytic enzymes with predominant effect on the degeneration of collagen and proteins in the tissue of epithelium and the basal membrane of blood vessels, contributing to the pathogenesis of atherosclerosis and remodelling of the left ventricle.
EC promotes the synthesis and release of metalloproteinase (matrix metalloproteinase) (91). It has been found hat collagenases MMP-8 and MMP-13 quadrupled at the end of the EC, pro-MMP-9 triples after the lifting of the blockade of the aorta and remains with the pro-MMP-2 at high levels for 24 hours after the end of the EC.
Angry blood
The blood during EC contains vocative agents, cytotoxic agents, activated blood cells and micro emboli. All these pro-inflammatory factors activate endothelial cells which allow the fluid to exit the extra vascular space (92).
Result of these mechanisms is the clinical appearance of inflammatory reaction, characterized by malaise, fever, frequency pulse, slight hypotension, fluid accumulation in the interstitium, temporary malfunction of organs particularly the brain, heart, lung and kidney.
Measures to avoid or reduce release of inflammatory reaction
Heart surgery without extracorporeal circulation
Gormley SMC and associates (93) in their study on the levels of cytokines in plasma and urine in two groups of patients who underwent heart surgery with or without EC found no statistically significant changes in the concentrations of cytokines. Early lesions in renal tubules were observed in both groups of patients, but the patients of group who were operated without EC lesions were shorter period of time. The authors argue that the variations in the concentrations of cytokines and early renal injury are not specific parameter indicative of the detrimental effect of extracorporeal circulation, but the EC causes elongation of the inflammatory response and its mediators in renal failure. Brasil LA and associates (78) studied the release of TNF-a in two groups of patients who underwent heart surgery with or without EC and found that this factor was detected only in patients operated with CE.
Interesting was the study of Song Wan et al. (94) who determined the levels of IL-8 and IL-10 in patients undergoing coronary artery bypass without the use of extracorporeal machine (with a beating heart). The authors found low levels of interleukins, suggesting reduced release of cytokines and consequently less extensive myocardial injury.
Elevated levels of inflammatory mediators, increased complement activation and adhesion molecules observed during cardiovascular surgery (95). Nevertheless, the complement activation and consumption of ICAM-1 is more evident in the group of patients operated on EC, but are not exclusive of this finding.
Regardless of the aforementioned parameters, patients with advanced age and patients with renal or pulmonary dysfunction have lower morbidity and mortality when operated on without EC, probably due to reduced body’s response to the impact of the EC (96-98).
Extracorporeal circulation without interference of oxygenator
In a prospective randomized trial of Josef AR and associates the technique of Drew-Anderson was used. It was observed a significantly reduced inflammatory response of the body (reduced levels of IL-6 & IL-8), while patients were faster postoperative recovery.
The coronary surgery with the off-pump technique s a reliable alternative to the classical technique (using EC). Associated with decreased levels of cytokines and in particular IL-8 (94,99) resulting in decreased activation of leukocytes/epithelial cells and consequently the extent of postoperative myocardial damage, as monitored by levels of troponin-I. The minimization of cell and activation of complement activation significantly limit the extent of postoperative inflammation in coronary surgery with off-pump technique.
Temperature circulating blood during EC
Studies in patients with coronary artery disease (94), of low risk showed that intermittent normothermic cardio protection against ischemic period of the heart causes less extensive myocardial damage than the cold cardioplegic protection.
Grunenfelder J and colleagues in their study (100) found increased levels of adhesion molecules and cytokines after EC. Particularly significant it has been the increase in the levels of sICAM-1, sE-selectin, IL-6 and IL-8 when systemic hypothermia was applied. Elevated levels were associated with prolonged blocking (occlusion) of the aorta and perfusion, but nevertheless did not affect the postoperative course of patients. Menasché et al. (101) argued that in patients where profound systemic hypothermia is applicable levels of inflammatory mediators increase during the reheating of the patient.
Heparinised extracorporeal circulation circuit (or circuits coated with heparin)
From studies found that the heparinised EC circuits reduce the concentration of complement (102-105) and production of TNF (106) without better clinical picture. The search of more biocompatible materials continues and new synthetic materials are used with encouraging results in the clinical experimental level (107,108). Generally heparinised circuits (oxygenator tubing) are in widespread clinical application.
Modified ultra filtration
Grunenfeldera et al. attempted after the EP the application of modified ultra filtration specific interpolation filter (Gambro FH22, membrane surface 0.2 m2, pore size 5 nm) between inter-arterial venous circuit EC. This technique was applied in two groups of patients: (I) in patients undergoing hypothermic EC and (II) in patients with normothermic EC. The levels of adhesion molecules and cytokines were determined and the following were found: (I) found higher levels of these indicators in patients operated with hypothermic EC than in those with normothermic; (II) the modified ultra filtration proved more beneficial to the group of patients in the hypothermia by reducing the levels of cytokines and adhesion molecules by 60% and 37% respectively, after 24 hours from the end of the EC. Nevertheless, the difference between the ultra filtration of both groups was not statistically significant, and this was attributed to the fact that although hypothermia produced larger amounts of cytokines and adhesion molecules (than in normothermia), but they are removed by ultra filtration and larger amounts of the substances given time. However, the modified ultra filtration did not receive widespread clinical application.
Removal of leukocytes using specific filters
Despite the initial enthusiasm of using special filters to trap white blood cells, in order to limit or prevent the inflammatory response of the organism however, most clinical studies failed to show a significant reduction in the activation of leukocytes or equivalent clinical benefit (109,110). Today these filters have found wide clinical application.
Inhibitors of complement
C1-inhibitor is found inside the body and it is the natural inhibitor of C1, while the factors C4BP and H inhibit the complement C3 and C5, respectively. Nonetheless Sahu et al. (44) in their study indicated that none of these factors did not inhibit satisfactory supplement during extracorporeal circulation. Newer literature data indicate that genetically recombinant inhibitors like h5G1.1-scFv antibody to complement C5, the monoclonal antibody anti-factor D, and a very small synthetic peptide the compstatin, inhibit supplements C5, C3a, sC5b-9, C5a and C3, with encouraging clinical results (38,39,111).
