Introduction
Lung biopsy is a relatively frequently performed procedure with considerable benefit for patient. It is a multidisciplinary procedure involving respiratory physicians, pathologists, and radiologists with an interest in chest diseases. In recent years, CT guidance has become the primary imaging modality, replacing fluoroscopy guided biopsies in many institutions. Also, the total number of lung biopsies performed has increased in the past decade. All invasive procedures have a morbidity and mortality rate associated with them, which are important in considering whether to subject the patient to a procedure, with the most common complications of lung biopsy being pneumothorax and hemoptysis. Several recent studies have evaluated possible reasons and management of complication after transthoracic needle biopsy (TTNB), which will be discussed in this review (1,2).
In about 90 percent of patients, needle biopsy provides enough tissue for the pathologist to determine the cause of the abnormality. The indications for TTNB have altered substantially since the technique was developed, reflecting changes in many areas, including needle technology, imaging techniques, and immunohistochemistry and cytochemistry. Further advances, in the field of Radiology, particularly in positron emission tomography (PET), may alter the indications for needle biopsy and, in particular, the management of the solitary nodule. TTNB is one of several methods available for tissue diagnosis of suspected lung cancer (3,4).
TTNB can be performed, depending on a needle type, as well as type of material providing, as fine needle aspiration biopsy (FNAB), providing samples for cytology, which can accurately diagnose malignancy, while the more recent development of core needle biopsy (CNB), providing histological material, has enabled a firm diagnosis of benign lesions to be made, thus improving overall diagnostic accuracy (5-7).
Needle biopsy is typically an outpatient procedure with infrequent complications and as such has become a standard and very useful tool in the routine diagnostic workup of lung lesions. The problem most responsible for complicating outpatient management is not the presence of the pneumothorax per se, but an increase in the size of the pneumothorax that requires chest tube placement and patient hospitalization (8,9).
Transthoracic needle biopsy (TTNB)
Classification of lung biopsies may be done according to the method of access (percutaneous, during bronchoscopy, or open lung operation) or by the reason for biopsy (sampling of diffuse lung disease or obtaining tissue from a mass when malignancy is suspected). Sometimes percutaneous biopsy is also defined by the tissue type obtained (cytological or histological).
TTNB is integral in the diagnosis and treatment of many thoracic diseases, and is an important alternative to more invasive surgical procedures (2).
A fine needle aspiration (FNA) gives cytological specimens which can be used to withdraw cells from a suspected cancer, although these needles tend to be of narrow bore, cutting needles that produce histological specimens can also be of similar gauge. Large CNB is a technique that uses cutting needles that enables the interventional radiologist to obtain a larger biopsy sample (1).
Fine needle aspiration biopsy (FNAB) is also known as FNA cytology or FNA. Aspiration needles are usually 20-25 gauge and provide material for cytological and microbiological examination. Cutting or core biopsy needles provide small linear tissue sections suitable for histological evaluation. These needles are usually larger in caliber than aspiration needles; however, nowadays small caliber (18-20 gauge) automated cutting needles are available. Most CT-guided lung biopsies in earlier reports were performed by fine-needle aspiration and were useful in differentiating malignant from benign lesions. More recently, the use of tissue core biopsy utilizing an automated cutting needle has been implemented in an attempt to further enhance diagnostic accuracy (10,11).
The diagnostic accuracy of aspiration biopsy is almost as good as core biopsy for the diagnosis of malignant lesions, especially if an onsite cytopathologist is present. However, for the diagnosis of benign lesions and lymphoma, core biopsy is preferred (1,12).
In earlier years, it has been reported that the diagnostic accuracy of TTNB is greater than 80% for benign disease and greater than 90% for malignant disease. Westcott and colleagues reported an overall sensitivity of 93% and specificity of 100% in pulmonary nodules of less than 15 mm diameter (13,14).
Although rates for pneumothorax are similar for aspiration needles and cutting biopsy needles, a slightly higher incidence of pulmonary bleeding is reported with cutting biopsy needles (1).
Advantages of needle biopsy are many and they include: with image guidance, the abnormality can be biopsied while important nearby structures (such as blood vessels and vital organs) can be seen and avoided. The patient is spared the pain, scarring and complications associated with open surgery. Recovery times are usually shorter and patients can more quickly resume normal activities (1). To further everyday use of this procedure, solutions to the complications inherent in its performance, the commonest being pneumothorax, are necessary (8,9).
Imaging guidance and procedure technique
Transthoracic needle lung biopsy is performed with the aid of equipment that creates a computer-generated image and allows radiologists to see an area inside the body from various angles. This “stereotactic” equipment helps them pinpoint the exact location of the abnormal tissue.
