Comparison of the 11th Japanese classification and the AJCC 7th and 8th staging systems in esophageal squamous cell carcinoma patients
Introduction
Staging systems for cancer are essential for determining treatment plans, communicating among medical personnel, and accurately predicting survival. A widely accepted and used cancer staging system is the tumor, node, and metastasis (TNM) system. The TNM system describes the anatomical extent of disease based on assessment of three components: T is the extent of the primary tumor, N is the absence or presence and extent of regional lymph node metastasis, and M is the absence or presence of distant metastasis. The American Joint Committee on Cancer (AJCC)/Union for International Cancer Control (UICC) system is based on the TNM descriptor and has been adopted worldwide. For esophageal cancer, the 7th edition of the AJCC staging system for esophageal cancer was published in 2009 based on a Worldwide Esophageal Cancer Collaboration (WECC) database (1) and 8th edition of the AJCC/UICC system was published in 2017. In addition to the AJCC system, Japan has its own esophageal cancer staging system. The current Japanese staging system is the 11th edition of the Japanese classification for esophageal cancer, based on a comprehensive esophageal cancer registry in Japan (2). Compared to the AJCC system, the Japanese classification has a different lymph node mapping system for esophageal cancer and different definitions of N staging and stage grouping compared to the AJCC system. The Japanese classification reflects the extensive experiences of Japanese surgeons who have performed uniform operations for esophageal cancer. The AJCC system is based on a worldwide international database and novel statistical methods.
Because of the two different staging systems for esophageal cancer, surgeons and clinicians are confused about which staging system is appropriate for clinical situations and should be applied. A cancer staging system has to predict survival well and be easy to use. Therefore, this retrospective study investigated which staging system was more appropriate for esophageal cancer. Predictive powers for prognosis of the 7th and 8th AJCC staging systems and the 11th Japanese classification for esophageal cancer were calculated and compared.
Methods
This retrospective study was approved by the Institutional Review Board of our hospital (4-2017-0769). Records were retrospectively reviewed for 143 patients who underwent esophagectomy with mediastinal and abdominal lymphadenectomy for esophageal squamous cell carcinoma (ESCC) between January 2006 and July 2015. During the study periods, total 184 patients received the esophagectomy, and 20 patients received the neoadjuvant therapy. Exclusion criteria were neoadjuvant therapy, incomplete resection (R1 or R2 resection), salvage operation, operative mortality, and other pathology including esophageal adenocarcinoma. Because more than 90% of esophageal cancer in East Asia is squamous cell carcinoma, we analyzed patients with ESCC patients only for data consistency. Chest and abdominal computed tomography (CT), positron emission tomography-computed tomography (PET-CT), and esophagogastroduodenoscopy with endoscopic ultrasound were performed preoperatively. After operation, adjuvant chemotherapy based on cisplatin was done if patients have metastatic lymph nodes. In case of T3 or T4a, adjuvant radiotherapy was also performed. During follow-up, chest and abdomen CT scans were obtained at 6-month intervals, and esophagogastroduodenoscopy was conducted annually after operation. Locoregional recurrence was defined as occurring at the anastomosis site or mediastinum and abdomen where lymph node dissection was performed. Distant recurrence was defined as that found in tissues outside of the operative field such as lung, brain, liver, adrenal glands, bone, or other locations. Recurrence was diagnosed based on PET-CT scans and chest-abdomen CT scans, with tissue biopsy of suspected recurrent lesions performed if clinically possible. All patients were followed without loss.
Staging and lymph node map
As an institutional policy, surgical specimens were routinely dissected after the operation to accurately define lymph node stations. We applied a Japanese lymph node map for defining lymph node station and pathology staging (2). Because the AJCC system and Japanese classification had different lymph node maps, all dissected lymph nodes were reclassified according to the AJCC 7th and 8th lymph node map of esophageal cancer (3,4). Based on these results, patients were assigned three stages based on 7th AJCC staging system, 8th AJCC staging system, and 11th Japanese classification.
Statistical analysis
Statistical analysis used the open source statistical software R (http://www.R-project.org). Clinical and pathological parameters were described as mean ± standard deviation for continuous variables and frequency (%) for categorical variables. Overall survival (OS) was measured from date of operation to date of death from any cause or last clinical follow-up. Disease-free survival (DFS) was measured from date of operation to date of recurrence or death from any cause or last clinical follow-up. The Kaplan-Meier method and log-rank test were used for univariate analysis for survival, and a Cox proportional hazard model was used to calculate hazard ratios (HRs). Survcomp packages for performance assessment and calculation of concordance index (C-index) were used to calculate predictive power for survival and to compare Cox proportional hazard models (5). All tests were two-sided, and the criterion for significance was P<0.05.