Corticosteroids-non-steroidal anti-inflammatory
Inaba et al. (112) argue that granting methylprednisolone (30 mg/kg) prior to cardiac surgery suppressed the production of IL-6 by the EC, while the same dose in patients who underwent surgery for lung did not affect its concentration (46). Steroids can reduce complement activation improving the recovery of the myocardium (113), and preventing the occurrence of cardiac arrhythmias (114). Steroids before surgery significantly inhibits the release of toxic cytokines TNF-a, IL-6, IL-8, reduces the production of endotoxins (115), promotes the synthesis of IL-10, but has no effect on clinical recovery of patients (116). Generally, the use of steroids before surgery improves systolic and diastolic function of the heart and contributes to hemodynamic stability after the EC, as found in experimental heart transplant model in sheep (117). Also preoperative steroids contribute to the rapid recovery of paediatric and adult patients and found that it reduces the time spent in the ICU and total hospital treatment time (113).
Corticosteroids act by interfering with the synthesis mechanism of prostaglandins which are responsible for the appearance of clinical assistance. The production of the PGE1 and PGE2 manifests signs of inflammation such as redness, temperature increase due to vasodilatation and oedema due to increased capillary permeability. The PGE2 induce chemo taxis of T lymphocytes, does not cause pain before the production of histamine and bradykinin, but they enhance the intensity and duration of pain due to these factors. Pyrogens activate the biosynthesis of prostaglandins resulting in the release of PGE2 by the hypothalamus which regulates the body temperature. NSAIDs inhibit cyclooxygenase (COX) and act as antipyretics. In particular inhibitors of COX-2 attenuate the inflammatory reaction induced after cardiac surgery. The COX-2 after extracorporeal circulation is overproduced in many tissues (e.g., brain), while increasing its derivatives, thromboxanes and pressor prostaglandins, leading to coronary artery spasm and increased pulmonary vascular resistance. Inhibition of COX-2 reduces myocardial suppression of the production of endotoxin and lung damage during ischemia-reperfusion phase.
Protease inhibitors-other inhibitors
Pentoxifillini a nonspecific inhibitor of phosphodiesterase, was found to cause complete inhibition of the expression of ICAM-1 and cytokines (118), as inhibits the action of IL-1 and TNF. This mechanism limits the tissue damage caused by other inflammatory cells.
Another substance, aprotinin, protease inhibitor, inhibits extravasation of plasma and interstitial oedema formation after EC, a process which is triggered by PAF (119). The use of aprotinin during EC, in order to inhibit the production and action of kallikrein appears to be an acceptable approach to the suppression of systemic inflammatory response, as well as aprotinin reduces the activation of proteases of the plasma, which ultimately leads to the SIRS. Nevertheless, Denizot Y and colleagues (120) failed, despite high doses of granted aprotinin to inhibit the inflammatory response in patients who underwent coronary surgery. Also in a randomized study (121) involving a total of 200 patients underwent coronary artery surgery and given high doses of aprotinin, it was demonstrated decrease in plasma pro-inflammatory cytokines and activation of leukocytes.
As regards to inhibitors of phosphodiesterase, the milrinone and enoximone these have given encouraging results in suppression of the inflammatory response, causing a decrease in production of IL-6 and IL-1b (122).
Similar results were given by simvastatin a lipid lowering agent which lowers levels of LDL in plasma lipoproteins and which exhibits strong inhibitory action on release of the inflammatory response (123).
Inhibitory activity and inflammatory response presents sodium nitroprusside which through the production of NO limits the complement activity and proinflammatory cytokines, and the leukocyte-platelet aggregation (124).
Finally a plurality of antioxidants, with the major vitamin E and C, have been used to limit the harmful effects of oxygen free radicals in tissues, during extracorporeal circulation but without causing overt clinical improvement in patients (122).
Lornoxicam
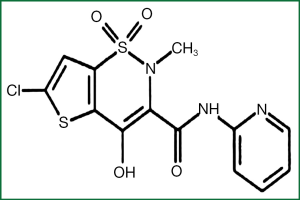
Lornoxicam (Figure 2) known as clortenoxicam is an NSAID and belongs to the category of oxicams. Its molecular weight is 371.81 and the colour yellow. Lornoxicam is a zwitterion at pH 2-5 and anion at pH ≥6.208. Lornoxicam has analgesic, antipyretic and anti-inflammatory action.
The lornoxicam inhibits the action of COX enzyme and as as result prevents the synthesis of prostaglandins, some of which have harmful effects on the body.
More specifically, the mechanism of action of lornoxicam is as follows: the linoleic acid of foods converted into phospholipids of the cell membrane, which under the influence of phospholipase A2 are converted into arachidonic acid, which essentially is the precursor of prostaglandins. Subsequently arachidonic acid undergoes oxidation and cyclization with the effect synthetase endoperoxide of prostaglandin, and the prostaglandins are produced PGG2, from which-again with the effect of the same enzyme-prostaglandins PGH2 are produced. The latter are precursors of various prostaglandins and thromboxanes
The lornoxicam acts by inhibiting the action of the enzyme COX (cyclization) and peroxidase (oxidation), an action which is characterized as “reversible” that lasts about 48 hours. Arachidonic acid follows simultaneously another metabolic road where the influence of 5-lipoxygenase neutrophil is converted into leukotrienes.
The synthesis of prostaglandins can be inhibited, except lornoxicam and other compounds, such as cortisol, aspirin and other NSAIDs.
Major interest in the mechanism of action of lornoxicam presents the participation of the enzyme COX. There are two types of cyclooxygenase: COX-1 and COX-2. The COX-1 is located in the gastric mucosa, platelets, vascular endothelium and kidney. COX-2 is produced in response to the inflammatory reaction and therefore is found in monocytes and macrophages when they are activated by the activated platelet factor (PAF), interleukin-1 or lip-polysaccharide bacteria. Also, COX-2 is located in the smooth muscle cells, epithelial and endothelial cells and neurons. Many of the non-selective NSAIDs inhibit both types of COXe, leading to undesirable side effects from the gastrointestinal tract after COX-1 is inhibited. Lornoxicam exhibits potent inhibitory activity in both isozymes.
Pharmacokinetic
Absorption
A dose of lornoxicam is rapidly absorbed from the gastro-intestinal system, with greater concentration in plasma 30 min (tmax) after dosing. It was found that, after taking tablets LNX the maximum value of concentration in plasma (Cmax) was 280 mg/L after 2.5 hours of dosing 4 mg. Ankier SI and associates peak in concentration is achieved after repeated administration of the drug. Receiving lornoxicam during meals causes decrease in absorption, as demonstrated both by the approximately 15% decrease in AUC (Area under the Curve) and secondly by increasing tmax from 1.5 to 2.3 hours. The bioavailability of lornoxicam after intramuscular administration is estimated to be 87%. Important is the fact that the bioavailability of lornoxicam shows no differences between per os administration and parenteral administration.
The lornoxicam shows strong association with plasma proteins and almost exclusively to serum albumin.