Modalities commonly employed for imaging-guided percutaneous needle biopsy include moving X-ray technique (fluoroscopy), ultrasound or magnetic resonance (MR), conventional CT, and helical CT, which has become more widely used.
Ultrasound is useful only where the tissue mass is in contact with the chest wall since the ultrasound beam does not pass through air and, hence, the aerated lung. Magnetic resonance imaging (MRI) currently has a limited use because of expense, difficulty accessing the patient within the magnet, the relatively poor visualization of lung lesions, and difficulties with ferromagnetic instruments within the magnetic field (1). CT fluoroscopy is technical advancement which enables real-time visualization of a lesion during needle manipulation. It is reported to be more accurate than conventional CT in diagnosing pulmonary lesions, with a significant reduction in complication rates (15). This technique is especially useful for targeting small lung lesions, juxtraphrenic lesions, and patients with poor breath holding capacity. It was developed most recently and is not universally available, and it has simplified the process and decreased the time requirements of CT-guided needle biopsies (16-18). It offers promising advantages and may permit accurate and rapid procedures, particularly in benign lesions. The decision as to which method to use should be tailored to each patient, and is preferably reached by a team consisting of pulmonary physicians, chest surgeons, oncologists, cytologists, and radiologists (3).
Biopsy technique
Ideally, the technique must not only be able to diagnose malignancy but also to make a definite diagnosis if the lesion is benign (1).
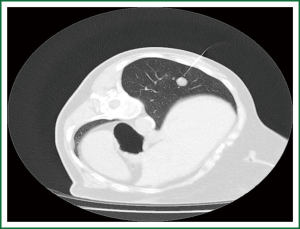
After appropriate patient positioning, a radiopaque marker or grid is placed on the patient’s skin over the area of interest to focus the optimal access point. A short spiral CT scan of the region of interest is obtained, and from these images, an appropriate table position and needle trajectory are chosen. The shortest straight pathway from the skin to the lesion is preferred over a longer oblique pathway. The depth from the skin entry site to the lesion is then measured (19) (Figure 1).
During the procedure, patients must be able to follow breathing instructions. Needle choice depends mostly upon lesion characteristics and location. There are techniques of needle tip repositioning that can be quite helpful for obtaining diagnostic material from lung lesions, particularly small nodules (2,4).
The number of passes needed per procedure has not been defined. Most operators perform at least two. Variables to consider are: the difficulty of the procedure, complications arising from each biopsy, the quality of the specimen obtained, the characteristics of the lesion biopsied, and the need for specimens for cytological, histological and microbiological examination (20,21).
A strong working relationship with pathologists experienced in lung cytology is a vital element of any successful biopsy program. Techniques available to the pathologist allow for quick and decisive determination of the adequacy of the aspirated specimen and help guide the radiologist performing the procedure. Newer cytopathologic techniques help the pathologist make more complex diagnoses from the aspirated material. Finally, techniques used to minimize complications should be considered by the operator before the performance of the biopsy (4).
Indications and contraindications
TTNB is used to investigate any solid or cystic lesion between the chest wall and the mediastinum, provided it is accessible to the needle (22).
Indications for TTNB are pulmonary lesion inaccessible to bronchoscopy, or in which prior bronchoscopic biopsy was nondiagnostic and mediastinal or pleural mass. The indications also include multiple nodules in a patient not known to have malignancy or who has had a prolonged remission. Persistent infiltrates, either single or multiple, for which no diagnosis has been made by sputum or blood culture, serology or bronchoscopy. As well as hilar mass following negative bronchoscopy. Although TTNB can be used to investigate interstitial lung disease (particularly in patients with focal areas of consolidation such as cryptogenic organizing pneumonia), transbronchial and thoracoscopic or open lung biopsy are preferred to minimize the risk of pneumothorax and to obtain larger and more representative diagnostic samples, particularly by open lung biopsy (23,24).
Pre-procedure evaluation should be consideration of the various factors that influence suitability and degree of risk, including site and size of the nodule/mass and its relationship to structures that must be avoided: vessels, bleb, bullae,fissures. On cavitating lesions the needle must be targeted at the periphery (19). A recent platelet count and complete coagulogram should be available before the procedure to exclude any coagulation abnormality. A good quality baseline contrast-enhanced CT should always be done and reviewed before the procedure for the lesion size, location, lesion vascularity, and important structures located in the biopsy path (12,25,26).
The risks and benefits of the procedure and knowledge of the wishes of the patient will enable the management decision to be tailored to the needs of the individual.
