Exploiting sensitization windows of opportunity in hyper and hypo-fractionated radiation therapy
Introduction
Approximately 60% of patients with solid tumors are treated with radiation therapy, which highlights its importance in cancer treatment. For 15% of patients radiation therapy is the only form of treatment and the remaining 45% are treated with radiation combined with chemotherapy. The latter includes breast, lung, prostate, head & neck, bladder, gynecological, pancreas, colorectal and anal cancers and brain tumors (1). The efficacy of radiation therapy, whether treated alone or in combination, can be further improved by adopting recent technological advances and biological approaches. These advances in technology include improved dose distribution with intensity modulated and image guided radiotherapy (IMRT and IGRT), dose escalation (higher dose) and dose intensification (higher and more focused dose). Biological approaches include (I) adopting time-honored, “classical” concepts such as DNA damage repair, tumor cell repopulation and cell cycle distribution; (II) exploiting tumor microenvironmental changes such as hypoxia, reoxygenation, vasculature, etc.; (III) use of different types of particles (e.g., protons and carbon ions), which may have a high-linear energy transfer for improved radiobiological effectiveness; (IV) use of altered dose and schedule such as hyper- and hypo-fractionation; and (V) use of radiation protectors and sensitizers including concurrent chemotherapy. In this paper, we define standard fractionation as conventional 1.8 to 2.2 Gy (one fraction per day, five days a week continuing for 3-7 weeks), hyperfractionation as 0.5 to 2.2 (two fractions per day, 2-5 days a week, for 2-4 weeks), and hypofractionation as doses of 3-20 Gy (one fraction a day given for 1-3 days for doses 8-20 Gy).
As with cancer treatment in general, progress in radiation therapy has been steady with much more organ preservation (e.g., head & neck cancer, anal and rectal cancer, esophageal cancer) because of (I) patient selection based on improved clinical parameters, mostly of tumor stage but some with biomarkers such as proliferation and metabolism (e.g., PET scanning); (II) modified surgical/radio-surgical approaches; and (III) use of chemo/hormonal therapy based on pathological and molecular subtype (e.g., breast cancer). Progress is likely to accelerate with the incorporation of emerging new knowledge in cancer biology including tumor classification by molecular characterization and precision medicine, i.e., providing right treatment to right patient. Key to progress relies on well done randomized clinical trials that need to be based on improved preclinical models and careful post-trial analysis because well-conceived hypotheses may not be confirmed for a variety of reasons (2).
It is always wise to exploit what can be exploited based on careful clinical observation—some of which may have been hypothesis driven but much of it may be hypothesis generating based on thorough observations and innovative analyses. Examples from clinical treatments based on so-called “classical” radiation biology includes modifying radiation dose and treatment volume based on the shape of the survival curve (alpha and beta components of the linear-quadratic curve) but it would be preferable to understand the benefits of a particular dose size at the molecular, cellular, and tissue levels. Understanding what happens in various tumor types and relevant normal tissues at the clinically relevant dose fractions of 2 Gy is important, as there are extensive historical clinical-outcome data over many decades. This may help identify targets such as radiation-induced pro-survival factors that can confer induced radiation resistance (IRR). Were those the situation, one could use a particular radiation dose window (below threshold IRR dose) and schedule it in such a way that it does not activate pro-survival events. Resistance to treatment could relate more to factors within the heterogeneous tumor microenvironment niche or to other factors that might benefit from the use of chemotherapy as part of the regimen. The first part of the review will focus on low-dose hyperfractionation (below IRR dose or HRS-inducing dose) and chemopotentiation providing evidence both at pre-clinical and clinical level. In the second part, we provide data that support the contention that high-dose radiation has the potency to induce a robust bystander effect, as well as abscopal (distant) effects (3). Since high-dose hypofractionation regimens are now commonly adopted in the clinic (such as stereotactic radiation surgery), is there a defined dose/fractionation window to exploit certain potential sensitization avenues initiated by abscopal factors that can be potentially combined with agents (including immune modulating agents) or subsequent radiotherapy?
Low-dose hyperfractionation and chemopotentiation
In the past 100 years, the biological effects of various size doses of low-LET radiation have been examined in the clinic as well as by in vitro clonogenic assay since first reported by Puck and Marcus in 1955. Radiation hormesis or an effect of radiation at very low doses which can stimulate the repair mechanisms on the cellular level and thereby potentially protect cells from future exposure, are known to be induced at 0.1 to 0.2 Gy (100 to 200 mGy) (4). There is controversy as to what is the lowest radiation dose that can produce radiation-inducible cancer however, at doses above 0.10 Gy there is a risk of radiation-induced carcinogenesis, which increases with dose (5). Generally, at doses above 1 Gy growth arrest occurs and cell killing predominates above 2 Gy. A daily dose size in the range of 2-3 Gy and multiple dose schedules had been empirically selected over the years based on both normal tissue sparing from fractionation and evidence of clinical efficacy. However, as the biological effects of dose have been examined, novel regimens are being explored.
Low dose hyper-radiosensitivity (HRS) and induced radiation resistance (IRR)
Although, there is an understanding of the mechanism of cell death by radiation at conventional doses (1.5-2.2 Gy per fraction), the mechanism of radiation effects at lower doses (<1 Gy) is still emerging (6). The initial slope of the radiation cell-survival curve (doses of 0.1-1 Gy) was presumed to be ineffective for human tumor therapy, however, with dynamic microscopic imaging to study the effects of low dose radiation on individual cells within a larger cell population, it was demonstrated that X-rays are effective at cell killing at very low doses, around 0.1 Gy, then become less effective as the dose increased with minimal effectiveness at about 0.6 Gy, and then becoming more effective again as the dose increased to 1.5 Gy and above. This phenomenon is referred to as hyper radiation sensitivity (HRS) (6,7). At doses <1 Gy, many cell lines show low dose HRS (8-10). Interestingly, the HRS is most pronounced in radio-resistant cells, defined in this case as those with mutant p53 expression (11,12). Enns et al. (13) examining the response of human A549 lung carcinoma, T98G glioma, and MCF7 breast carcinoma cell lines to gamma radiation in the dose range 0 to 2 Gy, showed marked HRS at doses below 0.5 Gy. It was further determined that low dose hypersensitivity is possibly related to p53-dependent apoptosis, as treatment of cells with Pifithrin, an inhibitor of p53 function, completely ablated HRS. Thus, the role of p53 function in HRS is still unclear and requires further investigation using p53 knockout cell lines and validation in GEMMs.
HRS is evident in murine models (14), but it appears to be an underexplored phenomenon in humans. Since development of resistance is a major cause of treatment failure, circumventing resistance by exploiting HRS would greatly benefit in the treatment of many cancer types. Further, as seen in vitro HRS does not involve activation of pro-survival pathways [found at higher doses (15)] (16), providing a mechanism to explain the efficacy of radiation at these low doses. However, as Short and Joiner have pointed out, in order to benefit from low dose-per-fraction radiation in the clinical setting, therapy needs to be extended over 7-12 weeks for sufficient total dose to be delivered. During this prolonged period of treatment, tumor proliferation can occur, which would abate the gain due to enhanced cell killing at HRS radiation doses (17). Prolonged treatment in clinic, lasting 7-12 weeks, will result in several logistic issues as well as increasing cost. Hence, it is logical to combine a radiation dose that results in HRS with chemotherapy to potentiate the effects of chemotherapy and also shorten the treatment time.
In summary, there is a functional evidence for the existence of HRS in vitro and its exploitation in the clinic can be challenging. One possibility is to benefit in the clinic from HRS is by using Low Doses Fractionated Radiation Therapy (LDFRT) as a potentiator of systemic chemotherapy that would not trigger the activation of pro-survival pathways in the tumors. Here below, we describe the preclinical evidence to this end.
HRS-inducing LDFRT as a potentiator of chemotherapy: preclinical evidence
Extensive data are available on the HRS/IRR phenomenon observed in more than 40 tumor cell lines in response to single low dose radiation (18,19). HRS occurs after fractionated low doses in in vitro (18,19). Pretreatment with paclitaxel followed by multi-fractionated low dose radiation (0.5- or 1-Gy fractions for a total dose of 2 Gy) significantly enhanced the radiosensitizing effect in both HCT-116 and HT-29 cells when compared to single fraction 2 Gy dose (12). LDFRT was found to potentiate the effects of taxanes in head and neck cancer cell lines in vitro (15,20) as well as cisplatin in lung cancer cells in vitro (21).