Results
Patient demographics, recurrences, and survival
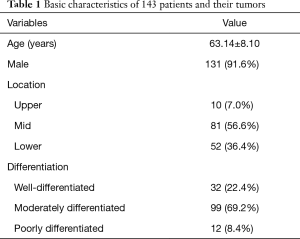
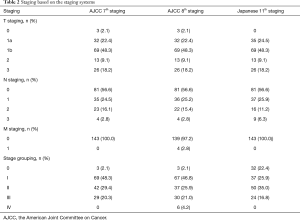
Basic patient characteristics are described in Table 1. Mean patient age was 63.14±8.10 years, and 131 (91.6%) patients were men. Median follow-up was 47.73 (6.27–134.40) months. Patients were classified in three stages (Table 2). Although T stagings were similar for the three staging systems, and in situ lesions were defined as T1a in the Japanese classification and Tis in the AJCC system. Patients showed different N and M staging. Four patients had metastatic lymph nodes at the supraclavicular fossa with M0 disease based on the AJCC 7th edition lymph node map, upstaged to M1 in the AJCC 8th edition because that map defines supraclavicular lymph node metastasis as distant metastasis. Patients also showed different stage groupings (Table 2).
Full table
Full table
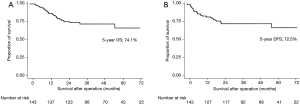
For all 143 patients, 5-year OS was 74.1% and DFS was 72.5% (Figure 1A,B). During follow-up, recurrences developed in 30 (20.9%) patients. Locoregional recurrences occurred in 4 (2.8%) patients; 2 in upper mediastinum, 1 subcarinal lymph node, 1 in right supraclavicular fossa. Distant recurrences occurred in 26 (18.2%) patients.
Comparison of staging systems according to N staging
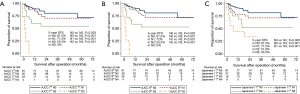
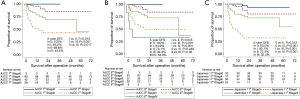
Survival differences were measured according to N staging. Based on N staging of the AJCC 7th staging system, DFS was well stratified (Figure 2A). The 5-year DFS by stage was 81.5% for N0, 71.3% for N1, 55.3% for N2, and 0% for N3 (P<0.001). Based on N staging of the AJCC 8th staging system, DFS was well stratified (Figure 2B). The 5-year DFS was 81.5% for N0, 72.0% for N1, 53.2% for N2, and 0% for N3 (P<0.001). For the Japanese N staging system, DFS was well stratified (Figure 2C). The 5-year DFS was 81.5% for N0, 71.2% for N1, 51.6% for N2, and 33.3% for and N3 (P<0.001). For patients with N3 cancer, N3 in the Japanese staging system had a higher survival than N3 in the AJCC staging system.
HRs for N staging for each stage system are documented in Table 3. HRs increased as N stage increased for each staging system. C-indexes for N staging were 0.755 [95% confidence interval (CI), 0.650–0.860] for AJCC 7th staging, 0.755 (95% CI, 0.650–0.860) for AJCC 8th staging, and 0.734 (95% CI, 0.634–0.835) for the Japanese 11th system. C-indexes for AJCC N staging was slightly higher than for the Japanese classification, with no significant different (AJCC 7thvs. Japanese 11th P=0.11, AJCC 8thvs. Japanese 11th P=0.11).
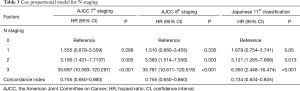
Full table
Comparison of the three staging systems according to grouping
Survival differences were measured according to stage grouping. Based on stage grouping for the AJCC 7th staging system, DFS was well stratified (Figure 3A). The 5-year DFS was 100% for patients with stage 0 cancer, 85.2% for stage I, 69.6% for stage II, and 43.4% for stage III (P<0.001). Based on stage grouping of the AJCC 8th staging system, DFS was well stratified (Figure 3B). The 5-year DFS was 100% for patients with stage 0 cancer, 84.9% for stage I, 73.1% for stage II, 49.3% for stage III, and 33.3% for stage IV (P<0.001). For the Japanese stage-grouping system, DFS was well stratified (Figure 3C). The 5-year DFS was 93.5% for patients with stage 0 cancer, 80.1% for stage I, 72.3% for stage II, and 32.4% for stage III (P<0.001).
