Comparison of open surgical versus hybrid endovascular repair for descending thoracic aortic aneurysms with distal arch involvement
Introduction
Isolated pathology of the descending aorta at present is managed safely and effectively through open or thoracic endovascular aortic repair (TEVAR). Open repair is an established and durable therapeutic option (1,2), whereas TEVAR is a newer alternative surgical repair option that offers acceptable results for high-risk patients (3-5). However, disease of the descending aorta that extends proximally to the arch is inherently more complex. There are hazards associated with accessing the arch during open repair, and endovascular repair requires a hybrid technique.
Open surgical repair of lesions involving the arch calls for total circulatory arrest (TCA), cerebral perfusion, and manipulation of arch atheroma, all contributing to the high risk of this procedure. A hybrid technique that combines supra-aortic vascular debranching with stent grafting of the aortic arch has been introduced as an attractive therapeutic alternative. Still, the short- and long-term outcomes of hybrid repairs remain unclear due to technical difficulties and complications imposed by supra-aortic vascular angulation and influences (6-10).
Several studies to date have examined both hybrid and conventional open repairs (via sternotomy) in managing arch aneurysms, although none have similarly addressed thoracotomy and hybrid repairs of descending thoracic aorta (DTA) that include the distal arch (11-13). A preferred approach has therefore yet to be determined. We thus sought to compare outcomes of conventional open (thoracotomy) and hybrid arch repairs in patients with descending aortic aneurysms, focusing on those that involve the distal arch.
Methods
Patient population
Between January 2005 and December 2016, 125 patients underwent descending aortic surgical repairs inclusive of the distal arch at the Yonsei Cardiovascular Hospital of Yonsei University Health System in Seoul, Republic of Korea. The patients were grouped by procedure, 79 (63.2%) undergoing conventional open repair via thoracotomy (open group) and 46 (36.8%) subjected to hybrid aortic repair (hybrid group). Surgeries entailing total arch replacement by elephant trunk technique (with sternotomy) and Zone 0 hybrid arch repairs were grounds for exclusion.
The hybrid group included those patients whose hybrid arch repairs incorporated supra-aortic branch revascularization and involved at least one common carotid artery bypass and simultaneous or staged endovascular thoracic stent grafting. As a prerequisite for successful stent-graft placement, a proximal landing zone of healthy and non-dissected aorta (or polyester graft) of at least 20 mm along the curvature of aortic arch and a diameter <40 mm at the level of proximal landing zone were stipulated. The decision for hybrid arch repair was based on discussions between cardiac and vascular surgeons, interventional cardiologists, and vascular radiologists.
Operative techniques
Conventional open repair
Each patient underwent hemi-arch and descending aortic replacement via left thoracotomy. Surgical access was via left 4th or 5th intercostal space. Left femoral artery and vein were routinely cannulated to establish cardiopulmonary bypass. Left ventricular venting was achieved via left low pulmonary vein or left ventricular apex, and TCA was induced at a core body temperature of 20 °C, without cerebral perfusion or cardioplegia. We performed routine electroencephalogram (EEG) monitoring, which served as the basis for initiating TCA. Bladder temperature was used as a core index for systemic organ protection.
Hybrid arch repair
The operations were performed under general endotracheal anesthesia with continuous monitoring by transesophageal echocardiography (TEE), rSO2, and arterial pressure. Five patients (12.8%) underwent one-stage procedures. Two-stage procedures, performing TEVAR after full recovery from cervical debranching, were conducted in another 32 patients (87.2%). Cervical debranching was achieved through cervicotomy, enabling supra-tracheal right-to-left common carotid artery bypass using an 8-mm Gore-Tex or Dacron graft. A carotid clamp was placed under rSO2 monitoring, but in no instance was a carotid shunt required. All TEVAR procedures were confined to a hybrid room, furnished with both endovascular and surgical equipment. The common femoral artery was regularly used for access in placing of stent grafts.
Classification
Aortic arch zones were designated according to Ishimaru14 as follows: Zone 0, ascending aorta proximal to innominate artery; Zone 1, aortic arch between innominate and left common carotid artery; Zone 2, aortic arch between left common carotid artery and left subclavian artery (LSCA); Zone 3, proximal DTA distal to LSCA; and Zone 4, mid-DTA (14).