Metabolism
Lornoxicam catabolized yielding three metabolites, one of which is 5-hydroxy-lornoxicam while the other two remain unidentified metabolites in their chemical structure, but they are retained for more than 120 hours after administration of the drug as it was demonstrated by radio isotopic studies of Turner P and partners. The concentration of lornoxicam and its metabolites in plasma after a single per os administration of 4 mg, while the concentration of lornoxicam in plasma after per os or intravenous administration to a higher dose (2 mg/KVS)
The oxidative metabolism of lornoxicam is made by cytochrome CYP2C9 (member of the family of cytochrome P450) (125). From the same isozyme other substances are catalyzed such as sulphaphenazole which acts competitively in the catabolism of lornoxicam. Surprisingly high concentration values of lornoxicam with slow clearance of the drug were observed in healthy volunteers (126) and it seems that high concentrations not related to pharmacogenetics but they are caused by mechanisms which affect the metabolism of the drug. Also lornoxicam can inhibit the metabolism of itself. Finally the hydroxylated metabolite has documented “inactive” in vitro and in vivo tests.
Catharsis
It has been found that only minimum amounts of lornoxicam excreted with urine. Degradation products of lornoxicam excreted in urine (42%) and feces (51%).
The half-life (t1/2) for lornoxicam varies from 3 to 5 hours as it appears personalization. Its hydroxylated metabolite has half-life time for a single dose of 4 mg and 8.5-9 hours for one or two daily doses of parenteral administration (126). The half-life time is independent of the route of administration with similar rates between intravenous and intramuscular administration. The clearance of lornoxicam after per os administration of 8 mg twice a day lasts from 1.5 to 3.4 hours in healthy individuals (126).
Advancing age, hepatic dysfunction, chronic arthropathy and nephropathy horror dysfunction may influence the association of lornoxicam in plasma proteins and alter its pharmacokinetics and pharmacodynamics. In addition high concentrations of lornoxicam can fully commit protein binding of lornoxicam in plasma.
In elderly subjects, the clearance of lornoxicam is reduced by 30-40%, with no significant change in the pharmacokinetics of lornoxicam in patients with renal or hepatic impairment.
Clinical pharmacology
Lornoxicam has anti-inflammatory, analgesic and antipyretic activity. The anti-inflammatory action is ten times stronger than that of piroxicam and tenoxicam.
Lornoxicam proved experimentally that slight inhibitory migration of polymorph nuclear. It was also found that lornoxicam reduces human platelets, in vitro release of platelet derived growth factor (PDGF), which is associated with the production of destructive pain in rheumatoid joints.
Another effect of lornoxicam, unlike other NSAIDs, which was proved to experimentally in pigs, is the stimulation of proteoglycan synthesis, a protein associated with the creation of the cartilage. That capacity makes lornoxicam a potential agent for reducing the harmfulness of rheumatoid arthritis cartilage.
Intravenous lornoxicam was proved beneficial in patients with low back pain which fell significantly. The mechanism of action in these cases is attributed to the creation of increased levels of endogenous morphine (dynorphin and β-endorphin) of the administered substance.
The antipyretic effects of lornoxicam are less than the corresponding anti-inflammatory. Nevertheless, to achieve necessary antipyretic activity is needed tenfold dose than that of the anti-inflammatory activity.
In a recent [2004] study of Antwerpen et al. (127), was proved that oxicams in order of potency piroxicam, lornoxicam, tenoxicam, which are effective antioxidants and more effective than nimesulide ivoproufaini.
Interactions with other drugs
Lornoxicam interacts with a significant number of pharmaceutical agents and it is possible that its action will be increased or decreased depending on the class of drug. There is also a group of drugs for which we do not know whether or not the synergy of lornoxicam with them and for this reason requires continual monitoring of the patient. A classic example is the lithium, which is commonly used in psychiatric patients.
It has not been assessed with certainty the effect of lornoxicam on digoxin. Hitoglou-Makedou and colleagues in their study in healthy volunteers who received lornoxicam 4 and 0.25 mg digoxin was found to have decreased by 21% Cmax of lornoxicam in plasma, while half-life of the drug (t1/2) was prolonged by 36%. Also in the same paper the authors indicate synergy, without specifying the mechanism, with antihypertensive agents such as angiotensin converting enzyme inhibitors, beta-blockers, diuretics and vasodilators.
Lornoxicam shows synergy with pharmaceutical agents which are used to treat certain rheumatic diseases (Disease Modifying anti-Rheumatic Drugs-DMARDS), such as methotrexate, cyclosporine, sulfasalazine. Lornoxicam found that at doses of 4 or 8 mg two times pre day, administered with methotrexate at a dose 5 mg twice a day, increases the activity of methotrexate at rates ≥20%.
The antacids do not affect the pharmacokinetics of lornoxicam.
Interesting is the co administration of lornoxicam in patients who have been underwent anticoagulation with various factors, such as warfarin (warfarin, panwarfin), derivatives koumarolis (sintrom). This knowledge is particularly useful in cardiac surgery for patients who are underwent replacement of one or more heart valves with mechanical valvular prostheses and should receive lifelong anticoagulation. Some of these patients due to postoperative appearance of pericarditis or pleural effusion (syndrome after pericardiotomy) should be undergone immediate in treatment with NSAIDs.
There is considerable synergy of lornoxicam with warfarin, which increases by 32% of the average concentration of warfarin in plasma, thereby increasing the anticoagulant activity and the risk of bleeding. In another study it has been suggested that co administration of lornoxicam in therapeutic doses does not affect the anticoagulant activity of acenocoumarol. This can be explained by the fact that acenocoumarol and warfarin exhibit significant pharmacokinetic differences and are metabolized by different isozymes of cytochrome P450.
The same authors found that the co-administration of lornoxicam with phenprocoumon significantly reduced the anticoagulant action. This is caused due to reduced clearance and increased bioavailability of phenprocoumon. Based on these reports it is indicated that co administration of lornoxicam with the three aforementioned anticoagulants require close monitoring of prothrombin time and INR in these groups of patients.
Side effects
The lornoxicam, like other NSAIDs, may experience a variety of undesirable effects of the various organ systems of the body. Thus:
- by the digestive system may occur epigastric pain, anorexia, nausea, diarrhea or constipation, and rarely liver function;
- from CNS headaches, vertigo;
- from the skin rash, pruritus, and rarely photosensitivity;
- from the hematopoietic system anaemia, leucopoenia, thrombocytopenia, and;
- rarely: oedema, amblyopic and tinnitus.