HRs for stage groping for each staging system are documented in Table 4. HRs increased as N stage increased for each staging system. C-indexes for staging grouping was 0.813 (95% CI, 0.732–0.896) for AJCC 7th staging, 0.805 (95% CI, 0.726–0.885) for AJCC 8th staging, and 0.837 (95% CI, 0.766–0.908) for 11th Japanese classification. The c-index for staging grouping for the Japanese system was slightly higher than the c-indexes for the AJCC system, although without significant differences (AJCC 7thvs. AJCC 8th P=0.250, AJCC 8thvs. Japanese 11th P=0.964, and AJCC 7thvs. Japanese 11th P=0.837).

Full table
Discussion
Cancer stage at the time of diagnosis is a key factor for defining prognosis and is a critical element in determining appropriate treatment based on the experience and outcomes of groups of patients with similar stage. A widely used cancer staging system is based on the TNM system developed by Pierre Denoix in the 1940s. For staging esophageal cancer, the AJCC/UICC TNM staging system is applied worldwide, although the Japanese classification is also used in Japan. Each staging system has its own characteristics. The first UICC esophageal cancer staging system was proposed in 1968, and the first AJCC staging system in 1977 (6). Since 1987, close collaboration between the AJCC and the UICC has resulted in uniform and identical definitions and stage groupings of cancers for all anatomical sites. At the request of the AJCC, the WECC was inaugurated in 2006 (6). The 7th and 8th AJCC/UICC staging systems were proposed by WECC. The 8th staging system analyzed the data of 22,653 patients from 33 WECC institutions in 6 continents and 13 countries (7). Since the 7th AJCC staging system, this system has applied modern machine-learning analysis and random forest analysis (8). Therefore, the AJCC esophageal cancer stage system is a scientific staging system based on large, worldwide databases and novel statistical techniques. However, Japanese institutions did not participate in WECC, and Japanese data were not included in the WECC database (7). Japanese institutions proposed the first edition of Japanese classification in 1969, and the latest is the 11th edition published in 2015 (2). The recent Japanese classifications are based on a comprehensive registry of esophageal cancer with very high consistency in surgical policies and techniques whereas WECC data included different countries and surgical policies.
The major differences in the two staging systems are in N category and stage grouping. In the AJCC staging system, N category is classified by the number of metastatic lymph nodes. In the Japanese classification, the N category is classified by the spread of metastasis-positive lymph nodes. The AJCC N category reflects the burden of the primary tumor, such as malignancies in the gastrointestinal tract, breast, and kidney. This system is easy to use with surgical specimens, but difficult to apply for preoperative clinical staging. The current preoperative imaging system for the work-up of esophageal cancer has limitations in resolution (9). The accurate prediction of the number of metastatic lymph nodes in clinical staging is nearly impossible for esophageal cancer and other malignancies. Inaccuracy and the high possibility of N stage migration are major limitations of the N staging system based on the number of metastatic lymph nodes. In contrast, the N category of the Japanese classification is based on the spread of lymph nodes, which is complex because the definition of N differs according to the location of the primary lesion. Pathologists have difficulty defining lymph node stations in resected pathologic specimens. Therefore, the operator must dissect lymph nodes from surgical specimens to define lymph nodes stations accurately. In spite of these complexities, the Japanese classification can give additional information to surgeons about the extent of lymphadenectomy by N category. In addition to N category, the Japanese classification defines the concepts of D1–D3 dissection, which indicates extent of lymphadenectomy. Using these definitions, a surgeon can plan for extent of dissection based on the location of primary lesions. Similar to these systems, the N categories of the AJCC system and Japanese classification have their own advantages and limitations. For survival prediction, these two N category systems were similar in our results. This finding implied that both N categories could be applied in clinical situations for survival prediction.