The most important contraindication are: poor respiratory function or reserve, abnormal coagulation indices. The condition of the underlying lung is evaluated for the presence of emphysema and bullous lesions. A violent uncooperative patient is one of the major contraindications for TTNB. Other relative contraindications include bleeding diatheses, severe bullous emphysema, contralateral pneumonectomy, hydatid cyst, pulmonary hypertension, highly vascular lesion, and the inability to tolerate a pneumothorax. If the patient has cough, cough suppressants should be prescribed (12,25-27).
Recommendations from the British Thoracic Society published in 2003 are: the balance of benefit against risk for TTNB should be assessed by a multidisciplinary team with a respiratory physician and radiologist as a minimum. All patients should have recent pulmonary function tests (spirometry) before needle biopsy. Patients with FEV1 <35% predicted should not undergo needle biopsy without further assessment by the multidisciplinary team (1).
Complications
Despite observing all precaution, complications may occur, early detection of complications and timely management is important. Complications of TTNB include pneumothorax, hemoptysis, hemothorax, infection, and air embolism, with the most common complication as pneumothorax. Definition of pneumothorax (PNTX) is presence of gas in the pleural space. Although it can be spontaneous and traumatic, most pneumothoraces are iatrogenic and caused by a physician during surgery, central line placement, lung biopsy, or bronchoscopy (2,28).
Risk factors for the development of biopsy-related pneumothorax include the presence of chronic obstructive pulmonary disease (COPD), small lesion size, a long needle path, repeated pleural puncture, and the absence of a history of ipsilateral surgery (27).
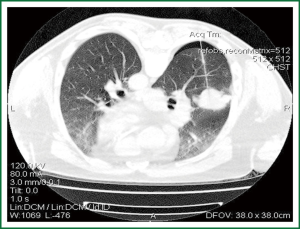
Pneumothoraces can occur during or immediately after the procedure, which is why it is important to perform a CT scan of the region following removal of the needle (2). The incidence of pneumothorax in patients undergoing TTNB has been reported to be from 9% to 54%, according to reports published in the past ten years (11,16,17,29-31), with an average of around 20% (32) (Figure 2).
Many researchers have investigated risk factors for this problem. It is thought that the risk of pneumothorax increases with increasing needle diameter, but some studies suggest no difference in pneumothorax rate between larger and smaller needles (33,34).
Factors increasing the risk of pneumothorax also include increased patient age, increased depth of lesion, increased time of needle across the pleura, and traversal of a fissure (17,29,33).
When comparing needle biopsy techniques, however, the incidence of pneumothorax is essentially the same for single-needle FNA, coaxial-needle FNA, and core biopsy using automated biopsy devices (35-37).
The frequency and risk factors associated with pneumothorax in the study of Takuji Y et al. (28), suggested that the subjects and the operator skill in CT-guided lung biopsies did not differ appreciably from earlier studies (11,16,37,38), and in this study (28) emphysema was detected on CT in 18 of 46 procedures (39.1%) complicated by pneumothorax, while 22 of 88 procedures (25.0%) were not complicated by pneumothorax, which was not statistically significant.
Many authors prefer core biopsy, as it provides tissue sample and permits more laboratory testing, such as electron microscopy, immunohistochemistry and analysis of tumor-markers, factors that enhance diagnostic specificity. Priola et al. stated in their study on 73 patients, that large cut needles of 14G have a higher complication rate, while small-sized needles (18G) do not increase the complication rate compared to FNAB. It is also stated that 18G core biopsy has a higher value than FNAB, for the confirmation of a benign lesion, characterization of malignant cell types, especially in lymphoproliferative diseases (lymphoma), metastatic lung cancer and mesothelioma (35,39,40).
According to some reports, core biopsy is superior to FNAB in diagnosis of benign thoracic lesions, mediastinal tumors, determination of cancer cell-type and predicting cancer-negative findings. As confirmed in large study by Beslic et al. in 2012 (41). On 242 patients, by performing core biopsy and getting adequate samples for patohistological diagnosis, it is possible to increase the rate of definitive diagnosis in benign lesions from 52% to 91% (35,40,41).
Computed tomography-guided transthoracic needle aspiration and core needle biopsy were performed in 105 patients, in a Turkish study published in November 2013, on peripheral pulmonary lesions. The most frequent complication during the transthoracic procedures was pneumothorax (10%), and there was no statistically significant difference in complication rates between the two procedures (42).