Aabakken et al. (128) studied in healthy volunteers the effect of lornoxicam (8 mg × 2 daily for 7 days) and naproxen (500 mg × 2 daily for 7 days) in the gastric mucosa, using the technique for the evaluation of gastroscopy mucosal. It was found that lornoxicam caused significantly fewer erosions, bleeding and irritation of stomach mucosa so as duodenal bulb (P=0.0001) and the average duodenum (P<0.0001) compared to naproxen. The risk of side effects from the gastrointestinal tract in patients with long-term treatment appears to be greater in elderly patients or in women.
Small doses lornoxicam (4 mg × 2 daily) did not appear to adversely affect the metabolism of the liver cell, as it is seen with other NSAIDs. The total daily administration of lornoxicam in doses of 2-16 mg up to 22 days does not seem to have a nephrotoxic effect.
Regarding the effect of lornoxicam in biochemical indices of blood, the study of Ankier SI and associates in healthy volunteers showed no changes in laboratory test values while it has been of crucial importance the of reference Fenzel claimed that there was no death due to administration of lornoxicam.
There are strong indications that lornoxicam displays endogenous toxic effect on genes, although they have been caused chromosomal abnormalities in bone marrow cells in vivo, but these had occurred after maximum dosage and were attributed to the toxic effects of the drug in the bone marrow and subsequent regenerative proliferation. Regarding the participation of lornoxicam in carcinogenesis, experimental studies in mice and rats where the drug was administered for 96 and 104 weeks, respectively, found no effect on the overall incidence of tumours and grade them. Also in experimental studies in rats and rabbits, found that lornoxicam has no adverse effects on fertility of male animals, whereas in females lornoxicam affect ovulation, implantation of the egg in the uterus and prolonged pregnancy and childbirth. Although there is no evidence of teratogenicity, lornoxicam is contraindicated during pregnancy due to lack of clinical experience in humans.
Clinical applications
Lornoxicam may be used as an analgesic in gynecological surgery, providing analgesia level similar to that of paracetamol, without even having difference in the incidence of postoperative bleeding (129,130). It also causes strong analgesia in spinal surgeries (131).
Lornoxicam may be used as an analgesic to patients after cardiac surgery and even its effectiveness is similar to that of diclofenac, and it does not increase the incidence of postoperative bleeding.
A contrary view was expressed by Griffin and colleagues (132) who argue that lornoxicam and NSAIDs because are generally associated with serious side effects (hypotension, bradycardia, gastric complaints, kidney damage, nausea, vomiting, bleeding disorders influence platelet function), should not be used in cardiac surgery. Also asserted that NSAIDs should not be used postoperatively and in other surgeries because of the risk of postoperative bleeding (133).
Formulations and dosage
So far in Europe is released a formulation of lornoxicam with the trademark XEFO (NYCOMED AUSTRIA GMBH). The drug is available in the form of tablets 4 and 8 mg, as well as a solution for injection in vials 8 mg/fialidio. As excipients are used: Magnesium stearate, Polyvidone K25, Croscarmellose sodium type A, Cellulose mi-crocrystalline, Lactose monohydrate, Macrogol 6000, Titanium dioxide, Talc, Hypromellose 2910.
A new granular form has been developed to provide more rapid absorption from the gastrointestinal tract with the same bioavailability of conventional tablets (134).
The recommended initial dose per os for lornoxicam is 4 mg twice or three times a day or 8 mg twice a day for chronic arthritis. Single or repeated per os or parenteral dosages of 4 or 8 mg are suggested for postoperative pain .
Future directions
The investigation continues today on the three metabolites of lornoxicam (the hydroxylated metabolite and two other metabolites of unknown chemical composition) to search for potential new pharmacological properties and activities (134).
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Gibbon JH Jr. Application of a mechanical heart and lung apparatus to cardiac surgery. Minn Med 1954;37:171-85. [PubMed]
- Lillehei CW, Cohen M, Warden HE, et al. The results of direct vision closure of ventricular septal defects in eight patients by means of controlled cross circulation. Surg Gynecol Obstet 1955;101:446-66. [PubMed]
- Dewall RA, Gott VL, Lillehei CW, et al. A simple, expendable, artificial oxygenator for open heart surgery. Surg Clin North Am 1956;1025-34. [PubMed]
- Kirklin JW, Dushane JW, Patrick RT, et al. Intracardiac surgery with the aid of a mechanical pump-oxygenator system (gibbon type): report of eight cases. Proc Staff Meet Mayo Clin 1955;30:201-6. [PubMed]
- Wan S, LeClerc JL, Vincent JL. Inflammatory response to cardiopulmonary bypass: mechanisms involved and possible therapeutic strategies. Chest 1997;112:676-92. [PubMed]
- Royston D, Minty BD, Higenbottam TW, et al. The effect of surgery with cardiopulmonary bypass on alveolar-capillary barrier function in human beings. Ann Thorac Surg 1985;40:139-43. [PubMed]
- Banerjee R, Chakraborty H, Sarkar M. Photophysical studies of oxicam group of NSAIDs: piroxicam, meloxicam and tenoxicam. Spectrochim Acta A Mol Biomol Spectrosc 2003;59:1213-22. [PubMed]
- Ritland SR, Gendler SJ. Chemoprevention of intestinal adenomas in the ApcMin mouse by piroxicam: kinetics, strain effects and resistance to chemosuppression. Carcinogenesis 1999;20:51-8. [PubMed]
- Grossman EM, Longo WE, Panesar N, et al. The role of cyclooxygenase enzymes in the growth of human gall bladder cancer cells. Carcinogenesis 2000;21:1403-9. [PubMed]
