One-week recovery from bronchopleural fistula by combined techniques
Introduction
Bronchopleural fistula (BPF) is an uncommon, but severe complication of pulmonary resection surgery (1). And the mortality rates associated with BPF are alarmingly high and ranges between 7.1% and 67% (1-3). It has been generally accepted that the three paramount management goals of BPF included securely blocking the direct fistulous tract, sustainable eliminating of all inflammatory effusion, and precisely administration of appropriate antibiotics for pleural space infection. Treatment options for BPF included chest drains, muscle flap, omental flap, transsternal bronchial closure, thoracoplasty and prolonged antibiotic regimens (3).
In this study, to avoid aggressive surgical intervention, we described 1-week recovery from combined techniques for one patient with BPF after lobectomy (Figure 1).
Case presentation
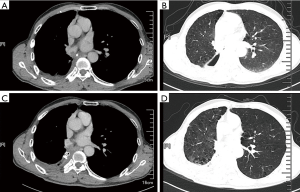
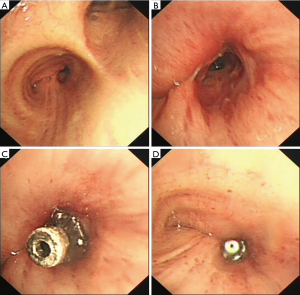
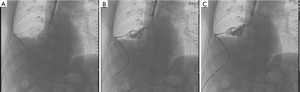
A 65-year-old male was admitted to our hospital with the chief complaint of repeated cough and vomiting white fluid while bending down for one month. The patient underwent a right lower lobectomy at local hospital three months ago. No significant high-risk factors for BPF, such as neoadjuvant therapy, postoperative complication, or diabetes mellitus, were reported. Chest computed tomography (CT) suggested a possible right lower lobe BPF with a chest tube drainage (Figure 1A,B). Immediate intravenous empirical antibiotics of piperacillin/sulbactam (Welman, HB, China) was prescribed with the dose of 3.0 g/q12h. Bacterial cultures of the chest drainage were taken, the results supported the prescription of antibiotics. A portable electronic chest drainage system (Thopaz, Medela Healthcare, McHenry, IL, USA) was attached to the chest tube for continuous drainage of thoracic effusion (4). A 5-mm diameter fistulous orifice on the opening of right lower lobe was observed by bronchoscopy (Figure 2A,B). A bronchoscopic self-expandable Amplatzer device (AD) (SQFDQ-1 08, SJM, MN, USA) was implanted (Figure 2C,D), based on the confirmation of right lower lobe BPF by digital subtraction angiography (DSA) (Figures 3,4). Three days after closure of the fistula, staphylococcus cohnii urealyticum (MRSCN) were cultivated from thoracic effusion. To suppress this MRSCN infection, iodine-based sterilizing solution, 0.09–0.11% Available iodine of An’erdian Skin-Mucous Rinse Antiseptic (500 mL/bottle, Shanghai Likang Disinfectant Hi-Tech Co., Ltd.), mixed with lidocaine (20 mL:20 mL) was irrigated into thoracic cavity through chest tube for 20 minutes with chest tube clip by a forcep. After removing the forceps, chest drainage continued. The patient ceased to complain fever the next day. Repeated chest CT was performed 5 days after the implantation of Amplatzer device, which revealed proper position of Amplatzer device and improvement of pleural space infection (Figure 1C,D). The Amplatzer device would be left in place permanently. On the 7th day after the implantation, repeated smear test of thoracic drainage was reported negative; along with the normal blood result, the chest tube was removed. And the patient was discharged the same day in good physical condition. Moxifloxacin tablet (BSP, Berlin, Germany) was further prescribed for 7 days with the dose of 0.4 g/qd.

The patients reported no significant symptom and the repeated CT revealed no recurrence 6 months after discharge.
Comments
The primary principles for managing BPF include initial drainage, tailored antibiotics and optimization of nutrition; closure of the fistula, typically with autologous flap buttressing; and appropriate management of the infected space. Although many treatment options are currently proven to be effective for BPF, including surgical and bronchoscopic operations, or conservative managements (6), there is no promise for single treatment to be omnipotent. And the recovery process could be painful and time-consuming.