The molecular mechanisms underlying the process of chemopotentiation by LDFRT are shown in Table 1. In brief, there is involvement of NFκB, NF-Y, bcl-2, XIAP and MDR1 in IRR and at the same time p53, bax, and pro-apoptotic effectors such as cytochrome C seems to be involved (Figure 1). Further, in a recent meeting presentation, HDAC inhibitor SAHA (Vorinostat) was combined with LDFRT in GBM cells lines D54 and U118. Findings of this study demonstrated that LDFRT potentiated the effect of Vorinostat in p53 dependent manner with the requirement of PTEN (22). It is important to note that at doses of approximately 0.5 Gy, ATM autophosphorylation occurs in normal cells such as skin fibroblast (23) and peripheral blood lymphocytes (24) resulting in activation of DNA repair programs, but in cancer cells the dose to activate ATM pathways is >1 Gy (25). Thus, it appears that HRS is due to a lack of activation of ATM autophosphorylation pro-survival pathways (Figure 1) (modification of apoptosis, NFκB). Thus, these mechanistic data from cell culture studies indicate that chemopotentiation by LDFRT is primarily due to cell killing, thus leading to further studies in vivo.
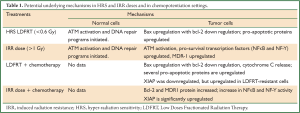
Full table
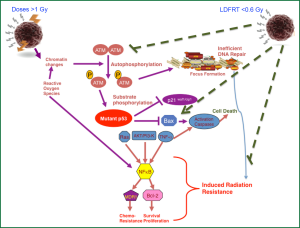
HRS inducing doses in fractionation setting were tested alone or with combination of chemotherapy in several mouse models and the results have not always been reflective of data obtained using cell cultures. For example one study, compared the effect of low dose ultra-fractionation schedule (0.4 Gy/fraction—126 fractions in six weeks; an approach to exploit the HRS) with the conventional fractionation schedule (1.68 Gy/fraction, 30 fractions in six weeks) of a total dose of 50.4 Gy for inhibiting A7 tumor growth in nude mice (26). Although, ultrafractionation resulted in a significant decrease in tumor growth delay, it also showed a significant increase of the top-up TCD50 dose (the dose needed to cure 50% of animals) compared with conventional fractionation dose, but failed to prove the existence of HRS in in vivo (26). Thus, despite a pronounced HRS phenomenon observed in vitro, ultrafractionation appeared to be significantly less effective than conventional fractionation in the above nude mice xenograft model. The results from this study simply indicate that extrapolation of such data on single dose exposure or a few fractionated doses in in vivo is not always predictive of in vitro data and does not exclude the potential clinical value (27).
Low dose fractionation allows the delivery of a higher total radiation dose to the tumor for a better result as indicated in the studies below. In a mouse glioma tumor xenograft model, repeated irradiation with low dose (0.8 Gy 3 times/day × 4 days/wk × 2 wks, total dose of 19.2 Gy) was markedly more effective compared to a conventional fractionated dose schedule (2 Gy/day × 4 days/wk × 2 wks; total dose of 16 Gy) in inhibiting tumor growth (28). Similarly, Spring et al. (29) showed that LDFRT (0.5 Gy 2 times/day × 2 days/wk × 6 wks; total dose of 12 Gy) significantly prolonged tumor re-growth delay compared to a conventional fractionation dose schedule (2 Gy one fraction/day × 1 wk × 6 wks) in a SCCHN xenograft mouse model (29).
Recently, Tyagi et al. demonstrated the capability to deliver ten 0.2 Gy pulses in 8 mins [referred to as Pulsed Low-Dose Radiation (PLRT)] (30). This approach of dose-escalated PLRT was compared with standard radiation therapy (Std-RT), where 2 Gy fractions were delivered continuously in a single fraction in eight minutes, in an intracranial U87MG GBM nude mice tumor model (31). Both PLRT and Std-RT groups received treatments for 5 days/wk. One cohort of mice was treated with 20 Gy Std-RT or 20 Gy PLRT; a second cohort was treated with 30 Gy. Results showed that the mean survival was significantly better with 34.2 days for 30 Gy PLRT compared to 29 days with Std-RT, although there was no tumor cure in either of the groups.
Even though these results imply a minimally a better outcome when radiation is used alone as LDFRT in preclinical models, because of the existence of HRS at lower radiation doses as described above, there exists potential to benefit from the effects of chemotherapy when LDFRT is used in conjunction with chemotherapy. However, demonstration of efficacy of combination of chemotherapy with LDFRT in animal model(s) optimizing dose, time, and sequence is a critical prerequisite for a successful clinical translation.
Below we discuss three such studies in which combination of LDFRT or PLRT with chemotherapy has been used that substantiate potential opportunities for enhancing chemotherapy effects for better treatment outcome. (I) Complete tumor cure was demonstrated in the studies by Spring et al. (29), that evaluated the efficacy of LDFRT in potentiating tumoricidal properties of taxotere in SCCHN tumor xenograft animal model. Tumor regression was significant in all LDFRT groups. Mechanistic studies involving molecular analyses of resected tumor specimens showed an increase in Bax levels with an increase in cytochrome c release suggesting an apoptotic mode of cell death in LDFRT chemopotentiation of taxotere effects rather than clonogenic inhibition, albeit G2M cell cycle arrest by taxotere also appears to be an important sequencing component of chemopotentiation. (II) PLRT in combination with Temozolamide (TMZ) was more effective in reducing tumor volume and normal tissue damage and improving survival compared to standard fractionation RT with TMZ in an orthotopic GBM xenograft murine model (32). Increased and differential vascularization and significantly fewer degenerating neurons were seen in normal brain after PLRT with TMZ compared to standard RT with TMZ. (III) Similarly, in an on-going study in a mouse ovarian cancer model, combination of LDFRT with paclitaxel showed significantly improved survival over paclitaxel alone or LDRFT alone. A similar trend was noted when cisplatin was combined with LDFRT in the treatment of ovarian cancer (33) as well as when TMZ was used with LDFRT in the treatment of GBM in mouse models (unpublished observations).
The above preclinical in vivo studies assessing the benefit of combining LDFRT or PLRT with chemotherapy demonstrating improved efficacy and survival as well as reducing normal tissue toxicity together with supporting mechanistic evidences provided adequate rationale for conducting safety and efficacy trials in the clinic as these studies might unlock novel treatment avenues for radio-resistant and/or aggressive tumors with poor clinical outcome (e.g., GBM and ovarian cancers). LDFRT can be exploited to potentiate the effect of chemotherapy for achieving maximum tumor cell killing with significantly reduced toxicity and a favorable clinical translation of the HRS phenomenon observed at low radiation doses to help overcome IRR at radiation doses above 0.6 Gy seen in standard fractionated chemo-radiotherapy regimen. In summary, there is strong pre-clinical evidence and mechanistic reasoning for using HRS low-doses of radiation to potentiate the effects of chemotherapy particularly in hyperfractionated settings.
HRS-inducing LDFRT as a potentiator of chemotherapy: clinical evidence
Several clinical trials have been conducted to assess the benefit of combining LDFRT with standard chemotherapeutic agents for improved outcome (Table 2). Arnold et al. (34) studied LDFRT as a chemopotentiator of paclitaxel and carboplatin in 40 patients with locally advanced SCCHN. LDFRT was given in two doses of 0.80 Gy (based on the average dose that yielded maximal HRS in four SCCHN cell lines each on days 1 and 2, administered 4-6 hours apart, and the sequence was repeated on days 22 and 23. Definitive RT began three weeks after the last dose of chemotherapy and LDFRT. The combinations of LDFRT, carboplatin and paclitaxel were extremely well-tolerated, with toxicity comparable to that of carboplatin and paclitaxel alone in a similar patient cohort.
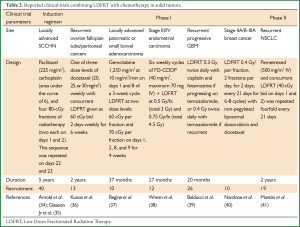
Full table
Recently, the Arnold group reported 5-year results of the above prospective Phase II SCCHN trial (35). After a median follow-up of 83 months, LRC was 80% and distant control was 77%. Out of 39 evaluable patients, 5-year OS, diseases specific survival (DSS), and PFS were 62%, 66%, and 58%, respectively. These data strongly indicate a favorable outcome compared to historical controls and excellent compliance with definitive therapy.