- Goldman AP, Williams CS, Sheng H, et al. Meloxicam inhibits the growth of colorectal cancer cells. Carcinogenesis 1998;19:2195-9. [PubMed]
- Anderson R, Eftychis HA, Weiner A, et al. An in vivo and in vitro investigation of the phototoxic potential of tenoxicam, a new non-steroidal anti-inflammatory agent. Dermatologica 1987;175:229-34. [PubMed]
- Serrano G, Fortea JM, Latasa JM, et al. Photosensitivity induced by fibric acid derivatives and its relation to photocontact dermatitis to ketoprofen. J Am Acad Dermatol 1992;27:204-8. [PubMed]
- Gebhardt M, Wollina U. Cutaneous side-effects of nonsteroidal anti-inflammatory drugs (NSAID). Z Rheumatol 1995;54:405-12. [PubMed]
- Bayerl C, Pagung R, Jung EG. Meloxicam in acute UV dermatitis--a pilot study. Photodermatol Photoimmunol Photomed 1998;14:167-9. [PubMed]
- Villegas I, Martin MJ, La Casa C, et al. Effects of oxicam inhibitors of cyclooxygenase on oxidative stress generation in rat gastric mucosa. A comparative study. Free Radic Res 2002;36:769-77. [PubMed]
- Yoshikawa T, Naito Y, Kishi A, et al. Role of active oxygen, lipid peroxidation, and antioxidants in the pathogenesis of gastric mucosal injury induced by indomethacin in rats. Gut 1993;34:732-7. [PubMed]
- Tanaka J, Yuda Y. Lipid peroxidation in gastric mucosal lesions induced by indomethacin in rat. Biol Pharm Bull 1996;19:716-20. [PubMed]
- Lugardon S, Lapeyre-Mestre M, Montastruc JL. Upper gastrointestinal adverse drug reactions and cyclo-oxygenase-2 inhibitors (celecoxib and rofecoxib): a case/non-case study from the French Pharmacovigilance Database. Eur J Clin Pharmacol 2004;60:673-7. [PubMed]
- Banerjee R, Chakraborty H, Sarkar M. Host-guest complexation of oxicam NSAIDs with beta-cyclodextrin. Biopolymers 2004;75:355-65. [PubMed]
- Mitchell IM, Prabhakar G, Maughan J, et al. Pulmonary artery versus left ventricular venting: a radioisotope study of left ventricular function. Ann Thorac Surg 1989;48:699-703. [PubMed]
- Sethia B, Martin W, Wheatley DJ. The effects of left atrial and left ventricular cannulation on left ventricular function. Int J Artif Organs 1985;8:331-4. [PubMed]
- Patterson RH Jr, Twichell JB. Disposable filter for microemboli. Use in cardiopulmonary bypass and massive transfusion. JAMA 1971;215:76-80. [PubMed]
- Heimbecker R, Robert A, McKenzie FN. The extracorporeal pump filter--saint or sinner? Ann Thorac Surg 1976;21:55-8. [PubMed]
- Taylor KM, Devlin BJ, Mittra SM, et al. Assessment of cerebral damage during open-heart surgery. A new experimental model. Scand J Thorac Cardiovasc Surg 1980;14:197-203. [PubMed]
- Mahoney CB. Heparin-bonded circuits: clinical outcomes and costs. Perfusion 1998;13:192-204. [PubMed]
- Aldea GS, Soltow LO, Chandler WL, et al. Limitation of thrombin generation, platelet activation, and inflammation by elimination of cardiotomy suction in patients undergoing coronary artery bypass grafting treated with heparin-bonded circuits. J Thorac Cardiovasc Surg 2002;123:742-55. [PubMed]
- Videm V, Mollnes TE, Bergh K, et al. Heparin-coated cardiopulmonary bypass equipment. II. Mechanisms for reduced complement activation in vivo. J Thorac Cardiovasc Surg 1999;117:803-9. [PubMed]
- Brasil LA, Gomes WJ, Salomão R, et al. Inflammatory response after myocardial revascularization with or without cardiopulmonary bypass. Ann Thorac Surg 1998;66:56-9. [PubMed]
- O’Connor GT, Birkmeyer JD, Dacey LJ, et al. Results of a regional study of modes of death associated with coronary artery bypass grafting. Northern New England Cardiovascular Disease Study Group. Ann Thorac Surg 1998;66:1323-8. [PubMed]
- Liu JY, Birkmeyer NJ, Sanders JH, et al. Risks of morbidity and mortality in dialysis patients undergoing coronary artery bypass surgery. Northern New England Cardiovascular Disease Study Group. Circulation 2000;102:2973-7. [PubMed]
- Buckberg GD. Oxygenated cardioplegia: blood is a many splendored thing. Ann Thorac Surg 1990;50:175-7. [PubMed]
- Quillen J, Kofsky ER, Buckberg GD, et al. Studies of controlled reperfusion after ischemia. XXIII. Deleterious effects of simulated thrombolysis preceding simulated coronary artery bypass grafting with controlled blood cardioplegic reperfusion. J Thorac Cardiovasc Surg 1991;101:455-64. [PubMed]
- Menasche P, Grousset C, Gauduel Y, et al. Prevention of hydroxyl radical formation: a critical concept for improving cardioplegia. Protective effects of deferoxamine. Circulation 1987;76:V180-5. [PubMed]
- Rosenkranz ER, Vinten-Johansen J, Buckberg GD, et al. Benefits of normothermic induction of blood cardioplegia in energy-depleted hearts, with maintenance of arrest by multidose cold blood cardioplegic infusions. J Thorac Cardiovasc Surg 1982;84:667-77. [PubMed]
- Teoh KH, Christakis GT, Weisel RD, et al. Accelerated myocardial metabolic recovery with terminal warm blood cardioplegia. J Thorac Cardiovasc Surg 1986;91:888-95. [PubMed]
- Lichtenstein SV, Ashe KA, el Dalati H, et al. Warm heart surgery. J Thorac Cardiovasc Surg 1991;101:269-74. [PubMed]
- Walport MJ. Complement. First of two parts. N Engl J Med 2001;344:1058-66. [PubMed]
- Wachtfogel YT, Harpel PC, Edmunds LH Jr, et al. Formation of C1s-C1-inhibitor, kallikrein-C1-inhibitor, and plasmin-alpha 2-plasmin-inhibitor complexes during cardiopulmonary bypass. Blood 1989;73:468-71. [PubMed]
- Fung M, Loubser PG, Undar A, et al. Inhibition of complement, neutrophil, and platelet activation by an anti-factor D monoclonal antibody in simulated cardiopulmonary bypass circuits. J Thorac Cardiovasc Surg 2001;122:113-22. [PubMed]
- Moat NE, Shore DF, Evans TW. Organ dysfunction and cardiopulmonary bypass: the role of complement and complement regulatory proteins. Eur J Cardiothorac Surg 1993;7:563-73. [PubMed]
