Early results of new endoscopic thoracic sympathectomy for craniofacial hyperhidrosis
Introduction
Craniofacial hyperhidrosis (CFH), a condition of excessive sweating of the head and face, is one of the most common cardinal symptoms of social phobia (1). Those with this condition may also likely experience a significant deterioration in the quality of life (2). Hence, to overcome these problems of CFH and to return to normal daily activities with self-esteem, treatment of CFH is highly recommended.
It has previously been established that CFH accounts for only 22.8% of all primary hyperhidrosis and is different from other forms of focal hyperhidrosis that are more common amongst males and elderly patients (3). Hence, primary CFH should be considered only after ruling out secondary conditions, such as menopause, diabetes mellitus, endocrine disorders, and certain medications, including hydrochloride and pilocarpine (4).
Endoscopic thoracic sympathectomy (ETS) has been considered as a definite treatment method for primary hyperhidrosis (5-9). It has been widely utilized for palmar and axillary hyperhidrosis, due not only to the relative ease of accessibility and short recovery time necessary, but also to its high success rate. Despite its wide application, however, to the best our knowledge, there have been only a few studies evaluating the applicability of ETS for CFH and risk of postoperative compensatory hyperhidrosis. Moreover, many institutions are avoiding surgical approaches to treat CFH due to compensatory hyperhidrosis after ETS. This motivated our team to develop a new method of ETS to minimize the degree and locations of postoperative compensatory hyperhidrosis, with the ultimate goal of reducing social phobia and discomfort in daily life in our patients. Therefore, this study attempted to examine whether our newly developed method of ETS is safe and efficient for managing CFH, and to analyze postoperative compensatory hyperhidrosis based on early results.
Methods
Patients and clinical data
We retrospectively analyzed the medical records of 72 patients who underwent ETS with our new technique for primary CFH between June 2016 and October 2017. All operations were performed on an outpatient basis. We excluded patients with severe pleural adhesion (n=1) and those who underwent previous thoracic surgery (n=1). The data were collected by a manual review of the electronic records of patients, which consisted of information on preoperative characteristics, disease status, operative technique, and postoperative outcomes. Moreover, we interviewed patients using a telephone questionnaire that has previously been reported, with modification, in June 2015 (10). The questionnaire included the degree of craniofacial sweating, grade of compensatory hyperhidrosis, and recurrence/failure. The degree of craniofacial sweating was defined as a percentage of post-operative craniofacial sweating compared with pre-operative craniofacial sweating. The grade of compensatory hyperhidrosis was categorized as none, mild (not bothering), moderate (sometimes bothering), severe (often bothering), or very severe (intolerable). The grade of satisfaction was categorized as very satisfied, satisfied, moderate, dissatisfied, or very dissatisfied. Recurrence or failure was considered in patients presented with a residual craniofacial sweating of 40% for those who underwent new ETS. This retrospective study was approved by the local institutional review board at the Gangnam Severance Hospital Korea, and the requirement for informed consent was waived due to its retrospective nature.
Surgery
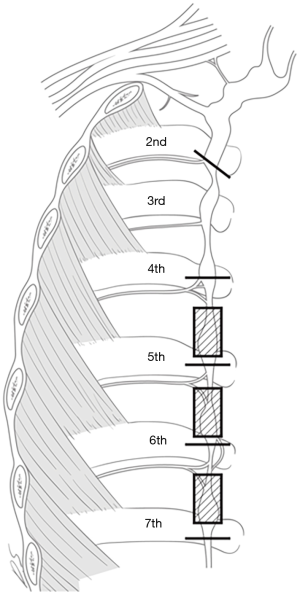
All patients, while under general anesthesia via one-lung ventilation in lateral decubitus position, underwent ETS with our new technique, using a bilateral simultaneous two-portal video-assisted thoracoscopic surgery (VATS). The new technique for ETS involved R2 and R4–R7 sympathectomy with R4–R7 truncal ablation using a CO2 gas instillation. Two tiny incisions of 2 and 3 mm were made on each side; the incision for 2 mm-thoracoscope was made at the 6th intercostal space (ICS) on the mid-axillary line, and the incision for instruments was made at the 5th ICS on the anterior axillary line. When performing R2 sympathectomy, the costal pleura on the surface of the sympathetic chain were transected at the level of the second lower rib bed with diathermy. To complete the transection of the potential bypass nerve fibers, we extended the transection range by approximately 2 cm, laterally along the surface; we then performed R4, R5, R6, and R7 sympathectomy and R4–R7 truncal ablation to prevent upper body hyperhidrosis (Figure 1). We routinely conducted the operation on the right side first, to get a view of the imbalanced innervation of the heart by bilateral sympathetic nerves (7). The corresponding procedure was performed on the left side. A 10-Fr chest tube was placed in the pleural cavity, the lung was inflated, and the chest tube was removed once the air ceased to escape. Postoperative routine chest radiography was obtained in all patients prior to extubation in the operating room for the early detection of pneumothorax. Most patients were discharged on the same day or the following day.