Known patient-related risk factor for pneumothorax is the presence of COPD and emphysema. Most studies report a 20% to 25% incidence of pneumothorax after TTNB of the lung, with higher rates when patients have moderate-to-severe emphysema or with core biopsy. In 1999, Cox et al. (38) found that increased rate of pneumothorax was correlated with presence of emphysema (P=0.01), also patient with emphysema were three times more likely to require a chest tube placement. Fish et al. (43) found that the pneumothorax rate was 46% in patients with pulmonary function test and chest radiographs showing obstructive airway disease compared with 7% in patients with normal findings for both criteria. In 2006, Heck et al. (44) confirmed that the presence of emphysema and obstructive pulmonary disease, strongly correlate with the occurrence of pneumothorax and the need for drainage, and the risk of pneumothorax were significantly greater if the lesions were completely surrounded by aerated lung.
Also the association of obstructive lung disease with chest tube placement was confirmed by Jane Ko et al. (45) on 150 coaxial computed tomography (CT)-guided lung biopsies. There were 58 (39%) pneumothoraces with eight (5%) biopsies resulting in chest tube placement. Emphysema along the needle path, pulmonary function tests showing ventilatory obstruction, and lesions along fissures predisposed patients to chest tube placement (P<0.05). Pleural thickening and prior surgery were associated with lower pneumothorax rates (P<0.05). Also investigated in this research was dwell time, the time between pleural puncture and needle removal. Longer dwell time did not correlate with pneumothorax. It was noted as well that shallow pleural puncture angle did increase the pneumothorax rate.
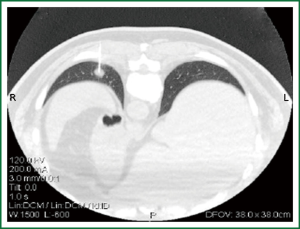
Mixed opinions exist on the effect of the nodule size on diagnostic accuracy of percutaneous lung biopsies and pneumothorax frequency. The diagnostic accuracy rates and yield reported in the literature vary from 65% to 96% because of differences in techniques, average nodule size, and the modality used for imaging guidance (46-49). Kothary et al. (49) reviewed studies that linked nodule size and accuracy when performing CT-guided percutaneous needle biopsy and reported that, in research of Ernst et al. (50) the prevalence of pneumothorax was 34.5% (48 of 139 patients); however, only seven patients (5%) required a thoracostomy tube and were hospitalized overnight. Prevalence of a pneumothorax was similar in patients with small nodules and in patients with large nodules (35% and 34%, respectively), and there was no statistically significant difference between the two groups overall (P=0.93) or the requirement for a thoracostomy tube (P=0.32). Geraghty et al. (34) did a retrospective review of 846 consecutive CT-guided TTNB procedures, with pneumothorax frequency of 26.7% (226 of 846 patients). Analyses at each threshold (smaller than 1 cm and larger than 1 cm, and of smaller than 2 cm and larger than 2 cm) failed to demonstrate a significant difference in the rates of pneumothorax between smaller and larger lesion groups. When multivariate analysis was performed to compare lesion size, needle size (18- or 19-gauge outer stabilizing needle), and pneumothorax rate, there was no significant difference in the rate of pneumothorax for small versus large lesions (33) (Figure 3).
On the contrary, in a study of Joseph Cox et al., (37) an increased rate of pneumothorax was correlated with smaller lesion size (P=0.001), in 346 patients.
Poe and Tobin (22) reported no difference in diagnostic yield between small and large nodules. However, lung nodules were considered small if they measured <4 cm, with a preponderance of nodules >4 cm. More recent literature on CT-guided lung biopsies demonstrates a definite difference in the diagnostic yield and accuracy when biopsies are performed on small nodules (≤2 cm) (46,47). Ohno et al. (46) also used a size criterion of ≤2 cm and reported a statistically significant drop in diagnostic accuracy to 77%. Despite technical challenges often associated with these small nodules, percutaneous transthoracic lung biopsy (FNAB and CNB) still is the modality of choice to establish tissue diagnosis in a safe and accurate manner (49).
Also in the previously mentioned study by Geraghty et al. (33) on 846 consecutive CT-guided TTNB procedures, patient age had a significant effect on pneumothorax rate. A significant increase in the rate of pneumothorax was seen in patients older than 60 years (P<0.02) and older than 70 years (P<0.01) compared with their younger counterparts. There was no significant difference in the rate of pneumothorax for age comparisons in patients 50 years or younger and those older than 75 years.
On the contrary, some authors disagree that gender, age, and the number of pleural passes correlates with the incidence of pneumothorax (16,30,37).
One of the reports supporting no correlation between the number of passes with the incidence of pneumothorax was of Halloush et al., (30) that reviewed 150 cases. 11.6% (16/138) developed pneumothorax, only three of 138 (2%) required a chest tube for treatment. The number of passes was identified in 118 patients (85.5%), and it did not have a statistically significant association with the development of a pneumothorax (P=0.747). This was also confirmed by Cox and Chiles (37).