- Kirklin JK, Chenoweth DE, Naftel DC, et al. Effects of protamine administration after cardiopulmonary bypass on complement, blood elements, and the hemodynamic state. Ann Thorac Surg 1986;41:193-9. [PubMed]
- van Oeveren W, Kazatchkine MD, Descamps-Latscha B, et al. Deleterious effects of cardiopulmonary bypass. A prospective study of bubble versus membrane oxygenation. J Thorac Cardiovasc Surg 1985;89:888-99. [PubMed]
- Cavarocchi NC, Schaff HV, Orszulak TA, et al. Evidence for complement activation by protamine-heparin interaction after cardiopulmonary bypass. Surgery 1985;98:525-31. [PubMed]
- Sahu A, Lambris JD. Complement inhibitors: a resurgent concept in anti-inflammatory therapeutics. Immunopharmacology 2000;49:133-48. [PubMed]
- Chenoweth DE, Cooper SW, Hugli TE, et al. Complement activation during cardiopulmonary bypass: evidence for generation of C3a and C5a anaphylatoxins. N Engl J Med 1981;304:497-503. [PubMed]
- Kirklin JK, Westaby S, Blackstone EH, et al. Complement and the damaging effects of cardiopulmonary bypass. J Thorac Cardiovasc Surg 1983;86:845-57. [PubMed]
- Tennenberg SD, Clardy CW, Bailey WW, et al. Complement activation and lung permeability during cardiopulmonary bypass. Ann Thorac Surg 1990;50:597-601. [PubMed]
- Cugno M, Nussberger J, Biglioli P, et al. Cardiopulmonary bypass increases plasma bradykinin concentrations. Immunopharmacology 1999;43:145-7. [PubMed]
- Brown GM, Drost E, Selby C, et al. Neutrophil sequestration in rat lungs. Ann N Y Acad Sci 1991;624:316-7. [PubMed]
- Markos J, Hooper RO, Kavanagh-Gray D, et al. Effect of raised alveolar pressure on leukocyte retention in the human lung. J Appl Physiol (1985) 1990;69:214-21. [PubMed]
- Gebb SA, Graham JA, Hanger CC, et al. Sites of leukocyte sequestration in the pulmonary microcirculation. J Appl Physiol (1985) 1995;79:493-7. [PubMed]
- Warshawski FJ, Sibbald WJ, Driedger AA, et al. Abnormal neutrophil-pulmonary interaction in the adult respiratory distress syndrome. Qualitative and quantitative assessment of pulmonary neutrophil kinetics in humans with in vivo 111indium neutrophil scintigraphy. Am Rev Respir Dis 1986;133:797-804. [PubMed]
- Ekeström S, Koul BL, Sonnenfeld T. Decreased red cell deformability following open-heart surgery. Scand J Thorac Cardiovasc Surg 1983;17:41-4. [PubMed]
- Stoll G, Jander S, Schroeter M. Inflammation and glial responses in ischemic brain lesions. Prog Neurobiol 1998;56:149-71. [PubMed]
- Naldini A, Borrelli E, Cesari S, et al. In vitro cytokine production and T-cell proliferation in patients undergoing cardiopulmonary by-pass. Cytokine 1995;7:165-70. [PubMed]
- Thomason JJ, Douglas JE, Sears W. Morphology of the laminar junction in relation to the shape of the hoof capsule and distal phalanx in adult horses (Equus caballus). Cells Tissues Organs 2001;168:295-311. [PubMed]
- Naldini A, Sower L, Bocci V, et al. Thrombin receptor expression and responsiveness of human monocytic cells to thrombin is linked to interferon-induced cellular differentiation. J Cell Physiol 1998;177:76-84. [PubMed]
- Tocci MJ. Structure and function of interleukin-1 beta converting enzyme. Vitam Horm 1997;53:27-63. [PubMed]
- Vane JR, Anggård EE, Botting RM. Regulatory functions of the vascular endothelium. N Engl J Med 1990;323:27-36. [PubMed]
- Rich EA, Panuska JR, Wallis RS, et al. Dyscoordinate expression of tumor necrosis factor-alpha by human blood monocytes and alveolar macrophages. Am Rev Respir Dis 1989;139:1010-6. [PubMed]
- Jacobs RF, Tabor DR, Burks AW, et al. Elevated interleukin-1 release by human alveolar macrophages during the adult respiratory distress syndrome. Am Rev Respir Dis 1989;140:1686-92. [PubMed]
- Butler J, Chong GL, Baigrie RJ, et al. Cytokine responses to cardiopulmonary bypass with membrane and bubble oxygenation. Ann Thorac Surg 1992;53:833-8. [PubMed]
- Cavaillon JM, Munoz C, Fitting C, et al. Circulating cytokines: the tip of the iceberg? Circ Shock 1992;38:145-52. [PubMed]
- Steinberg JB, Kapelanski DP, Olson JD, et al. Cytokine and complement levels in patients undergoing cardiopulmonary bypass. J Thorac Cardiovasc Surg 1993;106:1008-16. [PubMed]
- Jorens PG, Van Damme J, De Backer W, et al. Interleukin 8 (IL-8) in the bronchoalveolar lavage fluid from patients with the adult respiratory distress syndrome (ARDS) and patients at risk for ARDS. Cytokine 1992;4:592-7. [PubMed]
- Seitz M, Loetscher P, Dewald B, et al. Interleukin-10 differentially regulates cytokine inhibitor and chemokine release from blood mononuclear cells and fibroblasts. Eur J Immunol 1995;25:1129-32. [PubMed]
- Minty A, Ferrara P, Caput D. Interleukin-13 effects on activated monocytes lead to novel cytokine secretion profiles intermediate between those induced by interleukin-10 and by interferon-gamma. Eur Cytokine Netw 1997;8:189-201. [PubMed]
- Wan S, DeSmet JM, Barvais L, et al. Myocardium is a major source of proinflammatory cytokines in patients undergoing cardiopulmonary bypass. J Thorac Cardiovasc Surg 1996;112:806-11. [PubMed]
- Kalfin RE, Engelman RM, Rousou JA, et al. Induction of interleukin-8 expression during cardiopulmonary bypass. Circulation 1993;88:II401-6. [PubMed]
- Mojcik CF, Levy JH. Aprotinin and the systemic inflammatory response after cardiopulmonary bypass. Ann Thorac Surg 2001;71:745-54. [PubMed]
- McBride WT, Armstrong MA, Gilliland H, et al. The balance of pro and anti-inflammatory cytokines in plasma and bronchoalveolar lavage (BAL) at paediatric cardiac surgery. Cytokine 1996;8:724-9. [PubMed]
- Frering B, Philip I, Dehoux M, et al. Circulating cytokines in patients undergoing normothermic cardiopulmonary bypass. J Thorac Cardiovasc Surg 1994;108:636-41. [PubMed]