Statistical analysis
All descriptive statistics are presented as the mean ± standard deviation (SD) for continuous variables. All statistical analyses were performed using SPSS statistical software version 21.0 (SPSS, Inc., Chicago, IL, USA).
Results
A total of 70 patients who underwent ETS using our new technique were included in this study. All procedures were performed bilaterally. Among these patients, 55 patients were male (78.6%), and the mean age was 48 years, with a range of 22–75 years. The median operation time was 38 minutes, with a range of 28–75 minutes (Table 1).
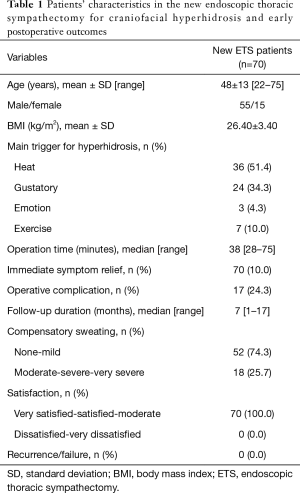
Full table
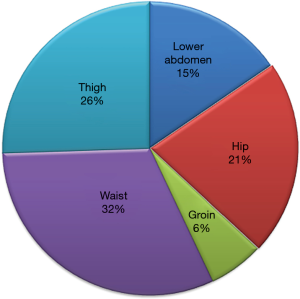
Bilateral ETS using our new technique was successful in all patients. There were no operative mortalities or major morbidities. The immediate operative results were excellent; all patients showed significant improvement with respect to sweating of the head and face. The median follow-up duration was 7 months (range, 1–17 months); 68 patients (97.1%) developed compensatory sweating in the lower limbs and lower trunk (Figure 2). The degree of compensatory hyperhidrosis was mostly mild to moderate in most patient (92.9%, 65/70); only 5 patients reported having severe compensatory hyperhidrosis in the lower abdomen and thigh with slight disturbance to everyday life. In terms of satisfaction, 46 (65.7%) patients were “very satisfied” and 24 (34.4%) patients were “satisfied” (Table 2). Neither transient nor permanent Horner’s syndrome occurred in any patient. We did not observe any patient with recurrence or failure of ETS using our new technique.
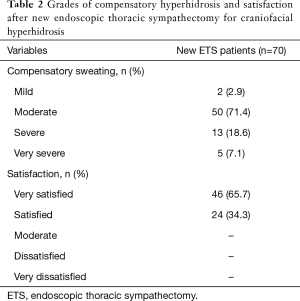
Full table
Discussion
Hyperhidrosis is defined as a pathologic condition characterized by excessive sweating (5). Although it is mainly a physical burden, hyperhidrosis can also negatively impact the psychological, emotional, and social well-being of an individual, severely debilitating one’s quality of life (5,11). More specially, CFH, which is a condition of excessive local sweating, on the head and face, is often associated with mental stress (1). Patients suffering from CFH are considered to be seriously handicapped, both socially and psychologically (2). Nonetheless, CFH has been a clinical problem overshadowed by palmar, plantar, and axillary hyperhidrosis.
Since Drott et al. recommended ablation of the first ganglion to manage CFH patients, several studies have presented satisfying results, and since then, the operative method has evolved (6). Still, the surgical approach of transecting the sympathetic chain in the lower margin of the second rib or in the lower third of the stellate ganglion level has been standardized and utilized in most institutions (9). An accurate identification of the second rib is very important. The superior intercostal artery, which arises from the subclavian artery, is very important in identifying the second rib. Lin and their colleagues suggested that the first intercostal muscle may be the best method in identifying of the second rib (2). After undergoing surgery to treat CFH, the incidence of Horner’s syndrome has been reported to be between 0% and 3.3% (2,5,9). In our study, we did not have any incidence of Horner’s syndrome. Thus, a transection of the lower third of the stellate ganglion or the lower margin of R2 does not result in Horner’s syndrome.
ETS is an elective procedure with great potential benefits. However, it is also associated with some significant risks. The major complication of the ETS is the high rates of compensatory hyperhidrosis (8). Postoperative compensatory hyperhidrosis after a standard ETS, which is usually performed at T2, is common in the buttocks, chest, upper limbs and lower limbs; but it is not limited to these areas and can manifest in other parts of the body (1). Although a clear mechanism for these complications has not been fully elucidated thus far, it has been proposed that a thermoregulatory response may be associated (2). Those who develop postoperative compensatory hyperhidrosis tend to regret receiving ETS for CFH, as they are left with a similar condition, just at another location of their body, posing a similar debilitative condition (12). Lin TS and their colleagues reported that 85.7% who underwent ETS for CFH developed compensatory hyperhidrosis, and 48% of them developed compensatory hyperhidrosis in lower chest and abdomen areas (2).