Endpoints and definitions
The primary study endpoints were early (in-hospital) and late mortality. Operative morbidities, including neurologic complications and reinterventions, constituted secondary endpoints. A reintervention was defined as any surgical or endovascular procedure for any complication related to the aorta in the aftermath of the primary procedure. Spinal cord ischemia (SCI) corresponded with any newly acquired lower-extremity deficit unrelated to an intracerebral event. SCIs were considered permanent if symptoms were not completely resolved at the time of discharge. Stroke was marked by any new global or focal neurologic deficit lasting more than 24 h, with an acute lesion observed on brain imaging. Technical success was equated with successful introduction and deployment of a device in the absence of mortality, surgical conversion, type I or III endoleaks, or graft limb obstruction.
Data collection and follow-up monitoring
Preoperative and perioperative data were collected prospectively from cardiac and vascular research databases at our institution. More detailed data on clinical outcomes and long-term survival were obtained by reviewing hospital charts or through telephone interviews. The Institutional Review Board of Yonsei University College of Medicine approved the study (Yonsei Institutional Review Board No. 4-2014-0810), waiving the customarily required individual patient consent. The protocol called for follow-up clinical examinations and computerized tomography (CT) scans done before discharge, at 6 months, and then annually thereafter.
Statistical analyses
All data were expressed as mean ± standard deviation (SD) or frequency and percentage. Continuous variables were compared via Student’s t-test, using x2 or Fisher’s exact test to compare categorical variables. To reduce the impact of selection bias and potential confounding factors, we adjusted the patient characteristics by using weighted logistic regression analysis and inverse-probability-of-treatment weighting (IPTW). Goodness-of-fit was assessed through Hosmer and Lemeshow chi-square test. Long-term survival and freedom of reintervention curves were estimated by Kaplan-Meier method, using log-rank test to compare differences. In all statistical testing, significance was defined as a two-tailed P value <0.05. Statistical analyses were performed with SAS version 9.4 (SAS Institute, Cary, NC, USA) and R, version 3.3.0 (The R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient characteristics
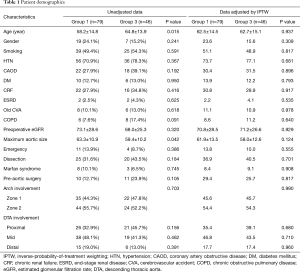
Patient demographics and clinical risk factors of the two groups are summarized in Table 1. Prior to adjustment, patient age in the hybrid (vs. open) group appeared significantly greater (64.8±13.9 vs. 58.2±14.8 years; P=0.015), and the percentage of patients with chronic obstructive pulmonary disease tended to be higher (hybrid: 17.4%, 8/46; open: 7.6%, 6/79; P=0.091), as did histories of previous aortic surgery (hybrid: 23.9%, 11/46; open: 12.7%, 10/79; P=0.105). In contrast, maximum aneurysm size was significantly greater in the open (vs. hybrid) group (63.3±10.9 vs. 59.4±10.2 mm; P=0.042). In examining the extent of arch and DTA involvement, proximal DTA lesions accounted for a sightly higher proportion in the hybrid group by comparison (hybrid: 45.7%, 21/46; open: 32.9%, 26/79; P=0.156). After adjustment, however, baseline characteristics and distributions within the two groups were found to be well balanced.
Full table
Operative data
Conventional open repair
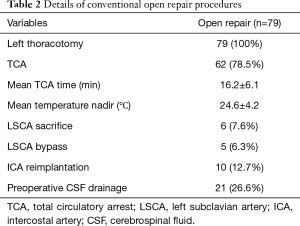
Each patient underwent hemi-arch and DTA replacement via left thoracotomy. TCA was performed in 62 patients (78.5%) and aortic cross clamping (between subclavian and carotid arteries) was performed in 17 patients (21.5%). Mean TCA time was 16.2±6.1 min, and mean nadir of body temperature was 24.6±4.2 °C. LSCA sacrifice was done in six patients. Details of open repair are summarized in Table 2.