- Finn A, Naik S, Klein N, et al. Interleukin-8 release and neutrophil degranulation after pediatric cardiopulmonary bypass. J Thorac Cardiovasc Surg 1993;105:234-41. [PubMed]
- Jorens PG, De Jongh R, De Backer W, et al. Interleukin-8 production in patients undergoing cardiopulmonary bypass. The influence of pretreatment with methylprednisolone. Am Rev Respir Dis 1993;148:890-5. [PubMed]
- Beutler B, Cerami A. Cachectin: more than a tumor necrosis factor. N Engl J Med 1987;316:379-85. [PubMed]
- Gomes WJ, Carvalho AC, Palma JH, et al. Vasoplegic syndrome: a new dilemma. Rev Assoc Med Bras 1994;40:304. [PubMed]
- Speziale G, Ruvolo G, Marino B. A role for nitric oxide in the vasoplegic syndrome. J Cardiovasc Surg (Torino) 1996;37:301-3. [PubMed]
- Brasil LA, Gomes WJ, Salomão R, et al. Inflammatory response after myocardial revascularization with or without cardiopulmonary bypass. Ann Thorac Surg 1998;66:56-9. [PubMed]
- Wei M, Kuukasjärvi P, Laurikka J, et al. Inflammatory cytokines and soluble receptors after coronary artery bypass grafting. Cytokine 2001;15:223-8. [PubMed]
- Albelda SM, Buck CA. Integrins and other cell adhesion molecules. FASEB J 1990;4:2868-80. [PubMed]
- Albelda SM. Endothelial and epithelial cell adhesion molecules. Am J Respir Cell Mol Biol 1991;4:195-203. [PubMed]
- Lefer AM, Weyrich AS, Buerke M. Role of selectins, a new family of adhesion molecules, in ischaemia-reperfusion injury. Cardiovasc Res 1994;28:289-94. [PubMed]
- Kalawski R, Bugajski P, Smielecki J, et al. Soluble adhesion molecules in reperfusion during coronary bypass grafting. Eur J Cardiothorac Surg 1998;14:290-5. [PubMed]
- Bevilacqua MP, Stengelin S, Gimbrone MA Jr, et al. Endothelial leukocyte adhesion molecule 1: an inducible receptor for neutrophils related to complement regulatory proteins and lectins. Science 1989;243:1160-5. [PubMed]
- Dustin ML, Rothlein R, Bhan AK, et al. Induction by IL 1 and interferon-gamma: tissue distribution, biochemistry, and function of a natural adherence molecule (ICAM-1). J Immunol 1986;137:245-54. [PubMed]
- Babior BM. Phagocytes and oxidative stress. Am J Med 2000;109:33-44. [PubMed]
- Taggart DP, Sundaram S, McCartney C, et al. Endotoxemia, complement, and white blood cell activation in cardiac surgery: a randomized trial of laxatives and pulsatile perfusion. Ann Thorac Surg 1994;57:376-82. [PubMed]
- Andersen LW, Baek L, Degn H, et al. Presence of circulating endotoxins during cardiac operations. J Thorac Cardiovasc Surg 1987;93:115-9. [PubMed]
- Kharazmi A, Andersen LW, Baek L, et al. Endotoxemia and enhanced generation of oxygen radicals by neutrophils from patients undergoing cardiopulmonary bypass. J Thorac Cardiovasc Surg 1989;98:381-5. [PubMed]
- Nilsson L, Kulander L, Nyström SO, et al. Endotoxins in cardiopulmonary bypass. J Thorac Cardiovasc Surg 1990;100:777-80. [PubMed]
- Joffs C, Gunasinghe HR, Multani MM, et al. Cardiopulmonary bypass induces the synthesis and release of matrix metalloproteinases. Ann Thorac Surg 2001;71:1518-23. [PubMed]
- Pacifico AD, Digerness S, Kirklin JW. Acute alterations of body composition after open intracardiac operations. Circulation 1970;41:331-41. [PubMed]
- Gormley SM, McBride WT, Armstrong MA, et al. Plasma and urinary cytokine homeostasis and renal function during cardiac surgery without cardiopulmonary bypass. Cytokine 2002;17:61-5. [PubMed]
- Wan S, Izzat MB, Lee TW, et al. Avoiding cardiopulmonary bypass in multivessel CABG reduces cytokine response and myocardial injury. Ann Thorac Surg 1999;68:52-6; discussion 56-7. [PubMed]
- Tárnok A, Hambsch J, Emmrich F, et al. Complement activation, cytokines, and adhesion molecules in children undergoing cardiac surgery with or without cardiopulmonary bypass. Pediatr Cardiol 1999;20:113-25. [PubMed]
- Hirose H, Amano A, Takahashi A. Off-pump coronary artery bypass grafting for elderly patients. Ann Thorac Surg 2001;72:2013-9. [PubMed]
- Ascione R, Lloyd CT, Underwood MJ, et al. On-pump versus off-pump coronary revascularization: evaluation of renal function. Ann Thorac Surg 1999;68:493-8. [PubMed]
- Cleveland JC Jr, Shroyer AL, Chen AY, et al. Off-pump coronary artery bypass grafting decreases risk-adjusted mortality and morbidity. Ann Thorac Surg 2001;72:1282-8; discussion 1288-9. [PubMed]
- Wan S, Yim AP. Cytokines in myocardial injury: impact on cardiac surgical approach. Eur J Cardiothorac Surg 1999;16 Suppl 1:S107-11. [PubMed]
- Grünenfelder J, Zund G, Schoeberlein A, et al. Expression of adhesion molecules and cytokines after coronary artery bypass grafting during normothermic and hypothermic cardiac arrest. Eur J Cardiothorac Surg 2000;17:723-8. [PubMed]
- Menasché P, Peynet J, Haeffner-Cavaillon N, et al. Influence of temperature on neutrophil trafficking during clinical cardiopulmonary bypass. Circulation 1995;92:II334-40. [PubMed]
- Wagner WR, Johnson PC, Thompson KA, et al. Heparin-coated cardiopulmonary bypass circuits: hemostatic alterations and postoperative blood loss. Ann Thorac Surg 1994;58:734-40; discussion 741. [PubMed]
- Gorman RC, Ziats N, Rao AK, et al. Surface-bound heparin fails to reduce thrombin formation during clinical cardiopulmonary bypass. J Thorac Cardiovasc Surg 1996;111:1-11; discussion 11-2. [PubMed]
- Muehrcke DD, McCarthy PM, Stewart RW, et al. Complications of extracorporeal life support systems using heparin-bound surfaces. The risk of intracardiac clot formation. J Thorac Cardiovasc Surg 1995;110:843-51. [PubMed]
- Butler J, Murithi EW, Pathi VL, et al. Duroflo II heparin bonding does not attenuate cytokine release or improve pulmonary function. Ann Thorac Surg 2002;74:139-42. [PubMed]
- Yamada H, Kudoh I, Hirose Y, et al. Heparin-coated circuits reduce the formation of TNF alpha during cardiopulmonary bypass. Acta Anaesthesiol Scand 1996;40:311-7. [PubMed]