In the above trial, the status of p16 was evaluated, which is a validated marker for HPV status and an important predictor of response to various treatment modalities for SCCHN (42). Immunohistochemistry analysis of available 42 pre-treatment specimens showed 15 HPV positive (ten were oropharynx sub group) and 27 (seven were oropharynx subgroup) were negative. Of 15 patients with p16 positive tumors CR, PR, SD and SD were 5 (33.3%), 8 (53.3%) 1 (6.7%), and 1 (6.7%) respectively, compared to 2 (7.4%), 18 (66.7%), 6 (22.2%) and no PD among 27 patients with p16 negative tumors (P=0.0616), respectively. Similar results were also found in HPV positive oropharynx sub-group. Two-year OS was 93.3% for p16 positive patients compared to 73.08% in p16 negative patients (P=0.0252); two-year PFS was 80% (p16 + ve) and 69.23% (p16 – ve). In oropharyngeal subgroup, the 2-year OS was 100% (p16 + ve) and 42.86% (p16 – ve) tumors respectively (P=0.001). These results stress the point that p16 status can be an important predictor of response to LDFRT mediated chemopotentiation induction treatment similar to that seen in standard of care, in head and neck cancer treatment an observation recently described (43,44).
Based on the pre-clinical data (33), the Gynecology Oncology Group (GOG) conducted a feasibility study (36), of whole abdomen LDFRT for patients with recurrent epithelial ovarian fallopian tube, or peritoneal cancers along with weekly treatment of docetaxel 25 mg/m2. LDFRT was delivered in 60 cGy fractions, twice daily for two days, with a minimum of 4 hr inter-fraction interval, starting on day 1 of each chemotherapy cycle. Three out of four patients completed therapy and none of the toxicities were dose limiting. Another phase I study (38), delivering once a week for six consecutive weeks of morning cisplatin followed 6-8 hours later by afternoon low dose-whole abdomen radiation therapy (LD-WART), enrolled 12 patients with optimally debulked Stage III/IV endometrial cancer. The results suggested feasibility of using LD-WART as a novel chemopotentiator to cisplatin in combination therapy as an adjuvant regimen (38). This trial showed no dose-limiting toxicities with follow-up that ranged from 4-36 months (median: 14 months). These data as well as the data from the GOG trial does indicate that 0.60 Gy/fraction was well tolerated.
Regine et al. (37) studied upper abdominal LDFRT given as a chemopotentiator for gemcitabine in patients with locally advanced pancreatic or small bowel adenocarcinoma. Gemcitabine was given at 1,250 mg/m2 at 10 mg/m2/min on days 1 and 8 of a 3-week cycle. Low-dose fractionated radiotherapy was tested at two dose levels: 0.6 Gy and 0.7 Gy/fraction. Radiotherapy was given b.i.d. on days 1, 2, 8, and 9. Two of the four patients at dose level 0.7 Gy/fraction experienced dose-limiting toxicity, therefore 0.6 Gy/fraction was deemed the MTD.
Balducci et al. (39) reported a study of LDFRT and chemotherapy for recurrent or progressive GBM in 17 patients who had previously received radiotheraqpy and recurred: they received total LDFRT dose of 7.2 Gy in 0.3 Gy fractions with concomitant chemotherapy (TMZ and Fotemustine). LDFRT regimen was well tolerated. In reality, a robust randomized clinical is warranted to establish as a new treatment modality for GBM patients with poor prognosis.
In recurrent NSCLC, Mantini et al. (41) found that LDFRT was safe when added to 500 mg/m2 Pemetrexed as a 10-minute intravenous infusion on day 1 of a 21-day cycle, concurrent with LDFRT on days 1 and 2 at 0.4 Gy twice daily with each faction given 5-6 hrs apart, and the median total dose was 6.40 Gy. LDFRT was also tested in combination with liposomal doxorubicin and docetaxel in stage IIA/B-IIIA breast cancer that led to higher histological response rates compared to the sequential application of the same two drugs (40).
There are three more clinical trials ongoing (http://www.clinicaltrials.gov), which are summarized in Table 3. Unfortunately, as with the trials discussed above, none of them is randomized for evaluating the efficacy of LDFRT using robust end-points such as survival or quality of life.
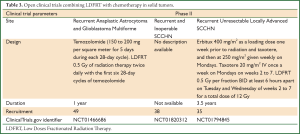
Full table
Summary of hyperfractionation
- Over the years clear evidence has emerged from the cell culture studies on the existence of HRS and IRR phenomena that have provided adequate mechanistic rationale for using radiation dose in the HRS range to potentiate the effects of chemotherapy.
- Preclinical in vivo animal studies using mouse xenograft tumor models, as discussed above, assessing the benefit of combining LDFRT or PLRT with chemotherapy demonstrate improved efficacy and survival as well as a reduction in normal tissue toxicity and have helped optimize dose, time, and sequence schedule in experimental setting and lead to clinical trials.
- Several Phase I/II clinical trials conducted in different cancer organ sites, such as SCCHN, GBM, ovarian, pancreatic, breast and lung cancers, are in process for an optimized LDFRT dose and schedule in order to potentiate the effects of chemotherapeutic drugs such as cisplatin, taxanes, TMZ, and also demonstrated improved efficacy.
- More randomized clinical trials are warranted to study the role of LDFRT as an adjuvant for chemotherapy in definite settings rather than induction regimen.
In conclusion, LDFRT has some very intriguing preclinical data, however, despite the fact that about ten clinical trials have been or are being performed, at present, it can be concluded that this technique appears to be relatively safe. Based on the reported as well as on-going clinical trials, it still remains unclear whether the patients can be benefited from the addition of LDFRT to chemotherapy and hence better designed prospective trials (randomized against chemotherapy-only controls, and with more robust endpoints such as survival and quality of life) must be conducted to ascertain the value of LDFRT in the management of solid tumors.
Hypofractionation: novel windows of opportunity
To take advantage of the technological ability to deliver precision radiation therapy and to utilize the biological effects of a large dose per fraction as well as the smaller dose per fraction just described, hypofractionated radiation therapy can provide a different pathway of biological effects either used alone or combined with chemoradiotherapy. A potential advantage of hypofractionated radiation therapy, which makes it an attractive approach for the management of advanced cancers, is the reduction in treatment time and cost and reduced burden of frequent and numerous radiotherapy sessions.
Hypofractionated radiation therapy can be approached in two different ways: (I) is to consider α/β ratio and Biologically Effective Dose (BED), where the “classical” concepts of repair, re-assortment, re-oxygenation and re-population (4-Rs) are applicable. This is a categorical approach for hypofractionated radiotherapy that uses 3 to 6 Gy dose fractions; (II) Hypofractionation schedule that uses above 8 Gy doses/fraction in radiotherapy, in which the biological changes different than the “classical” 4-Rs are felt to be applicable, generally known as high-dose hypofractionation radiation therapy (HDHRT). This section of the review will focus HDHRT with more detailed understanding of new radiobiology.
There are data to suggest that the use of HDHRT radiation is effective as an alternative means of dose escalation with conventional fractionation treatment schedule. The results with HDHRT in the early-stage lung cancer population have thus far been very encouraging with local control rates up to 90% (45,46), being superior to the control rates obtained with conventionally fractionated radiation. Biologically, new mechanistic insights suggest that HDHRT may cause four unique effects that can be further exploited for sensitization. HDHRT can (I) cause non-targeted pharmacodynamics effects (such as intra-tumoral bystander as well as abscopal effects) mediated by TNF-α, TRAIL, PAR-4 and ceramide (47-49); (II) robustly induce tumor endothelial death at doses above 8-11 Gy (50); (III) increase host immune recognition of radiation-induced enhanced antigen presentation, such that a single fraction may incite an immune response that enhances the effects of radiation (51); and (IV) result in a better response of that tumors that are heterogeneous with different cell populations, whose clonal radiosensitivity considerably differ (52).