Still, many authors have found that multiple punctures have been associated with increased chances of pneumothorax and procedure failure. So to minimize complications, one should aim to puncture the pleura once only. Although the development of pneumothorax usually required abandoning the procedure, CT guided biopsy of lung lesions could be completed under stable pneumothorax if the lesion was close to the pleura (51,52).
Management of complications
The majority of cases of pneumothorax developing after needle biopsy resolve spontaneously, but a few intractable cases require chest tube placement.
A large review of all studies published in the last ten years, revealed a frequency of chest tube placement ranging from 2.0% (33,37) to 15.0% (11) of all biopsy procedures, and 5-18% of cases complicated by pneumothorax (2,11,16, 31,32,37,38,53,54).
In comparison, frequency in Takuji et al. (28) study on 134 patients was at the low end, with only 2.2% (3 of 134) and 6.5% (3 of 46) of biopsies and pneumothoraces requiring chest tubes, respectively. Also in large study, in 2001, by Dennie at al. (27) on 506 cases, confirmed that only minority of patients, 2% to 5%, required a chest tube or catheter for drainage of the pneumothorax.
In most cases, patients treated with chest tubes typically have been admitted to the hospital. Recently, however, CT-guided lung biopsy has been performed in the outpatient setting, as a way of minimizing costs and inconvenience to the patient. Thus, efforts are required to decrease the necessity of chest tube placement for pneumothoraces that result from complications incurred from the biopsy (28).
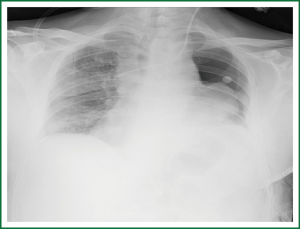
In recent study by Yaffe D et al. which was published in September 2013, among 129 CT image-guided percutaneous biopsies, both FNAB and CNB, pneumothorax was detected by CT scan in 54 (42%). In the cases with symptomatic minimal pneumothorax and in all patients with pneumothorax >10 mm, immediate, simple, manual aspiration was performed—in 27 of 129 procedures (21%). Ipsilateral opposite-side aspiration was performed when simple aspiration failed, in seven cases (26%) and was successful in 86% of the cases. Two of 129 procedures (1.55%) required chest tube placement. It appears, at least from the results that study, that the immediate aspiration of a pneumothorax, irrespective of the presence of symptoms, caused by CT-guided needle lung biopsy is effective at preventing progression of the pneumothorax, thus avoiding chest tube placement (28,55) (Figure 4).
Many measures can be taken to help prevent the development of a pneumothorax and reduce the number of pneumothoraces requiring chest tube placement. Patients are instructed not to move, talk, cough, or breathe deeply during and immediately after the procedure. The use of a coaxial technique allows multiple specimens to be obtained with a single pleural puncture. To reduce the number of pleural punctures, interlobar fissures should be avoided. Careful planning is necessary to traverse the least amount of aerated lung, without puncturing bullae or pneumatoceles if possible. Infusion of normal saline to expand the extrapleural space and displace the adjacent lung can be performed to avoid traversing aerated lung when biopsying a subpleural lesion (56). Oxygen can be administered through a nasal cannula during and after the procedure to speed the resorption of the pneumothorax, if one does develop (52). Finally, after removal of the introducer needle after the biopsy, patients should immediately be positioned in a “biopsy side down” position for at least 1 hour immediately after completing the procedure. The patient should remain in the recumbent position for the next four hours. Although some of the authors have reported no benefits of putting patients in the “biopsy down position”, this has been advocated by many others (38,52,57). The rationale behind this maneuver is reducing the aeration of the punctured lung, resulting in the reduction of alveolar-to-pleural pressure gradient at the puncture site and accumulation of hemorrhagic fluid around needle path, with an early sealing of the puncture tract (38,58).
Second most common complication after TTNB is hemorrhage. Less than 1% of patients experience significant (30 to 50 mL) hemoptysis. Hemoptysis was reported in 5% to 15% of cases in study done by Dennie et al. (27) on 506 patients, with most patients having minimal hemoptysis. Studies show that most pulmonary hemorrhage is self-limited, with a conclusion that significant hemorrhage can be avoided by careful attention to technique (27).
Wiener et al. (59) did a cross-sectional analysis on over 15,000 patients who had TTNB of a pulmonary nodule, to determine population-based estimates of risks for complications after TTNB of a pulmonary nodule. They reported that although hemorrhage was rare, complicating 1.0% of biopsies, 17.8% of patients with hemorrhage required a blood transfusion. In contrast, the risk for any pneumothorax was 15.0%, and 6.6% of all biopsies resulted in pneumothorax requiring a chest tube. Compared with patients without complications, those who experienced hemorrhage or pneumothorax requiring a chest tube had longer lengths of stay (P<0.001) and were more likely to develop respiratory failure requiring mechanical ventilation (P=0.020). Coming up from their results was that patients aged 60 to 69 years (as opposed to younger or older patients), smokers, and those with chronic obstructive pulmonary disease had higher risk for complications.