- Rubens FD, Labow RS, Lavallée GR, et al. Hematologic evaluation of cardiopulmonary bypass circuits prepared with a novel block copolymer. Ann Thorac Surg 1999;67:689-96; discussion 696-8. [PubMed]
- Suhara H, Sawa Y, Nishimura M, et al. Efficacy of a new coating material, PMEA, for cardiopulmonary bypass circuits in a porcine model. Ann Thorac Surg 2001;71:1603-8. [PubMed]
- Hurst T, Johnson D, Cujec B, et al. Depletion of activated neutrophils by a filter during cardiac valve surgery. Can J Anaesth 1997;44:131-9. [PubMed]
- Baksaas ST, Videm V, Mollnes TE, et al. Leucocyte filtration during cardiopulmonary bypass hardly changed leucocyte counts and did not influence myeloperoxidase, complement, cytokines or platelets. Perfusion 1998;13:429-36. [PubMed]
- Fitch JC, Rollins S, Matis L, et al. Pharmacology and biological efficacy of a recombinant, humanized, single-chain antibody C5 complement inhibitor in patients undergoing coronary artery bypass graft surgery with cardiopulmonary bypass. Circulation 1999;100:2499-506. [PubMed]
- Inaba H, Kochi A, Yorozu S. Suppression by methylprednisolone of augmented plasma endotoxin-like activity and interleukin-6 during cardiopulmonary bypass. Br J Anaesth 1994;72:348-50. [PubMed]
- Engelman RM, Rousou JA, Flack JE 3rd, et al. Influence of steroids on complement and cytokine generation after cardiopulmonary bypass. Ann Thorac Surg 1995;60:801-4. [PubMed]
- Bruins P, te Velthuis H, Yazdanbakhsh AP, et al. Activation of the complement system during and after cardiopulmonary bypass surgery: postsurgery activation involves C-reactive protein and is associated with postoperative arrhythmia. Circulation 1997;96:3542-8. [PubMed]
- Wan S, LeClerc JL, Huynh CH, et al. Does steroid pretreatment increase endotoxin release during clinical cardiopulmonary bypass? J Thorac Cardiovasc Surg 1999;117:1004-8. [PubMed]
- Fillinger MP, Rassias AJ, Guyre PM, et al. Glucocorticoid effects on the inflammatory and clinical responses to cardiac surgery. J Cardiothorac Vasc Anesth 2002;16:163-9. [PubMed]
- Segel LD, Follette DM, Castellanos LM, et al. Steroid pretreatment improves graft recovery in a sheep orthotopic heart transplantation model. J Heart Lung Transplant 1997;16:371-80. [PubMed]
- Krakauer T. Pentoxifylline inhibits ICAM-1 expression and chemokine production induced by proinflammatory cytokines in human pulmonary epithelial cells. Immunopharmacology 2000;46:253-61. [PubMed]
- O’Brien JG, Battistini B, Zaharia F, et al. Effects of tranexamic acid and aprotinin, two antifibrinolytic drugs, on PAF-induced plasma extravasation in unanesthetized rats. Inflammation 2000;24:411-29. [PubMed]
- Denizot Y, Karoutsos S, Nathan N. Differential alterations in plasma colony-stimulating factor concentrations after coronary artery bypass graft surgery with extracorporeal circulation. Cytokine 2001;13:314-6. [PubMed]
- Defraigne JO, Pincemail J, Larbuisson R, et al. Cytokine release and neutrophil activation are not prevented by heparin-coated circuits and aprotinin administration. Ann Thorac Surg 2000;69:1084-91. [PubMed]
- Paparella D, Yau TM, Young E. Cardiopulmonary bypass induced inflammation: pathophysiology and treatment. An update. Eur J Cardiothorac Surg 2002;21:232-44. [PubMed]
- Weis M, Pehlivanli S, Meiser BM, et al. Simvastatin treatment is associated with improvement in coronary endothelial function and decreased cytokine activation in patients after heart transplantation. J Am Coll Cardiol 2001;38:814-8. [PubMed]
- Massoudy P, Zahler S, Freyholdt T, et al. Sodium nitroprusside in patients with compromised left ventricular function undergoing coronary bypass: reduction of cardiac proinflammatory substances. J Thorac Cardiovasc Surg 2000;119:566-74. [PubMed]
- Bonnabry P, Leemann T, Dayer P. Role of human liver microsomal CYP2C9 in the biotransformation of lornoxicam. Eur J Clin Pharmacol 1996;49:305-8. [PubMed]
- Ravic M, Salas-Herrera I, Johnston A, et al. A pharmacokinetic interaction between cimetidine or ranitidine and lornoxicam. Postgrad Med J 1993;69:865-6. [PubMed]
- Van Antwerpen P, Nève J. In vitro comparative assessment of the scavenging activity against three reactive oxygen species of non-steroidal anti-inflammatory drugs from the oxicam and sulfoanilide families. Eur J Pharmacol 2004;496:55-61. [PubMed]
- Aabakken L, Osnes M, Frenzel W. Gastrointestinal tolerability of lornoxicam compared to that of naproxen in healthy male volunteers. Aliment Pharmacol Ther 1996;10:151-6. [PubMed]
- Hein A, Norlander C, Blom L, et al. Is pain prophylaxis in minor gynaecological surgery of clinical value? a double-blind placebo controlled study of paracetamol 1 g versus lornoxicam 8 mg given orally. Ambul Surg 2001;9:91-4. [PubMed]
- Trampitsch E, Pipam W, Moertl M, et al. Preemptive randomized, double-blind study with lornoxicam in gynecological surgery. Schmerz 2003;17:4-10. [PubMed]
- Thienthong S, Jirarattanaphochai K, Krisanaprakornkit W, et al. Treatment of pain after spinal surgery in the recovery room by single dose lornoxicam: a randomized, double blind, placebo-controlled trial. J Med Assoc Thai 2004;87:650-5. [PubMed]
- Griffin MR, Yared A, Ray WA. Nonsteroidal antiinflammatory drugs and acute renal failure in elderly persons. Am J Epidemiol 2000;151:488-96. [PubMed]
- Møiniche S, Rømsing J, Dahl JB, et al. Nonsteroidal antiinflammatory drugs and the risk of operative site bleeding after tonsillectomy: a quantitative systematic review. Anesth Analg 2003;96:68-77. [PubMed]
- Warner TD, Giuliano F, Vojnovic I, et al. Nonsteroid drug selectivities for cyclo-oxygenase-1 rather than cyclo-oxygenase-2 are associated with human gastrointestinal toxicity: a full in vitro analysis. Proc Natl Acad Sci U S A 1999;96:7563-8. [PubMed]