Full table
Hybrid arch repair
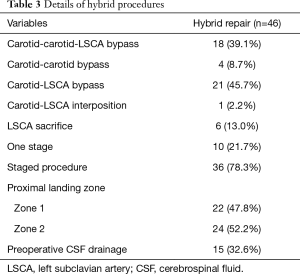
Twenty-two patients (47.8%) underwent cervical debranching procedures, including right-to-left carotid bypass. LSCA revascularization was performed in 38 patients (82.6%). Staged procedures were conducted in 36 patients (78.3%), with 22 patients (47.8%) undergoing Zone 1 TEVAR. Preoperative drainage of cerebrospinal fluid (CSF) was undertaken in 15 patients (32.6%). The types of stents used for TEVAR were as follows: Seal (S&G Biotech Inc., Seoul, Republic of Korea); Valiant (with Captivia delivery system; Medtronic, Santa Rosa, CA, USA); and Cook TX2 device (Cook Medical Inc., Bloomington, IN, USA). Details of the hybrid procedures are summarized in Table 3.
Full table
Early outcomes
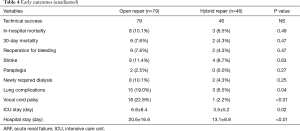
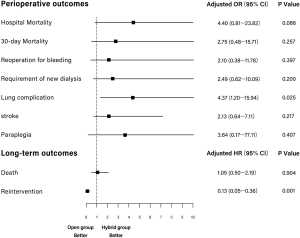
Unadjusted early perioperative outcomes are summarized in Table 4. There were no early open conversions for failed hybrid procedures. In-hospital mortality did not differ significantly by group (open: 10.1%, 8/79; hybrid: 6.5%, 3/46; P=0.49), and strokes occurred with similar frequency (open: 11.4%, 9/79; hybrid: 8.7%, 4/46; P=0.63). In two patients, conventional open repair resulted in paraplegia, compared with none in the hybrid group, but the difference was not significant (P=0.27). Postoperative morbidity rates recorded for lung complications (open, 19.0%; hybrid, 6.5%; P=0.04) and vocal cord palsy (open, 22.8%; hybrid, 2.2%; P<0.01) proved to be significantly higher in those undergoing conventional open repair. Adjusted ORs of adverse outcomes in open and hybrid groups are summarized in Table 5. After IPTW adjustment, there were no significant differences in 30-day mortality and perioperative stroke (P=0.257 and P=0.217, respectively). However, compared with the hybrid group, the risk of hospital mortality tended to be higher for the open group (OR =4.396; 95% CI: 0.811–23.816; P=0.086), and risk of lung complications was significantly higher (OR =4.372, 95% CI: 1.200–15.935; P=0.025) (Figure 1).
Full table

Full table
Long-term survival and reintervention
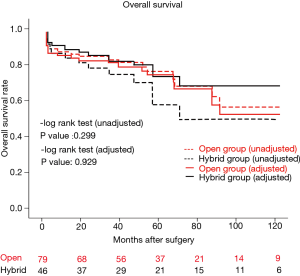
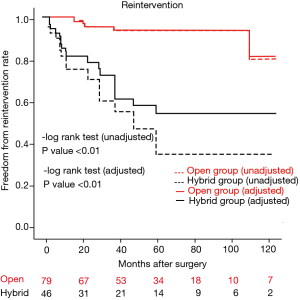
Mean follow-up duration was 51.9±34.9 months (range, 1–116 months). The two groups did not differ significantly in this regard (open: 50.3±33.9 months; hybrid: 54.6±36.6 months; P=0.50), nor did overall survival rates at 10 years (open: 59.4%±8.8%; hybrid: 48.8%±10.8%; P=0.299, unadjusted; P=0.929, adjusted) (Figure 2). In the hybrid group, 16 (34.8%) patients required reintervention or surgery, including endovascular reintervention in 12 patients and open conversion for aneurysm enlargement (n=2) or retrograde type A dissection (n=2). Subsequently, the freedom from reintervention rates at 10 years differed significantly (open: 85.2%±7.1%; hybrid: 41.3%±11.3%; P<0.01, unadjusted; P<0.01, adjusted) (Figure 3).
Discussion
In patients with disease of the descending aorta and distal arch involvement, optimal treatment is unclear, given the current lack of data. There are no studies to date comparing thoracotomy and hybrid techniques in such scenarios. Based on the present investigation, it is apparent that hybrid repairs carry equivalent early and late mortality risk, surpassing conventional open repair by virtue of fewer pulmonary complications and shorter ICU or hospital stays. However, the hybrid approach seems less durable than open repair.