Conclusions
CT-guided TTNB is generally regarded as a safe procedure with limited morbidity and extremely rare mortality. Its findings are reproducible with high interobserver agreement. Immediate adequacy evaluation and triage by a pathologist guarantees adequate sample in most instances. Pneumothorax remains the most frequent complication of TNNB, and a tube thoracostomy is occasionally required for treatment. The reported frequency of pneumothorax for CT-guided procedures varies from 8% to 64%.
CT-guided FNAB is a useful tool in the diagnosis and management of small pulmonary nodules, despite the lower diagnostic accuracy and higher complication rate than those of larger pulmonary lesions. Moreover, there is little evidence in the literature that needle gauge affects the complication rate within the size range of the smaller needles available for lung biopsy; needles larger than 18-gauge are considered a risk for causing both bleeding and pneumothorax.
Immediate simple, percutaneous aspiration of iatrogenic a pneumothorax, irrespective of the presence of symptoms, caused by CT-guided needle lung biopsy, proved successful in 74% of patients needing treatment, and it has been recommended at preventing progression of the pneumothorax, thus avoiding chest tube placement. Some authors also propose a new method of ipsilateral opposite-side aspiration, as it offers a solution for patients who remain with resistant pneumothorax after simple aspiration.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Manhire A, Charig M, Clelland C, et al. Guidelines for radiologically guided lung biopsy. Thorax 2003;58:920-36. [PubMed]
- Birchard KR. Transthoracic Needle Biopsy. Semin Intervent Radiol 2011;28:87-97. [PubMed]
- Shaham D. Semi-invasive and invasive procedures for the diagnosis and staging of lung cancer. I. Percutaneous transthoracic needle biopsy. Radiol Clin North Am 2000;38:525-34. [PubMed]
- Yankelevitz DF, Vazquez M, Henschke CI. Special techniques in transthoracic needle biopsy of pulmonary nodules. Radiol Clin North Am 2000;38:267-79. [PubMed]
- Komiya T, Kusunoki Y, Kobayashi M, et al. Transcutaneous needle biopsy of the lung. Acta Radiol 1997;38:821-5. [PubMed]
- Westcott JL, Rao N, Colley DP. Transthoracic needle biopsy of small pulmonary nodules. Radiology 1997;202:97-103. [PubMed]
- Greif J, Marmur S, Schwarz Y, et al. Percutaneous core needle biopsy vs. fine needle aspiration in diagnosing benign lung lesions. Acta Cytol 1999;43:756-60. [PubMed]
- Brown KT, Brody LA, Getrajdman GI, et al. Outpatient treatment of iatrogenic pneumothorax after needle biopsy. Radiology 1997;205:249-52. [PubMed]
- Gurley MB, Richli WR, Waugh KA. Outpatient management of pneumothorax after fine-needle aspiration: economic advantages for the hospital and patient. Radiology 1998;209:717-22. [PubMed]
- Boiselle PM, Shepard JA, Mark EJ, et al. Routine addition of an automated biopsy device to fine needle biopsy of the lung: a prospective assessment. AJR Am J Roentgenol 1997;169:661-6. [PubMed]
- Klein JS, Salomon G, Stewart EA. Transthoracic needle biopsy with a coaxially placed 20-gauge automated cutting needle: results in 122 patients. Radiology 1996;198:715-20. [PubMed]
- Cham MD, Henschke CI, Yankelevitz DF. Biopsy procedures of the lung, mediastinum, and chest wall. Handbook of interventional radiologic procedures. Wolters Kluwer/Lippincott Williams & Wilkins, 2011:478-85.
- Westcott JL, Rao N, Colley DP. Transthoracic needle biopsy of small pulmonary nodules. Radiology 1997;202:97-103. [PubMed]
- Lederer W, Schlimp CJ, Glodny B, et al. Air embolism during CT-guided transthoracic needle biopsy, BMJ Case Rep 2011;2011. pii: bcr0420114113.