The interaction between HDHRT and hypoxia needs to more fully understood. The effects would depend in part on the initial hypoxic fraction, the dose size used and fractionation, as reoxygenation could occur. Brown et al. (53), Song et al. (54), suggest the need for drugs to treat the hypoxic fraction whereas Meyer et al. (55) suggest that reoxygenation and the selection of a dose at the “hypoxia transition zone” could overcome hypoxia. With other potential mechanism of action of HDHRT, as noted above, studies that determine changes in hypoxia including imaging and biomarkers of hypoxia, as well as studies to modify hypoxia and or use cytotoxic agents would be needed to dissect out the complexity of the effect of hypoxia. Another interesting consideration could be the use of conventional radiation therapy following single high dose or high dose in combination with chemotherapeutic drugs to improve the response of tumors to treatment. There are strong biological data to suggest that a large induction dose of radiation preceding conventional fractionated radiation therapy results in significantly greater tumor regression (56,57). However, high doses of radiation prescribed uniformly to large tumor volumes are generally associated with significant side effects and potentially serious late toxicity, which can take many years to be manifest. At this point in time, there is limited use of high-dose-per-fraction radiation to smaller targets, as in the case of SABR for T1-2N0 lung cancer. In patients with stage III lung cancer, high-dose-per-fraction radiation to the entire target volume is precluded due to normal tissue tolerance. Therefore, future approaches could combine the capability of new imaging and treatment technology for target selection, including novel approaches described next, including HDHRT and its biological properties.
Technical aspects of hypofractionated radiation therapy
The challenges of hypofractionated radiotherapy for better treatment outcome primarily include development of optimal radiation dose delivery techniques. We provide a very brief account of technical development of SRS, SBRT and 3D lattice radiotherapy (LRT), with the understanding that high-dose rate brachytherapy with radionuclides or miniature X-ray source can also be an effective way of delivering highly localized radiation.
Traditionally SRS refers to single fraction stereotactic delivery of an intended ablative dose (58). The first full-scale successful radiosurgery system, Leksell Gamma Knife, was developed in the late 1960s. Since then its successful clinical utilization has established the foundations for intracranial radiosurgery and radiosurgery, in general. Following its success, a number of LINAC-based systems were developed since 1980s (59) and protons beams are also being used (60).
The concept of intracranial radiosurgery was first applied to other body sites in the early 1990s using modified conventional LINACs. The introduction of dedicated radiosurgery systems has widened the application, most noticeably from the early 2000s and clinical efficacy has been well demonstrated (61). In current terminology, SBRT refers to stereotactic body radiation treatments delivered in more than one fraction. While the term SBRT has been widely adopted, it is noteworthy that the difference between radiotherapy and radiosurgery is in the fractional-dose size that ostensibly leads to their differences in therapeutic effects—as a result of different radiobiological effects. The term stereotactic only indicates the method of target localization.
The goal of SABR is to administer a markedly higher dose to the treatment target volume without damaging the surrounding normal tissue thereby achieving enhanced local control and less normal tissue toxicity compared to conventional radiotherapy. The unique physical characteristics of traditional SRS are: high precision (sub-millimeter), highly-focused dose distribution (about a 10% dose fall-off per millimeter outside the treatment margin) and high dose (10 Gy and higher) (58).
In traditional SRS or SBRT, the coverage of prescribed dose to the treatment target volume is to be maximized. In contrast, the spatially fractionated high dose radiation therapy delivered in forms of spatially fractionated GRID radiotherapy (SFGRT) technique covers only partial tumor volume with the prescribed dose (48,49,62).
In the last decade, improvements in GRID design, ability to deliver higher tumor dose by improved target penetration along with reduced normal tissue damage as well as superior dosimetry have resulted in dramatic improvements in clinical responses (62-67). Unnecessary high dose exposure of the surrounding normal tissue can be significantly reduced by reconfiguring the GRID treatment into a 3D GRID dose in form of LATTICE. We now define 3D GRID as LATTICE which is a new approach to spatially fractionated radiation that takes advantage of modern-era technology of SABR systems in a safer and efficient way (68). The difference in the dose delivery is shown at the URL (http://assets.cureus.com/uploads/figure/file/538/13fig1.png) published by Wu et al. (68). Using this technique, high doses of radiation are concentrated at vertices within the tumor volume, with drastically lower dose between vertices (peak-to-valley effect) and leaving anything outside of tumor volume minimally exposed. Because more pronounced radiation dose peaks and valleys are generated using LATTICE technique compared to 2D-GRID, it may be more radio-biologically effective, with lower radiation dose to adjacent normal tissues resulting in a reduction in normal tissue toxicity.
Hypofractionation and normal tissue toxicity
The α/β ratios derived from linear quadratic model of the radiation survival curve describes the effectiveness of the dose and is used to model cell survival at different conventional doses used in radiation therapy (69). A similar approach has also been adapted to model cell survival with the large doses for hypofractionation studies (70,71). However, this approach may overestimate tumor control. Because of the improvements in radiotherapy planning and delivery, targeting accuracy of radiation to the tumor is also improved with a reduction in surrounding normal tissue damage. It is feasible to use higher doses of radiation per fraction without inducing significant acute and late radiation induced toxicity with SABR. However, concerns still remain on the late toxicity with high dose hypofractionation and it must be emphasized that these may take many years or a decade or more to be seen. An intriguing concept for both technological limitations and capabilities and also for biological advantages is to consider irradiating only limited portions of the tumor and still achieve similar or better outcomes with SABR as discussed next.
When large doses of radiation are delivered to only a fraction of the target volume, scaling back on the irradiated tumor volume invariably results in a reduction of dose to the adjacent normal tissues. Such scaling back of target treatment volume may not compromise the benefits of high dose per fraction for better control because underlying radiobiological mechanisms of damage by large dose per fractions remain the same. SFGRT (2D-Grid) and now LATTICE (3D-Grid), results in a better dose distribution in tumor spatially rather than temporally, which results in significantly improved sparing of normal tissue achieving a better tumor control.
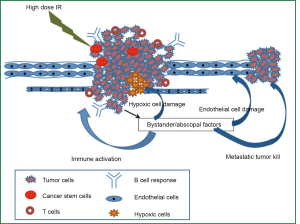
Next we discuss the role of three underlying radiobiological mechanisms of bystander/abscopal effects, activation of immune system, and damage to endothelial cells, that might contributing to a better tumor control with SFGRT and LATTICE in salvage settings, however, needs randomized trials for definitive treatment practices.
High dose radiation-induces factors leading to bystander/abscopal effects
Brooks et al. reported the first observation of radiation-induced non-targeted effects in a hamster model (72). Although evidences for these effects have accumulated over time, the exact mechanisms by which they cause tumor regression distant to site of irradiation remains somewhat speculative. A few major mechanistic categories have been proposed to account for abscopal effects based on studies involving different malignancies: immune system, cytokines and pseudo-abscopal effect (73).
Cell-cell communication appears to play an important role in mediating the bystander effect, and there may also be contributions from the transfers of soluble mediators generated in irradiated medium. It is most likely that multiple mechanisms are involved in bystander effects. The presence of gap junctions is not essential. Transfer of radiation-conditioned medium (RCM) from confluent cell culture is more effective, a phenomenon that is termed as “indirect radiation effects” (74-77). Irradiated cells may release clastogenic factors into serum that will induce chromosomal damage when transferred to cultured cells from unirradiated donors (78-80). In a study in rats, for example, clastogenic activity persisted in circulating plasma of irradiated animals for the 10-week duration of the study, and was not abrogated by diluting with non-irradiated serum. Serum irradiated in vitro was not clastogenic suggesting that these factors were released from the irradiated cells (81).
Although evidence for the presence of these factors has been accumulating over past decades, their exact nature as well as the mechanisms by which they cause the distant bystander effects (more of an abscopal effect) has proven elusive. One such mechanism might be through radiation-induced early genes and induction of cytokines. Indeed, TNF-α and TRAIL are directly involved in apoptosis and are induced by ionizing radiation (82-86). There is a demonstrated correlation of therapeutic efficacy following SFGRT with TNF-α induction in the serum obtained from these patients as well as ceramide production (48,49).
For SFGRT, the “bystander effect” is within the GRID irradiated tumor volume that falls directly under shielded regions (low-dose regions) of the GRID. Bystander factors, such as TNF-α shown by Sathishkumar et al. (49) and Shareef et al. (47); TRAIL shown by Shareef et al. (47) and ceramide shown by Sathishkumar et al. (48) are induced in cells that are under the open field of the high-dose GRID areas and are hypothesized to be responsible for initiating the cell death cascade both in the epithelial and endothelial compartments of the tumor micro-environment. Recent reports have demonstrated the presence of radiation-induce signal transduction leading to significant DNA damage and cellular stress (87,88). In addition to the bystander effect within the GRID-irradiated tumor, Peters et al. (3) reported that there is robust “abscopal effect” in distant tumors or metastatic lesions that are not irradiated or treated and has been reported clinically with the use of large doses (89).