The successful treatment of BPF by endoscopic treatment is variable and mainly depend on the proximity, size of the fistula and the underlying disease. As for our patient, three combined techniques were adopted for this patient. It was reported of BPF closed with cellulose patch and fibrin glue, yet the patient with diameter of the fistula larger than 5 mm failed in bronchial dehiscence (7). In recent years, Amplatzer device, which have been developed for the transcatheter closure of cardiac atrial septum defects, have been successfully used for the bronchoscopic occlusion of BPF (8). To ensure a sound closure of the fistula, we implanted self-expandable Amplatzer device, which isolated airway from the infected pleural effusion. This novel method is suitable for patients with large BPF and long tract communicating between the bronchial system and the pleural cavity (8). However, this report also observed nine patients (9/19, 47.4%) died due to sepsis. To eliminate latent infection, two other techniques were applied. First, the portable electronic chest drainage system was originally applied in post-lobectomy drainage to achieve shorter duration of chest tube placement (4). Given the similar, even urgent intention, this system was applied for continuous drainage of thoracic effusion. Second, based the favorable results of iodine-based sterilizing solution in establishing pleurodesis (9) and treating empyema (10), similar solution was irrigated to the chest cavity, and the drainage cultivation became asepsis one day after this intervention.
There were several limitations about this case report. First, beside the combined techniques, this quick recovery process should also be given credit to prompt drainage before admitting to our hospital. Second, this patient suffered from limited pleural infection, and he reported no diabetes or systematic bacteremia. This method should be cautiously applied to high-risk patients.
In conclusion, the combined techniques for BPF might be an effective alternative option. Though, based on the protean nature of the disease, the optimal treatment strategy needs to be individualized in each patient.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Hu XF, Duan L, Jiang GN, et al. A clinical risk model for the evaluation of bronchopleural fistula in non-small cell lung cancer after pneumonectomy. Ann Thorac Surg 2013;96:419-24. [Crossref] [PubMed]
- Pierson DJ, Horton CA, Bates PW. Persistent bronchopleural air leak during mechanical ventilation. A review of 39 cases. Chest 1986;90:321-3. [Crossref] [PubMed]
- Sirbu H, Busch T, Aleksic I, et al. Bronchopleural fistula in the surgery of non-small cell lung cancer: incidence, risk factors, and management. Ann Thorac Cardiovasc Surg 2001;7:330-6. [PubMed]
- Pompili C, Detterbeck F, Papagiannopoulos K, et al. Multicenter international randomized comparison of objective and subjective outcomes between electronic and traditional chest drainage systems. Ann Thorac Surg 2014;98:490-6; discussion 496-7. [Crossref] [PubMed]
- Liu J, Li J, He J, et al. The right lower lobe BPF was confirmed by endobronchial digital subtraction angiography. Asvide 2018;5:585. Available online: http://www.asvide.com/article/view/25560
- Mao R, Ying PQ, Xie D, et al. Conservative management of empyema-complicated post-lobectomy bronchopleural fistulas: experience of consecutive 13 cases in 9 years. J Thorac Dis 2016;8:1577-86. [Crossref] [PubMed]
- Fiorelli A, Frongillo E, Santini M, et al. Bronchopleural fistula closed with cellulose patch and fibrin glue. Asian Cardiovasc Thorac Ann 2015;23:880-3. [Crossref] [PubMed]
- Fruchter O, El Raouf BA, Abdel-Rahman N, et al. Efficacy of bronchoscopic closure of a bronchopleural fistula with amplatzer devices: long-term follow-up. Respiration 2014;87:227-33. [Crossref] [PubMed]
- Agarwal R, Aggarwal AN, Gupta D. Efficacy and safety of iodopovidone pleurodesis through tube thoracostomy. Respirology 2006;11:105-8. [Crossref] [PubMed]
- Schneiter D, Grodzki T, Lardinois D, et al. Accelerated treatment of postpneumonectomy empyema: a binational long-term study. J Thorac Cardiovasc Surg 2008;136:179-85. [Crossref] [PubMed]