- Paulson EK, Sheafor DH, Enterline DS, et al. CT fluoroscopy-guided interventional procedures: techniques and radiation dose to radiologists. Radiology 2001;220:161-7. [PubMed]
- Laurent F, Latrabe V, Vergier B, et al. Percutaneous CT- guided biopsy of the lung: comparison between aspiration and automated cutting needle using a coaxial technique. Cardiovasc Intervent Radiol 2000;23:266-72. [PubMed]
- Hirose T, Mori K, Machida S, et al. Computed tomographic fluoroscopy-guided transthoracic needle biopsy for diagnosis of pulmonary nodules. Jpn J Clin Oncol 2000;30:259-62. [PubMed]
- Daly B, Templeton PA. Real-time CT fluoroscopy: evolution of an interventional tool. Radiology 1999;211:309-15. [PubMed]
- Walker TG. Interventional Procedures. Lippincott Williams & Wilkins, 2012. ISBN:1931884862.
- Williams SM, Gray W, Gleeson FV. Macroscopic assessment of pulmonary fine needle aspirate biopsies: correlation with cytological diagnostic yield. Br J Radiol 2002;75:28-30. [PubMed]
- Hahn PF, Eisenberg PJ, Pitman MB, et al. Cytopathologic touch preparations (Imprints) from core needle biopsies: accuracy compared with that of fine-needle aspirates. AJR Am J Roentgenol 1995;165:1277-9. [PubMed]
- Poe RH, Tobin RE. Sensitivity and specificity of needle biopsy in lung malignancy. Am Rev Respir Dis 1980;122:725-9. [PubMed]
- Li Y, Du Y, Yang HF, et al. Re: CT-guided percutaneous core needle biopsy for small (≤20 mm) pulmonary lesions. Clin Radiol 2013;68:e354. [PubMed]
- The diagnosis, assessment and treatment of diffuse parenchymal lung disease in adults. Thorax 1999;54:S1-14. [PubMed]
- Lal H, Neyaz Z, Nath A, et al. CT-Guided percutaneous biopsy of intrathoracic lesions. Korean J Radiol 2012;13:210-6. [PubMed]
- Cham MD, Lane ME, Henschke CI, et al. Lung biopsy: special techniques. Semin Respir Crit Care Med 2008;29:335-49. [PubMed]
- Dennie CJ, Matzinger FR, Mariner JR, et al. Transthoracic needle biopsy of the lung: results of early discharge in 506 outpatients. Radiology 2001;219:247-51. [PubMed]
- Yamagami T, Nakamura T, Iida S, et al. Management of pneumothorax after percutaneous CT-guided lung biopsy. Chest 2002;121:1159-64. [PubMed]
- Choi CM, Um SW, Yoo CG, et al. Incidence and risk factors of delayed pneumothorax after transthoracic needle biopsy of the lung. Chest 2004;126:1516-21. [PubMed]
- Halloush RA, Khasawneh FA, Saleh HA, et al. Fine needle aspiration cytology of lung lesions: a clinicopathological and cytopathological review of 150 cases with emphasis on the relation between the number of passes and the incidence of pneumothorax. Cytopathology 2007;18:44-51. [PubMed]
- Ng YL, Patsios D, Roberts H, et al. CT-guided percutaneous fine-needle aspiration biopsy of pulmonary nodules measuring 10 mm or less. Clin Radiol 2008;63:272-7. [PubMed]
- Freund MC, Petersen J, Goder KC, et al. Systemic air embolism during percutaneous core needle biopsy of the lung: frequency and risk factors. BMC Pulm Med 2012;12:2. [PubMed]
- Geraghty PR, Kee ST, McFarlane G, et al. CT-guided transthoracic needle aspiration biopsy of pulmonary nodules: needle size and pneumothorax rate. Radiology 2003;229:475-81. [PubMed]
- Swischuk JL, Castaneda F, Patel JC, et al. Percutaneous transthoracic needle biopsy of the lung: review of 612 lesions. J Vasc Interv Radiol 1998;9:347-52. [PubMed]
- Connor S, Dyer J, Guest P. Image-guided automated needle biopsy of 106 thoracic lesions: a retrospective review of diagnostic accuracy and complication rates. Eur Radiol 2000;10:490-4. [PubMed]
- Laurent F, Michel P, Latrabe V, et al. Pneumothoraces and chest tube placement after CT-guided transthoracic lung biopsy using a coaxial technique: incidence and risk factors. AJR Am J Roentgenol 1999;172:1049-53. [PubMed]
- Cox JE, Chiles C, McManus CM, et al. Transthoracic needle aspiration biopsy: variables that affect risk of pneumothorax. Radiology 1999;212:165-8. [PubMed]