In this respect, recently using SFGRT we found both bystander and abscopal effects in mice bearing A549 lung adenocarcinoma xenograft contra-lateral tumors (90). Maximal abscopal effect was observed in unirradiated right tumor when mice was exposed to 15 Gy SFGRT followed by 5 fractions of 2 Gy to the left tumor suggesting that the abscopal effect can be amplified by sequential combination of SFGRT with conventional fractionation. More recently, using LATTICE therapy we obtained similar results in mice bearing syngenic Lewis Lung Carcinoma (LLC) contra-lateral tumors (91). These findings strongly suggest that SFGRT is more potent in eliciting evident abscopal effect in the un-irradiated tumor than conventional dosimetric approaches.
High dose radiation activates immune system
There are quite a few reports that support the important role of immune factors in mediating the abscopal effects (92,93). In contrast to the generally believed notion that radiation therapy is immunosuppressive, recent reports indicate ablative high dose radiation therapy could activate immune system and reduce the primary tumor burden as well as distant metastasis (51,94). These effects were mediated by radiation therapy induced disruption of physical and immunologic barriers, stimulation of danger signaling pathways, increase in dendritic cells cross-presentation of tumor antigen, and possibly reversal of T-cell unresponsiveness in tumor-bearing hosts, leading to a rejection of local and distant tumors (51). Subsequently these authors demonstrated that IFN-α/β produced by tumor-infiltrating myeloid cells in an autocrine fashion is required to endow tumor-infiltrating dendritic cells with T-cell cross-priming capacity following local RT; however, T cells do not need to bear the type I IFN receptor to mediate tumor rejection (94). Together, these results score the importance of cytotoxic T-cell mediated antitumor immunity that mediates tumor regression. Our unpublished results show that RCM obtained from lymphoblasts is able to induce killing of lung cancer (A549) cells, suggesting that the immune factors in addition to cytokines and ceramide pathway may be involved. However, in our contra-lateral tumor xenograft athymic nude mice, we observed significant bystander and abscopal effects indicating that not only the T-cell mediated immune factors but also humoral immunity may play an important role in the radiation-induced abscopal effects. These observations suggest potential therapeutic role for immune factors.
Lee et al. (51) reported that reduction of tumor burden after ablative radiation depends largely on T-cell responses as it dramatically increases T-cell priming in draining lymphoid tissues, leading to reduction/eradication of the primary tumor or distant metastasis in a CD8(+) T cell-dependent fashion. Interestingly, this study observed that ablative radiation-initiated immune responses and tumor reduction are abrogated by conventional fractionated RT or adjuvant chemotherapy (if given after a week of single ablative dose) but greatly amplified by local immunotherapy. However, in SFGRT settings we observed significant enhanced response when the high dose radiation was followed by fractionated 2 Gy fractions (given after 24 hrs), implying that spatial fractionation of radiation delivery might activate immune factors that can synergize with the conventional fractionated radiation. These results strongly argue for more detailed investigations to elucidate the role of immune factors in radiation therapy.
High dose radiation induces damage to endothelium
Engagement of the vascular component in tumor response to radiation therapy has been a topic of interest in recent literature. However, in addition to release of cytokines, impaired blood vessel formation and induction of endothelial cell death in tumors not exposed to radiation have been demonstrated to play a role in abscopal effect (95). Endothelial cells generate 20-fold more of a unique form of acid sphingomyelinase (ASMase), termed Secretory ASMase, than any other cell type in the body. Secretory ASMase activation is required for ionizing radiation to kill endothelium (96), as endothelium in lung, gut, and brain are totally resistant to radiation-induced apoptotic death in the absence of ASMase. Garcia-Barros et al. (50) have postulated that high dose radiation-induced damage (15 Gy) to the endothelial cells could convert Potentially Lethal Damage (PLD) in tumor cells and cancer stem cells to lethal damage resulting in tumor cell death. Animal studies have shown that radiation at doses higher than 10 Gy induces endothelial apoptosis by activation of acid sphingomyelinase (ASMase) and ceramide generation (50,96-99); these effects that are not observed with conventional radiation doses. Findings by Garcia-Barros et al. (50) suggest that high-dose radiation-induced tumor regression can be entirely dependent on tumor endothelium apoptosis since these effects were abolished in ASMase knockout animals implanted with functional ASMase MCA/129 fibrosarcomas and B16F1 melanomas and restored upon bone marrow transplantation of ASMase functional stem cells. Further, elevated sphingomyelinase activity and ceramide concentration in the serum of patients undergoing high dose spatially fractionated radiation treatment were observed (48). Our unpublished findings in A549 xenografts showed increased elevation of ceramide in the serum of nude mice treated with SFGRT (90).
Although direct killing effect of tumor cells with SFGRT occurs, it cannot completely account for tumor regression observed after treatment. Recently, we demonstrated that treatment of 11 patients with various types of cancer with 15 Gy SFGRT therapy followed by multiple consecutive doses of 2 Gy each led to an increase in the activity of ASMase in serum and a corresponding elevation in the concentration of LDL-enriched ceramide. These changes correlated with the clinical outcome of the treatment, as they were found only in the 76% of patients with CR or PR and not in non-responders (48). It is evident that there is a biologic/therapeutic consequence of this response, whereby high single dose radiotherapy requires ceramide-driven endothelial apoptosis for tumor cure (50,100). This observation has broad implications for cancer treatment and is a subject of active debate in the field, as it is generally believed that radiation therapy works by partly targeting tumor stem cells and it is unclear which components of tumor microenvironment play important role in radiation cure.
There exists data on ceramide production, its relation to endothelial apoptosis and induction of abscopal regression of distant tumor with radiation exposure, however, there is little or any information available on the impact of negative regulators of ceramide pathway in radioresistance/radiosensitivity, their association with release of cytokines, and finally any possible cross-talks during cellular events associated with abscopal phenomena.
Hypofractionation and hypoxia
Tumor hypoxia has been observed in many human cancers and has been a major impediment for the success of radiotherapy. Generally, the phenomenon of re-oxygenation of hypoxic cells between several fractions of conventionally fractionated radiation therapy is considered to increase the sensitivity of the cells that were previously hypoxic. With the encouraging results using SABR or other hypofractionation strategies, this is a point of considerable debate whether the issue of hypoxia under such therapy settings. Taking into account several factors such as the potential over-estimation of cell killing and tumor control by the linear quadratic model at large doses, high dose hypofractionation has actually resulted in greater than expected tumor control. It is possible that single dose hypofractionation induced specific mechanisms abate hypoxia, or that the extreme ablative doses currently used in many SABR protocols are already high enough to overcome hypoxic radioresistance or both. The latter hypothesis implies that concurrent strategies (such as hypoxic cytotoxin) targeted directly at hypoxic cells might improve the therapeutic ratio of SABR and allow clinicians to treat with a larger fraction in the patient population.
Fractional doses in hypofractionation schemes vary significantly in clinical practice, from 3 Gy/fraction to 20 Gy/fraction. There are a number of processes that will be effected by dose size and fractionation that could be exploited, including changes in the “4-R’s” (repair, repopulation, redistribution and reoxygenation), consequence of endothelial damage (which could worsen hypoxia) or tumor shrinkage (which could lessen hypoxia) and impact of the high dose on factors secreted by the tumor.
An example of the latter comes from our unpublished results (101). In two lung cancer cell lines, we observed that conditioned media collected from 10 Gy-irradiated hypoxic A549 cells (H-RCM) showed highly reduced cell proliferation effect on normoxic A549 cells when compared to media collected from irradiated normoxic A549 cells (N-RCM). Interestingly, with H-RCM obtained from 10 Gy irradiated hypoxic H-460 cells showed a significantly decreased cell proliferation in H460 cells but such reduced cell proliferation was absent with H-RCM obtained from 2 Gy irradiated hypoxic H-460 cells (101). This suggests that oxygen may potentially negate bystander effect. Nonetheless more data are needed, including modeling that would help define the potential complexities, for example, one recently published that aims to account for intercellular signaling (102).
How to best take advantage of the high dose effect but also not damage normal tissue remains to be established. This could include partial treatment of the tumor to high dose using a variety of technique such as the high-dose LATTICE approach. That might have positive effects on damaging the endothelial compartment and/or immune activation. Another important aspect that is not discussed in detail could be differential effect of hypofractionation on cancer stem cells.