Potential negative side-effects of thoracotomy for aortic arch lesions
Although clinical outcomes of open repair have been well documented in isolated DTA segments, such documentation is lacking for distal arch involvement. Okita et al. showed that atherosclerotic arch lesions accompanying descending thoracic aneurysms introduce considerable risk, affecting early mortality and stroke rates (15). Thoracotomy increases the chance of embolic events. When the arch is opened during deep hypothermic arrest, without aortic clamping, the embolic burden of an atherosclerotic arch may disperse retrograde into carotid vessels, leading to embolic strokes (16). Even if the arch is clamped, it inevitably requires manipulation, again posing the threat of embolic stroke. The current study findings likewise linked early mortality in the thoracotomy group to embolic events such as stroke or coronary embolism, associated with manipulation of arch lesions.
Pulmonary complications associated with thoracotomy are readily acknowledged as consequences of aortic arch repair and reportedly are as high as 30% (17). In the present study, the incidence of pulmonary complications after conventional open repair was significantly higher than that following hybrid surgery (open: 19%, 15/79; hybrid: 6.5%, 3/46; P=0.04). The demands of prolonged mechanical ventilation and tracheostomy have greater impact in the context of open (vs. hybrid) repair. It is thought that lung manipulation during thoracotomy is a source of parenchymal bleeding and edema; and vocal cord palsy may promote further pulmonary compromise owing to aspiration or impaired bronchial toilet.
These findings demonstrate that when arch lesions are involved, an open (i.e., thoracotomy) approach is fraught with potential risks, particularly embolic events or lung complications, which may contribute to perioperative mortality.
Potential negative side-effects of hybrid arch repair
There are inherent difficulties in endovascular procedures as well, because aortic arch characteristics/properties, including its angulation, high-velocity blood flow, and substantial pulsatile movement, are apt to be problematic. According to previous studies, arch lesions are more often subject to type I endoleaks than are isolated TEVAR procedures (18-20). Our results proved similar to outcomes of reintervention or open conversion, with 75% (12/16) of type I endoleaks or new entry tears occurring at proximal lesion endografts. Stent migration may also occur more frequently in arch lesions. As mentioned above, the combination of angulation and high-velocity flow are key factors in stent migration after hybrid arch repair. On one occasion, stent migration and type I endoleak presented 2 years after a hybrid arch repair. The patient was treated by reintervention.
Finally, retrograde type I dissection (RTAD) may rarely develop after TEVAR. RTAD has been reported at rates of 1–2% after TEVAR (21-23). In hybrid arch repairs, the incidence of RTAD appears to be higher than after single TEVAR procedures (19). Recently Gandet et al. reported that in 32 patients undergoing hybrid arch repairs, six experienced RTAD (18.7%) (24). Again, factors such as severe angulation, high-velocity flow, and pulsatile movement may contribute to the development of RTAD. In the current study, the incidence of RTAD after hybrid repairs was 4.3% (2/46). Of note, RTAD may occur during initial procedures or in the course of follow-up monitoring. One year postoperatively, RTAD was discovered by CT scan in one of our patients, who then required emergency surgery.
Comparing early outcomes of open and hybrid repairs
The superiority of any approach in treating diseases of the descending aorta that involve the arch is still debatable. Although many sources credit hybrid repairs with less invasiveness, there is little research that truly validates any claimed advantages over conventional open repair. As cited in earlier reports, outcomes of hybrid arch (vs. open) repair may be more favorable in terms of operative morbidities and early recovery (11-13). Herein, we have also shown that pulmonary complications are reduced and that ICU or hospital stays are shortened.
It is important to note that stroke and early mortality percentages after hybrid repairs are still high. In a meta-analysis undertaken by Moulakakis et al., stroke (7.6%) and mortality (11.6%) rates for hybrid arch repair showed no significant improvement relative to open approach (25). Despite certain advantages (fewer pulmonary or renal complications and reduced ICU/hospital stays), we similarly discovered no distinct mortality (open: 10.1%; hybrid: 6.5%; P=0.49) or stroke (open: 11.4%; hybrid: 8.7%; P=0.63) benefits. Three of our patients suffered early mortality after hybrid repairs, two dying of massive cerebral infarction and the other of sudden death due to hemothorax; and all occurred after TEVAR procedures. Five strokes were also recorded in the aftermath of TEVAR, but none after debranching. Hence, it appears that the endovascular aspect of a hybrid approach to diseased arch lesions is implicated in the ominous outcomes (i.e., stroke or death) thereafter.