- Collings CL, Westcott JL, Banson NL, et al. Pneumothorax and dependent versus nondependent patient position after needle biopsy of the lung. Radiology 1999;210:59-64. [PubMed]
- Priola AM, Priola SM, Cataldi A, et al. CT-guided percutaneous transthoracic biopsy in the diagnosis of mediastinal masses: evaluation of 73 procedures. Radiol Med 2008;113:3-15. [PubMed]
- Montaudon M, Latrabe V, Pariente A, et al. Factors influencing accuracy of CT-guided percutaneous biopsies of pulmonary lesions. Eur Radiol 2004;14:1234-40. [PubMed]
- Beslic S, Zukic F, Milisic S. Percutaneous transthoracic CT guided biopsies of lung lesions; fine needle aspiration biopsy versus core biopsy. Radiol Oncol 2012;46:19-22. [PubMed]
- Tuna T, Ozkaya S, Dirican A, et al. Diagnostic efficacy of computed tomography-guided transthoracic needle aspiration and biopsy in patients with pulmonary disease. Onco Targets Ther 2013;6:1553-7. [PubMed]
- Fish GD, Stanley JH, Miller KS, et al. Postbiopsy pneumothorax: estimating the risk by chest radiography and pulmonary function tests. AJR Am J Roentgenol 1988;150:71-4. [PubMed]
- Heck SL, Blom P, Berstad A. Accuracy and complications in computed tomography fluoroskopy- guided needle biopsies of lung masses. Eur Radiol 2006;16:1387-92. [PubMed]
- Ko JP, Shepard JO, Drucker EA, et al. Factors influencing pneumothorax rate at lung biopsy: are dwell time and angle of pleural puncture contributing factors? Radiology 2001;218:491-6. [PubMed]
- Ohno Y, Hatabu H, Takenaka D, et al. CT-guided transthoracic needle aspiration biopsy of small (< or = 20 mm) solitary pulmonary nodules. AJR Am J Roentgenol 2003;180:1665-9. [PubMed]
- Wallace MJ, Krishnamurthy S, Broemeling LD, et al. CT-guided percutaneous fine-needle aspiration biopsy of small (< or = 1-cm) pulmonary lesions. Radiology 2002;225:823-8. [PubMed]
- Ohno Y, Hatabu H, Takenaka D, et al. Transthoracic CT-guided biopsy with multiplanar reconstruction image improves diagnostic accuracy of solitary pulmonary nodules. Eur J Radiol 2004;51:160-8. [PubMed]
- Kothary N, Lock L, Sze DY, et al. Computed tomography-guided percutaneous needle biopsy of pulmonary nodules: impact of nodule size on diagnostic accuracy. Clin Lung Cancer 2009;10:360-3. [PubMed]
- Ernst A, Silvestri GA, Johnstone D, et al. Interventional Pulmonary Procedures: Guidelines from the American College of Chest Physicians. Chest 2003;123:1693-717. [PubMed]
- Tsai IC, Tsai WL, Chen MC, et al. CT-guided core biopsy of lung lesions: a primer. AJR Am J Roentgenol 2009;193:1228-35. [PubMed]
- Wu CC, Maher MM, Shepard JA. Complications of CT-guided percutaneous needle biopsy of the chest: prevention and management. AJR Am J Roentgenol 2011;196:W678-82. [PubMed]
- Yeow KM, Su IH, Pan KT, et al. Risk factors of pneumothorax and bleeding: multivariate analysis of 660 CT-guided coaxial cutting needle lung biopsies. Chest 2004;126:748-54. [PubMed]
- Khan MF, Straub R, Moghaddam SR, et al. Variables affecting the risk of pneumothorax and intrapulmonal hemorrhage in CT-guided transthoracic biopsy. Eur Radiol 2008;18:1356-63. [PubMed]
- Yaffe D, Shitrit D, Gottfried M, et al. Ipsilateral Opposite-Side Aspiration in Resistant Pneumothorax After CT Image Guided Lung Biopsy: Complementary Role After Simple Needle Aspiration. Chest 2013;144:947-51. [PubMed]
- Klose KC. CT-guided large-bore biopsy: extrapleural injection of saline for safe transpleural access to pulmonary lesions. Cardiovasc Intervent Radiol 1993;16:259-61. [PubMed]
- Wallace AB, Suh RD. Percutaneous transthoracic nnedle biopsy:special considerations and techniques used in lung transplant recipients. Semin Intervent Radiol 2004;21:247-58. [PubMed]
- Kinoshita F, Kato T, Sugiura K, et al. CT-guided transthoracic needle biopsy using a puncture site-down positioning technique. AJR Am J Roentgenol 2006;187:926-32. [PubMed]
- Wiener RS, Schwartz LM, Woloshin S, et al. Population-based risk for complications after transthoracic needle lung biopsy of a pulmonary nodule: an analysis of discharge records. Ann Intern Med 2011;155:137-44. [PubMed]