Summary of new biology of hypofractionation
- Hypofractionated radiotherapy (>12 Gy) is an attractive approach in the management of cancer although long-term toxicity in patients with curative tumors remains to be evaluated as series mature.
- Success of hypofractionated radiotherapy is dependent on its ability to deliver a markedly higher dose to the target volume without damage to surrounding normal tissue. Over the last decade, technological improvement in terms of dose delivery and intra-tumoral spatial distribution of dose seems to have been achieved, with long-term data needed to see if the spatial distribution of dose can reduce normal tissue injury and maintain or even improve tumor control.
- The underlying radiobiological mechanisms for improved outcome obtained by high dose hypofractionated radiation therapy could be multifactorial, which include differential endothelial and cancer stem cell killing, overcoming hypoxic radioresistance, activation of complex immunological pathways, and bystander/abscopal tumoricidal effects, resulting in improved treatment outcome (Figure 2).
- There appears to be opportunities to achieve better response of tumors to high dose fractionated radiotherapy by the use of chemotherapeutic drugs or hypoxic cell radiosensitizers.
- While speculative, the use of spatial fractionation in the form of 2D SFGRT and 3D LATTICE in combination with conventional fractionated radiation therapy or chemotherapeutic drugs or hypoxic cytotoxins might be able to counteract the effects of hypoxia with simultaneous normal tissue sparing. In conclusion, ablative hypofractionation schemes are effective in certain solid tumors that may take advantage of new aspects of radiation biology by involving certain components of tumor microenvironment such as effects on vasculature as well as immunologic modulation.SFGRT provided some mechanistic insights pre-clinically as well as from patients (who received SFGRT as salvage therapy), however, to bring SFGRT in the mainstream needs more well designed trials Lattice (3D-Grid) has some promise in the main realm of definitive treatment, however, this approach warrants robust randomized trials. Overall, it is the ablative dose (delivery approaches may differ with or without homogenous dose distribution) that needs further exploration based on clinical observation of its efficacy and preclinical studies.
Overall conclusions
While hyper- and hypo-fractionation are presented as distinctly different, a key point to emphasize is that radiation fraction size and schedule have properties that can be exploited using radiation alone and in combination with immunotherapy, molecular target treatment and cytotoxic chemotherapy. Improvements in imaging and technology of treatment delivery can allow improvement in anatomical targeting and also in treating based on the physiological and biological processes as they present and evolve. New techniques such as LATTICE may be able to take advantage of heterogeneous dose delivery.
While there is a good deal of new and exciting data there is much research to do and, of course, the ultimate proof will be from well-designed clinical trials. Radiation therapy and radiation biology are far from static and with the ability for precision targeting and dose delivery, radiation “as a drug” can have a major impact in multi-modality cancer treatment.
Acknowledgements
Authors acknowledge Dr. Bhadrasain Vikram for critical reading and suggestions.
Disclosure: The authors declare no conflict of interest.
References
- Delaney G, Jacob S, Featherstone C, et al. The role of radiotherapy in cancer treatment: estimating optimal utilization from a review of evidence-based clinical guidelines. Cancer 2005;104:1129-37. [PubMed]
- Liu FF. Lessons learned from radiation oncology clinical trials. Clin Cancer Res 2013;19:6089-100. [PubMed]
- Peters ME, Shareef MM, Gupta S, et al. Potential Utilization of Bystander/Abscopal-Mediated Signal Transduction Events in the Treatment of Solid Tumors. Current Signal Transduction Therapy 2007;2:129-43.
- Feinendegen LE. Evidence for beneficial low level radiation effects and radiation hormesis. Br J Radiol 2005;78:3-7. [PubMed]
- Brenner DJ, Doll R, Goodhead DT, et al. Cancer risks attributable to low doses of ionizing radiation: assessing what we really know. Proc Natl Acad Sci U S A 2003;100:13761-6. [PubMed]
- Joiner MC. Induced radioresistance: an overview and historical perspective. Int J Radiat Biol 1994;65:79-84. [PubMed]
- Lambin P, Marples B, Fertil B, et al. Hypersensitivity of a human tumour cell line to very low radiation doses. Int J Radiat Biol 1993;63:639-50. [PubMed]
- Wouters BG, Skarsgard LD. The response of a human tumor cell line to low radiation doses: evidence of enhanced sensitivity. Radiat Res 1994;138:S76-80. [PubMed]
- Marples B, Wouters BG, Collis SJ, et al. Low-dose hyper-radiosensitivity: a consequence of ineffective cell cycle arrest of radiation-damaged G2-phase cells. Radiat Res 2004;161:247-55. [PubMed]
- Marples B, Wouters BG, Joiner MC. An association between the radiation-induced arrest of G2-phase cells and low-dose hyper-radiosensitivity: a plausible underlying mechanism? Radiat Res 2003;160:38-45. [PubMed]
- Short S, Mayes C, Woodcock M, et al. Low dose hypersensitivity in the T98G human glioblastoma cell line. Int J Radiat Biol 1999;75:847-55. [PubMed]
- Chendil D, Oakes R, Alcock RA, et al. Low dose fractionated radiation enhances the radiosensitization effect of paclitaxel in colorectal tumor cells with mutant p53. Cancer 2000;89:1893-900. [PubMed]
- Enns L, Bogen KT, Wizniak J, et al. Low-dose radiation hypersensitivity is associated with p53-dependent apoptosis. Mol Cancer Res 2004;2:557-66. [PubMed]
- Joiner MC, Johns H. Renal damage in the mouse: the response to very small doses per fraction. Radiat Res 1988;114:385-98. [PubMed]
- Dey S, Spring PM, Arnold S, et al. Low-dose fractionated radiation potentiates the effects of Paclitaxel in wild-type and mutant p53 head and neck tumor cell lines. Clin Cancer Res 2003;9:1557-65. [PubMed]
- Short SC, Mitchell SA, Boulton P, et al. The response of human glioma cell lines to low-dose radiation exposure. Int J Radiat Biol 1999;75:1341-8. [PubMed]
- Skov KA. Radioresponsiveness at low doses: hyper-radiosensitivity and increased radioresistance in mammalian cells. Mutat Res 1999;430:241-53. [PubMed]
- Joiner MC, Marples B, Lambin P, et al. Low-dose hypersensitivity: current status and possible mechanisms. Int J Radiat Oncol Biol Phys 2001;49:379-89. [PubMed]
- Short SC, Kelly J, Mayes CR, et al. Low-dose hypersensitivity after fractionated low-dose irradiation in vitro. Int J Radiat Biol 2001;77:655-64. [PubMed]
- Shareef MM, Brown B, Shajahan S, et al. Lack of P-glycoprotein expression by low-dose fractionated radiation results from loss of nuclear factor-kappaB and NF-Y activation in oral carcinoma cells. Mol Cancer Res 2008;6:89-98. [PubMed]
- Gupta S, Koru-Sengul T, Arnold SM, et al. Low-dose fractionated radiation potentiates the effects of cisplatin independent of the hyper-radiation sensitivity in human lung cancer cells. Mol Cancer Ther 2011;10:292-302. [PubMed]
- Carrier F, Diss E, Nalabothula N, et al. The Histone Deacetylase Inhibitor Vorinostat Induces Hyper-radiosensitivity (HRS) In P53 Wild Type Glioblastoma Cells. In The 53rd Annual Meeting of American Society for Radiation Oncology. 2011. Miami Beach, FL: ASTRO.