Limitations of hybrid strategy reintervention
Reintervention is unquestionably required more often after endovascular aortic repair than after open repair. Hybrid strategies also have limitations in terms of long-term endograft durability. After hybrid arch repair, reintervention rates range from 0–30% (6-14). In our study, 16 patients (34.8%) in the hybrid group required reintervention or open surgery for various reasons, such as type I or II endoleak, stent migration, and RTAD. Hybrid arch repairs were also associated with significantly higher percentages of reintervention during a 10-year follow-up period, compared with open repair [34.8% (16/46) vs. 7.6% (6/79); P<0.01]; and such complications may occur at any time. In our study, five reinterventions performed >4 years after hybrid aortic repairs were necessitated by type I endoleaks and stent migration. Moreover, one patient died suddenly from a recurring type I endoleak (while awaiting open conversion), having previously survived reintervention after Zone 1 TEVAR. Although many late complications of hybrid arch repairs may be resolved through endovascular technique, patients with no hope of endovascular salvage will need open conversion to avoid sudden death. The findings herein suggest that long-term survival of patients undergoing hybrid arch repairs may decline over time.
Study limitations
This study has several clear limitations. Its retrospective design imparts selection bias. Not all patients with aortic aneurysms were anatomically suited for hybrid repair, adding further to selection bias. There was also heterogeneity of patient characteristics, aortic pathologies, and degrees of involvement. Nevertheless, the latter were minimized by focusing on descending aortas with arch involvement. The relatively small number of patients recruited was another limitation.
Conclusions
Outcomes of both open and hybrid surgical techniques were satisfactory. Hybrid arch repair conferred a significantly lower incidence of pulmonary complications, but without apparent benefits for midterm survival and perioperative stroke. However, open repair proved more reliable, showing greater durability than hybrid repair. Long-term follow-up is needed to confirm the viability and safety of hybrid repair as an alternative treatment in this setting.
Acknowledgements
Funding: This study was supported by a faculty research grant of Yonsei University College of Medicine for 2014 (6-2014-0155).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The Institutional Review Board of Yonsei University College of Medicine approved the study (Yonsei Institutional Review Board No. 4-2014-0810), waiving the customarily required individual patient consent.
References
- Coselli JS, LeMaire SA, Conklin LD, et al. Left heart bypass during descending thoracic aortic aneurysm repair does not reduce the incidence of paraplegia. Ann Thorac Surg 2004;77:1298-303; discussion 1303. [Crossref] [PubMed]
- Estrera AL, Miller CC 3rd, Chen EP, et al. Descending thoracic aortic aneurysm repair: 12-year experience using distal aortic perfusion and cerebrospinal fluid drainage. Ann Thorac Surg 2005;80:1290-6; discussion 1296. [Crossref] [PubMed]
- Leurs LJ, Bell R, Degrieck Y, et al. Endovascular treatment of thoracic aortic diseases: combined experience from the EUROSTAR and United Kingdom Thoracic Endograft registries. J Vasc Surg 2004;40:670-9; discussion 679-80. [Crossref] [PubMed]
- Criado FJ, Abul-Khoudoud OR, Domer GS, et al. Endovascular repair of the thoracic aorta: lessons learned. Ann Thorac Surg 2005;80:857-63; discussion 863. [Crossref] [PubMed]
- Patel HJ, Shillingford MS, Williams DM, et al. Survival benefit of endovascular descending thoracic aortic repair for the high-risk patient. Ann Thorac Surg 2007;83:1628-33; discussion 1633-4. [Crossref] [PubMed]
- Szeto WY, Bavaria JE, Bowen FW, et al. The hybrid total arch repair: brachiocephalic bypass and concomitant endovascular aortic arch stent graft placement. J Card Surg 2007;22:97-102; discussion 103-4. [Crossref] [PubMed]
- Czerny M, Zimpfer D, Fleck T, et al. Initial results after combined repair of aortic arch aneurysms by sequential transposition of the supra-aortic branches and consecutive endovascular stent-graft placement. Ann Thorac Surg 2004;78:1256-60. [Crossref] [PubMed]
- Gottardi R, Seitelberger R, Zimpfer D, et al. An alternative approach in treating an aortic arch aneurysm with an anatomic variant by supraaortic reconstruction and stent-graft placement. J Vasc Surg 2005;42:357-60. [Crossref] [PubMed]
- Koullias GJ, Wheatley GH 3rd. State-of-the-art of hybrid procedures for the aortic arch: a meta-analysis. Ann Thorac Surg 2010;90:689-97. [Crossref] [PubMed]
- Cao P, De Rango P, Czerny M, et al. Systematic review of clinical outcomes in hybrid procedures for aortic arch dissections and other arch diseases. J Thorac Cardiovasc Surg 2012;144:1286-300, 1300.e1-2.