Given such high incidence of postoperative compensatory hyperhidrosis using the standard ETS for CFH, we used the following logic in developing our approach: If postoperative compensatory hyperhidrosis is unavoidable in most cases, we may be able to obtain greater satisfaction if we could localize the site of compensatory hyperhidrosis to the lower extremity. To achieve this, R4–R7 sympathectomy with truncal ablation, in addition to the conventional R2 sympathectomy was performed. R4–R7 sympathectomy was performed to eliminate compensatory hyperhidrosis in the upper part trunk, and R4–R7 truncal ablation was performed to provide similar advantages of sympathectomy. As expected, we observed a high incidence (97.1%) of compensatory hyperhidrosis in our study population. However, we were able to achieve localization of compensatory hyperhidrosis to the lower extremity, with no incidence of postoperative compensatory sweating in the chest area. We observed five patients with severe compensatory hyperhidrosis in the waist and lower abdomen. These patients are currently receiving oxybutynin at the outpatient clinic and are reporting that their compensatory hyperhidrosis is under control without much interference with daily living. It is unclear why, despite the same procedural approach, there is a variance in the degree and location of compensatory hyperhidrosis between individuals. To better understand and such variance, a future study analyzing the risk factors is warranted. Nonetheless, all our patients were satisfied with the operation, and reported that postoperative compensatory hyperhidrosis was relatively tolerable.
There are some limitations to consider when interpreting our findings. Our study included a small number of sample size, with a short follow-up duration. In addition, compensatory hyperhidrosis and symptoms of our patients were not measured in an objective manner; they were based on a survey. A future study with larger sample size and longer follow-up duration with objective indicators are necessary.
Conclusions
We believe that this study is meaningful in that it provides a new surgical approach to overcome the issue of compensatory hyperhidrosis. Although ETS may not be the treatment of choice for CFH, our method of ETS offers a relatively safe and efficient treatment option for those who do not respond to other conservative treatments, such as topical glycopyrrolate, oral oxybutynin, and intradermal botulinum toxin A injection.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This retrospective study was approved by the local institutional review board at the Gangnam Severance Hospital Korea and the requirement for informed consent was waived.
References
- Nicholas R, Quddus A, Baker DM. Treatment of Primary Craniofacial Hyperhidrosis: A Systematic Review. Am J Clin Dermatol 2015;16:361-70. [Crossref] [PubMed]
- Lin TS, Fang HY. Transthoracic endoscopic sympathectomy for craniofacial hyperhidrosis: analysis of 46 cases. J Laparoendosc Adv Surg Tech A 2000;10:243-7. [Crossref] [PubMed]
- Lear W, Kessler E, Solish N, Glaser DA. An epidemiological study of hyperhidrosis. Dermatol Surg 2007;33:S69-75. [Crossref] [PubMed]
- Glaser DA, Hebert AA, Pariser DM, et al. Facial hyperhidrosis: best practice recommendations and special considerations. Cutis 2007;79:29-32. [PubMed]
- Cerfolio RJ, De Campos JR, Bryant AS, et al. The Society of Thoracic Surgeons expert consensus for the surgical treatment of hyperhidrosis. Ann Thorac Surg 2011;91:1642-8. [Crossref] [PubMed]
- Drott C, Göthberg G, Claes G. Endoscopic transthoracic sympathectomy: an efficient and safe method for the treatment of hyperhidrosis. J Am Acad Dermatol 1995;33:78-81. [Crossref] [PubMed]
- Liu Y, Yang J, Liu J, et al. Surgical treatment of primary palmar hyperhidrosis: a prospective randomized study comparing T3 and T4 sympathicotomy. Eur J Cardiothorac Surg 2009;35:398-402. [Crossref] [PubMed]
- Moraites E, Vaughn OA, Hill S. Endoscopic thoracic sympathectomy. Dermatol Clin 2014;32:541-8. [Crossref] [PubMed]
- Wait SD, Killory BD, Lekovic GP, et al. Thoracoscopic sympathectomy for hyperhidrosis: analysis of 642 procedures with special attention to Horner's syndrome and compensatory hyperhidrosis. Neurosurgery 2010;67:652-6; discussion 656-7. [Crossref] [PubMed]
- Mohebbi HA, Mehrvarz S, Manoochehry S. Thoracoscopic Sympathicotomy vs Sympathectomy in Primary Hyperhidrosis. Trauma Mon 2012;17:291-5. [Crossref] [PubMed]
- Reisfeld R, Berliner KI. Evidence-based review of the nonsurgical management of hyperhidrosis. Thorac Surg Clin 2008;18:157-66. [Crossref] [PubMed]
- Karlqvist M, Rosell K, Rystedt A, et al. Botulinum toxin B in the treatment of craniofacial hyperhidrosis. J Eur Acad Dermatol Venereol 2014;28:1313-7. [Crossref] [PubMed]