- Bakkenist CJ, Kastan MB. DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature 2003;421:499-506. [PubMed]
- Suzuki K, Kodama S, Watanabe M. Low-dose radiation effects and intracellular signaling pathways. Yakugaku Zasshi 2006;126:859-67. [PubMed]
- Tichý A, Záskodová D, Rezácová M, et al. Gamma-radiation-induced ATM-dependent signalling in human T-lymphocyte leukemic cells, MOLT-4. Acta Biochim Pol 2007;54:281-7. [PubMed]
- Krause M, Hessel F, Wohlfarth J, et al. Ultrafractionation in A7 human malignant glioma in nude mice. Int J Radiat Biol 2003;79:377-83. [PubMed]
- Krause M, Joiner M, Baumann M. Ultrafractionation in human malignant glioma xenografts. Int J Cancer 2003;107:333-author reply 334. [PubMed]
- Beauchesne PD, Bertrand S, Branche R, et al. Human malignant glioma cell lines are sensitive to low radiation doses. Int J Cancer 2003;105:33-40. [PubMed]
- Spring PM, Arnold SM, Shajahan S, et al. Low dose fractionated radiation potentiates the effects of taxotere in nude mice xenografts of squamous cell carcinoma of head and neck. Cell Cycle 2004;3:479-85. [PubMed]
- Tyagi N, Yang K, Sandhu R, et al. External beam pulsed low dose radiotherapy using volumetric modulated arc therapy: planning and delivery. Med Phys 2013;40:011704. [PubMed]
- Dilworth JT, Krueger SA, Dabjan M, et al. Pulsed low-dose irradiation of orthotopic glioblastoma multiforme (GBM) in a pre-clinical model: effects on vascularization and tumor control. Radiother Oncol 2013;108:149-54. [PubMed]
- Lee DY, Chunta JL, Park SS, et al. Pulsed versus conventional radiation therapy in combination with temozolomide in a murine orthotopic model of glioblastoma multiforme. Int J Radiat Oncol Biol Phys 2013;86:978-85. [PubMed]
- Modesitt SC, Gupta S, Brandon J, et al. Low dose fractionated radiation as a chemopotentiator for in vitro and in vivo ovarian cancer models. Proc Amer Assoc Cancer Res 2005;46:abstr 4748.
- Arnold SM, Regine WF, Ahmed MM, et al. Low-dose fractionated radiation as a chemopotentiator of neoadjuvant paclitaxel and carboplatin for locally advanced squamous cell carcinoma of the head and neck: results of a new treatment paradigm. Int J Radiat Oncol Biol Phys 2004;58:1411-7. [PubMed]
- Gleason JF Jr, Kudrimoti M, Van Meter EM, et al. Low-dose fractionated radiation with induction chemotherapy for locally advanced head and neck cancer: 5 year results of a prospective phase II trial. Int J Radiat Oncol Biol Phys 2013;2:35-42.
- Kunos CA, Sill MW, Buekers TE, et al. Low-dose abdominal radiation as a docetaxel chemosensitizer for recurrent epithelial ovarian cancer: a phase I study of the Gynecologic Oncology Group. Gynecol Oncol 2011;120:224-8. [PubMed]
- Regine WF, Hanna N, Garofalo MC, et al. Low-dose radiotherapy as a chemopotentiator of gemcitabine in tumors of the pancreas or small bowel: a phase I study exploring a new treatment paradigm. Int J Radiat Oncol Biol Phys 2007;68:172-7. [PubMed]
- Wrenn DC, Saigal K, Lucci JA 3rd, et al. A Phase I Study using low-dose fractionated whole abdominal radiotherapy as a chemopotentiator to full-dose cisplatin for optimally debulked stage III/IV carcinoma of the endometrium. Gynecol Oncol 2011;122:59-62. [PubMed]
- Balducci M, Chiesa S, Diletto B, et al. Low-dose fractionated radiotherapy and concomitant chemotherapy in glioblastoma multiforme with poor prognosis: a feasibility study. Neuro Oncol 2012;14:79-86. [PubMed]
- Nardone L, Valentini V, Marino L, et al. A feasibility study of neo-adjuvant low-dose fractionated radiotherapy with two different concurrent anthracycline-docetaxel schedules in stage IIA/B-IIIA breast cancer. Tumori 2012;98:79-85. [PubMed]
- Mantini G, Valentini V, Meduri B, et al. Low-dose radiotherapy as a chemo-potentiator of a chemotherapy regimen with pemetrexed for recurrent non-small-cell lung cancer: a prospective phase II study. Radiother Oncol 2012;105:161-6. [PubMed]
- Silver NL, Arnold S, Gleason JF, et al. p16 status predicts resp onse to low dose fractionated radiation as a chemopotentiator of neoadjuvant chemotherapy for locally advanced squamous cell carcinoma of the head and neck. In 8th International Conference on Head and Neck Cancer. 2012. Toronto, Ontario, Canada: American Head & Neck Society.
- Ang KK, Sturgis EM. Human papillomavirus as a marker of the natural history and response to therapy of head and neck squamous cell carcinoma. Semin Radiat Oncol 2012;22:128-42. [PubMed]
- Snow AN, Laudadio J. Human papillomavirus detection in head and neck squamous cell carcinomas. Adv Anat Pathol 2010;17:394-403. [PubMed]
- Baumann P, Nyman J, Hoyer M, et al. Outcome in a prospective phase II trial of medically inoperable stage I non-small-cell lung cancer patients treated with stereotactic body radiotherapy. J Clin Oncol 2009;27:3290-6. [PubMed]
- Fakiris AJ, McGarry RC, Yiannoutsos CT, et al. Stereotactic body radiation therapy for early-stage non-small-cell lung carcinoma: four-year results of a prospective phase II study. Int J Radiat Oncol Biol Phys 2009;75:677-82. [PubMed]
- Shareef MM, Cui N, Burikhanov R, et al. Role of tumor necrosis factor-alpha and TRAIL in high-dose radiation-induced bystander signaling in lung adenocarcinoma. Cancer Res 2007;67:11811-20. [PubMed]
- Sathishkumar S, Boyanovsky B, Karakashian AA, et al. Elevated sphingomyelinase activity and ceramide concentration in serum of patients undergoing high dose spatially fractionated radiation treatment: implications for endothelial apoptosis. Cancer Biol Ther 2005;4:979-86. [PubMed]
- Sathishkumar S, Dey S, Meigooni AS, et al. The impact of TNF-alpha induction on therapeutic efficacy following high dose spatially fractionated (GRID) radiation. Technol Cancer Res Treat 2002;1:141-7. [PubMed]
- Garcia-Barros M, Paris F, Cordon-Cardo C, et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science 2003;300:1155-9. [PubMed]
- Lee Y, Auh SL, Wang Y, et al. Therapeutic effects of ablative radiation on local tumor require CD8+ T cells: changing strategies for cancer treatment. Blood 2009;114:589-95. [PubMed]
- Bao S, Wu Q, McLendon RE, et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006;444:756-60. [PubMed]
- Brown JM, Diehn M, Loo BW Jr. Stereotactic ablative radiotherapy should be combined with a hypoxic cell radiosensitizer. Int J Radiat Oncol Biol Phys 2010;78:323-7. [PubMed]
- Song CW, Levitt SH, Park H. Response to “tereotactic ablative radiotherapy in the framework of classical radiobiology: response to Drs. Brown, Diehn, and Loo.”(Int J Radiat Oncol Biol Phys 2011;79:1599-1600) and “nfluence of tumor hypoxia on stereotactic ablative radiotherapy (SABR): response to Drs. Mayer and Timmerman.” Int J Radiation Oncol Biol Phys 2011;78:1600). Int J Radiat Oncol Biol Phys 2011;81:1193-author reply 1193-4. [PubMed]
- Meyer J, Timmerman R. Stereotactic ablative radiotherapy in the framework of classical radiobiology: response to Drs. Brown, Diehn, and Loo. Int J Radiat Oncol Biol Phys 2011;79:1599-600; author reply 1600. [PubMed]
- Schenken LL, Poulakos L, Hagemann RF. Responses of an experimental solid tumour to irradiation: A comparison of modes of fractionation. Br J Cancer 1975;31:228-36. [PubMed]
- Sakamoto K, Sakka M. The effect of bleomycin and its combined effect with radiation on murine squamous carcinoma treated in vivo. Br J Cancer 1974;30:463-8. [PubMed]
- Phillips MH, Stelzer KJ, Griffin TW, et al. Stereotactic radiosurgery: a review and comparison of methods. J Clin Oncol 1994;12:1085-99. [PubMed]
- Thariat J, Hannoun-Levi JM, Sun Myint A, et al. Past, present, and future of radiotherapy for the benefit of patients. Nat Rev Clin Oncol 2013;10:52-60. [PubMed]