- Murashita T, Matsuda H, Domae K, et al. Less invasive surgical treatment for aortic arch aneurysms in high-risk patients: a comparative study of hybrid thoracic endovascular aortic repair and conventional total arch replacement. J Thorac Cardiovasc Surg 2012;143:1007-13. [Crossref] [PubMed]
- Milewski RK, Szeto WY, Pochettino A, et al. Have hybrid procedures replaced open aortic arch reconstruction in high-risk patients? A comparative study of elective open arch debranching with endovascular stent graft placement and conventional elective open total and distal aortic arch reconstruction. J Thorac Cardiovasc Surg 2010;140:590-7. [Crossref] [PubMed]
- Benedetto U, Melina G, Angeloni E, et al. Current results of open total arch replacement versus hybrid thoracic endovascular aortic repair for aortic arch aneurysm: a meta-analysis of comparative studies. J Thorac Cardiovasc Surg 2013;145:305-6. [Crossref] [PubMed]
- Ishimaru S. Endografting of the aortic arch. J Endovasc Ther 2004;11 Suppl 2:II62-71. [Crossref] [PubMed]
- Okita Y, Ando M, Minatoya K, et al. Predictive factors for mortality and cerebral complications in arteriosclerotic aneurysm of the aortic arch. Ann Thorac Surg 1999;67:72-8. [Crossref] [PubMed]
- Yoo JS, Kim JB, Jung SH, et al. Surgical repair of descending thoracic and thoracoabdominal aortic aneurysm involving the distal arch: open proximal anastomosis under deep hypothermia versus arch clamping technique. J Thorac Cardiovasc Surg 2014;148:2101-7. [Crossref] [PubMed]
- Schechter MA, Shah AA, Englum BR, et al. Prolonged postoperative respiratory support after proximal thoracic aortic surgery: Is deep hypothermic circulatory arrest a risk factor? J Crit Care 2016;31:125-9. [Crossref] [PubMed]
- Weigang E, Parker J, Czerny M, et al. Endovascular aortic arch repair after aortic arch de-branching. Ann Thorac Surg 2009;87:603-7. [Crossref] [PubMed]
- Piffaretti G, Mariscalco G, Lomazzi C, et al. Predictive factors for endoleaks after thoracic aortic aneurysm endograft repair. J Thorac Cardiovasc Surg 2009;138:880-5. [Crossref] [PubMed]
- Hughes GC, Daneshmand MA, Balsara KR, et al. "Hybrid" repair of aneurysms of the transverse aortic arch: midterm results. Ann Thorac Surg 2009;88:1882-7; discussion 1887-8.
- Eggebrecht H, Thompson M, Rousseau H, et al. Retrograde ascending aortic dissection during or after thoracic aortic stent graft placement: insight from the European registry on endovascular aortic repair complications. Circulation 2009;120:S276-81. [Crossref] [PubMed]
- Kpodonu J, Preventza O, Ramaiah VG, et al. Retrograde type A dissection after endovascular stenting of the descending thoracic aorta. Is the risk real? Eur J Cardiothorac Surg 2008;33:1014-8. [Crossref] [PubMed]
- Geisbüsch P, Kotelis D, Müller-Eschner M, et al. Complications after aortic arch hybrid repair. J Vasc Surg 2011;53:935-41. [Crossref] [PubMed]
- Gandet T, Canaud L, Ozdemir BA, et al. Factors favoring retrograde aortic dissection after endovascular aortic arch repair. J Thorac Cardiovasc Surg 2015;150:136-42. [Crossref] [PubMed]
- Moulakakis KG, Mylonas SN, Markatis F, et al. A systematic review and meta-analysis of hybrid aortic arch replacement. Ann Cardiothorac Surg 2013;2:247-60. [PubMed]