- Levin WP, Kooy H, Loeffler JS, et al. Proton beam therapy. Br J Cancer 2005;93:849-54. [PubMed]
- Kavanagh BD, Timmerman RD. Stereotactic radiosurgery and stereotactic body radiation therapy: an overview of technical considerations and clinical applications. Hematol Oncol Clin North Am 2006;20:87-95. [PubMed]
- Mohiuddin M, Fujita M, Regine WF, et al. High-dose spatially-fractionated radiation (GRID): a new paradigm in the management of advanced cancers. Int J Radiat Oncol Biol Phys 1999;45:721-7. [PubMed]
- Meigooni AS, Parker SA, Zheng J, et al. Dosimetric characteristics with spatial fractionation using electron grid therapy. Med Dosim 2002;27:37-42. [PubMed]
- Reiff JE, Huq MS, Mohiuddin M, et al. Dosimetric properties of megavoltage grid therapy. Int J Radiat Oncol Biol Phys 1995;33:937-42. [PubMed]
- Mohiuddin M, Curtis DL, Grizos WT, et al. Palliative treatment of advanced cancer using multiple nonconfluent pencil beam radiation. A pilot study. Cancer 1990;66:114-8. [PubMed]
- Huhn JL, Regine WF, Valentino JP, et al. Spatially fractionated GRID radiation treatment of advanced neck disease associated with head and neck cancer. Technol Cancer Res Treat 2006;5:607-12. [PubMed]
- Neuner G, Mohiuddin MM, Vander Walde N, et al. High-dose spatially fractionated GRID radiation therapy (SFGRT): a comparison of treatment outcomes with Cerrobend vs. MLC SFGRT. Int J Radiat Oncol Biol Phys 2012;82:1642-9. [PubMed]
- Wu X, Ahmed MM, Wright J, et al. On Modern Technical Approaches of Three-Dimensional High-Dose Lattice Radiotherapy (LRT). Cureus 2010. doi: . [PubMed]
- Williams MV, Denekamp J, Fowler JF. A review of alpha/beta ratios for experimental tumors: implications for clinical studies of altered fractionation. Int J Radiat Oncol Biol Phys 1985;11:87-96. [PubMed]
- Joiner MC. A simple alpha/beta-independent method to derive fully isoeffective schedules following changes in dose per fraction. Int J Radiat Oncol Biol Phys 2004;58:871-5. [PubMed]
- Hoban PW, Jones LC, Clark BG. Modeling late effects in hypofractionated stereotactic radiotherapy. Int J Radiat Oncol Biol Phys 1999;43:199-210. [PubMed]
- Brooks AL, Benjamin SA, McClellan RO. Toxicity of 90Sr-90Y in Chinese hamsters. Radiat Res 1974;57:471-81. [PubMed]
- Kaminski JM, Shinohara E, Summers JB, et al. The controversial abscopal effect. Cancer Treat Rev 2005;31:159-72. [PubMed]
- Lyng FM, Seymour CB, Mothersill C. Early events in the apoptotic cascade initiated in cells treated with medium from the progeny of irradiated cells. Radiat Prot Dosimetry 2002;99:169-72. [PubMed]
- Lyng FM, Seymour CB, Mothersill C. Initiation of apoptosis in cells exposed to medium from the progeny of irradiated cells: a possible mechanism for bystander-induced genomic instability? Radiat Res 2002;157:365-70. [PubMed]
- Hall EJ. The bystander effect. Health Phys 2003;85:31-5. [PubMed]
- Hall EJ, Hei TK. Genomic instability and bystander effects induced by high-LET radiation. Oncogene 2003;22:7034-42. [PubMed]
- Goh K, Sumner H. Breaks in normal human chromosomes: are they induced by a transferable substance in the plasma of persons exposed to total-body irradiation? Radiat Res 1968;35:171-81. [PubMed]
- Hollowell JG Jr, Littlefield LG. Chromosome damage induced by plasma of x-rayed patients: an indirect effect of x-ray. Proc Soc Exp Biol Med 1968;129:240-4. [PubMed]
- Sharpe HB, Scott D, Dolphin GW. Chromosome aberrations induced in human lymphocytes by x-irradiation in vitro: the effect of culture techniques and blood donors on aberration yield. Mutat Res 1969;7:453-61. [PubMed]
- Faguet GB, Reichard SM, Welter DA. Radiation-induced clastogenic plasma factors. Cancer Genet Cytogenet 1984;12:73-83. [PubMed]
- Ahmed MM, Sells SF, Venkatasubbarao K, et al. Ionizing radiation-inducible apoptosis in the absence of p53 linked to transcription factor EGR-1. J Biol Chem 1997;272:33056-61. [PubMed]
- Hallahan DE, Spriggs DR, Beckett MA, et al. Increased tumor necrosis factor alpha mRNA after cellular exposure to ionizing radiation. Proc Natl Acad Sci U S A 1989;86:10104-7. [PubMed]
- Hallahan DE, Haimovitz-Friedman A, Kufe DW, et al. The role of cytokines in radiation oncology. Important Adv Oncol 1993;71-80. [PubMed]
- Hallahan DE, Virudachalam S, Sherman ML, et al. Tumor necrosis factor gene expression is mediated by protein kinase C following activation by ionizing radiation. Cancer Res 1991;51:4565-9. [PubMed]
- Unnithan J, Macklis RM. TRAIL induction by radiation in lymphoma patients. Cancer Invest 2004;22:522-5. [PubMed]
- Asur R, Butterworth KT, Penagaricano JA, et al. High dose bystander effects in spatially fractionated radiation therapy. Cancer Lett 2013. [Epub ahead of print]. [PubMed]
- Asur RS, Sharma S, Chang CW, et al. Spatially fractionated radiation induces cytotoxicity and changes in gene expression in bystander and radiation adjacent murine carcinoma cells. Radiat Res 2012;177:751-65. [PubMed]
- Konoeda K. Therapeutic efficacy of pre-operative radiotherapy on breast carcinoma: in special reference to its abscopal effect on metastatic lymph-nodes. Nihon Gan Chiryo Gakkai Shi 1990;25:1204-14. [PubMed]
- Gupta S, Zagurovskaya M, Wu X, et al. Spatially Fractionated Grid High-dose radiation-induced tumor regression in A549 lung adenocarcinoma xenografts: cytokines and ceramide regulators balance in abscopal phenomena. Sylvester Comprehensive Cancer Center, 2014:20.
- Kanagavelu S, Gupta S, Wu X, et al. In vitro and in vivo effects of lattice radiation therapy on local and distant lung cancer. Sylvester Comprehensive Cancer Center, 2014:19.
- Dewan MZ, Galloway AE, Kawashima N, et al. Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Clin Cancer Res 2009;15:5379-88. [PubMed]
- Ilnytskyy Y, Koturbash I, Kovalchuk O. Radiation-induced bystander effects in vivo are epigenetically regulated in a tissue-specific manner. Environ Mol Mutagen 2009;50:105-13. [PubMed]
- Burnette BC, Liang H, Lee Y, et al. The efficacy of radiotherapy relies upon induction of type i interferon-dependent innate and adaptive immunity. Cancer Res 2011;71:2488-96. [PubMed]
- Camphausen K, Moses MA, Ménard C, et al. Radiation abscopal antitumor effect is mediated through p53. Cancer Res 2003;63:1990-3. [PubMed]
- Santana P, Peña LA, Haimovitz-Friedman A, et al. Acid sphingomyelinase-deficient human lymphoblasts and mice are defective in radiation-induced apoptosis. Cell 1996;86:189-99. [PubMed]
- Lin X, Fuks Z, Kolesnick R. Ceramide mediates radiation-induced death of endothelium. Crit Care Med 2000;28:N87-93.
- Haimovitz-Friedman A, Balaban N, McLoughlin M, et al. Protein kinase C mediates basic fibroblast growth factor protection of endothelial cells against radiation-induced apoptosis. Cancer Res 1994;54:2591-7. [PubMed]
- Haimovitz-Friedman A, Kan CC, Ehleiter D, et al. Ionizing radiation acts on cellular membranes to generate ceramide and initiate apoptosis. J Exp Med 1994;180:525-35. [PubMed]
- Truman JP, García-Barros M, Kaag M, et al. Endothelial membrane remodeling is obligate for anti-angiogenic radiosensitization during tumor radiosurgery. PLoS One 2010;5:e12310. [PubMed]
- Gupta S, Tubin S, Ahmed MM. Radiation-induced bystander effects in normoxic and hypoxic conditions in human lung cancer cells. In 14th International Congress of Radiation Research, Warsaw, Poland, 2011:71.
- McMahon SJ, McGarry CK, Butterworth KT, et al. Implications of intercellular signaling for radiation therapy: a theoretical dose-planning study. Int J Radiat Oncol Biol Phys 2013;87:1148-54. [PubMed]