Another difference between the two systems is stage grouping. In the AJCC system, prognostic stage grouping and anatomic stage grouping are used. The AJCC system included non-anatomical factors into stage classification to improve the prediction power of survival. Differentiation and location have been included in prognostic staging in the AJCC system since the 7th edition. In the 8th edition, the AJCC system had different staging groupings according to treatment status: clinical, pathological, and neoadjuvant pathologic (7,10,11). In addition, the AJCC staging system has different stage groupings according to pathology. Esophageal adenocarcinoma and squamous cell carcinoma have had different stage groupings since the 7th edition. Finally, in the 8th edition, the AJCC system has five stage-grouping systems staging groupings according to treatment status (clinical, pathological, and neoadjuvant pathologic stage) and pathology. As a result, the AJCC system has five stage grouping systems in the 8th edition (3). This strategy results in complex stage grouping that is difficult to apply in clinical situations. The original intent of the TNM system was to be a staging system based on anatomical factors only. The inclusion of non-anatomical factors in the TNM staging system does not follow the original intent of the TNM system. In contrast, the Japanese classification does not include non-anatomical factors such as differentiation and location in stage grouping and it has uniform stage grouping regardless of pathology and treatment status. Therefore the stage grouping of Japanese system is easy to use. A comparison of the three stage groupings showed that the stage grouping of the Japanese classification showed slightly better survival prediction although not significantly different. This finding implies that even a simple stage grouping without non-anatomical factors could have similar or slightly better survival prediction compared to a complex stage grouping system. If a simple and easy-to-use staging system is desired, further editions of stage groupings need to be simplified.
Each staging system had its own lymph node map and definition of regional lymph nodes. For example, the supraclavicular lymph nodes (No. 1 in the AJCC 7th edition and No. 104 in the 11th Japanese classification) are defined as regional lymph nodes in the AJCC 7th edition and the 11th Japanese classification but as distant lymph nodes in the AJCC 8th edition. In the 11th Japanese classification, lymph node metastasis to the supraclavicular lymph nodes is defined as N2 in cervical, upper, and middle thoracic esophageal cancer; N3 in lower thoracic esophageal cancer; and distant lymph nodes in abdominal esophageal cancer. If patients have suspicious metastatic lymph nodes in the supraclavicular area, surgical indications could differ based on staging system. In the 11th Japanese classification, a patient would have loco-regional disease and could be indicated for surgical resection; in the AJCC 8th edition, the patient would have distant metastasis, and surgical resection would not be indicated. The issue of which staging system to use is related to prediction of survival, convenience of use, and selection of the indication for surgery. However, the issues of which lymph node map is appropriate for esophageal cancer can be studied only in a comparison of survival predictions. In our data, only 4 patients had metastasis in the supraclavicular lymph nodes, and the survival differences according to supraclavicular lymph node metastasis could not be analyzed. To study this issue, a large cohort is needed.
Our study has several limitations. First, we analyzed patients with ESCC and excluded patients who received neoadjuvant treatment. Because more than 90% of esophageal cancers are ESCCs in East Asia and a small number of patients received neoadjuvant therapy in our institution, we analyzed pathologic stage only in patients with ESCC. For esophageal adenocarcinoma, our results should be applied cautiously. Second, differences such as in the definition of esophagogastric junction tumors were not analyzed. Last, the number of patients was relatively small. Although stage grouping changed from the AJCC/UICC 7th to 8th editions, the predictive powers for survival did not increase in our data. This result might be related to the small number of patients. In the AJCC staging system, stages I, II, and III are subdivided into IA, IB, IIA, IIB, IIIA, and IIIB, but we did not calculate survival differences between subdivided stages due to the small number of patients.
In conclusion, N staging and stage grouping of the 11th Japanese classification and 7th and 8th AJCC staging systems for ESCC showed similar predictive power for DFS. Both systems can be applied in clinical situations.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This retrospective study was approved by the Institutional Review Board of our hospital (4-2017-0769).
References
- Rice TW, Blackstone EH, Rusch VW. 7th edition of the AJCC Cancer Staging Manual: esophagus and esophagogastric junction. Ann Surg Oncol 2010;17:1721-4.
- Japan Esophageal Society. Japanese Classification of Esophageal Cancer, 11th Edition: part I. Esophagus 2017;14:1-36.
- Rice TW, Ishwaran H, Ferguson MK, et al. Cancer of the Esophagus and Esophagogastric Junction: An Eighth Edition Staging Primer. J Thorac Oncol 2017;12:36-42.
- Greene F, Page D, Fleming I. AJCC Cancer Staging Manual. New York: Springer, 2002.
- Schröder MS, Culhane AC, Quackenbush J, et al. survcomp: an R/Bioconductor package for performance assessment and comparison of survival models. Bioinformatics 2011;27:3206-8. [Crossref] [PubMed]
- Rice TW, Blackstone EH. Esophageal cancer staging: past, present, and future. Thorac Surg Clin 2013;23:461-9. [Crossref] [PubMed]
- Rice TW, Apperson-Hansen C, DiPaola LM, et al. Worldwide Esophageal Cancer Collaboration. clinical staging data. Dis Esophagus 2016;29:707-14. [Crossref] [PubMed]
- Ishwaran H, Blackstone EH, Apperson-Hansen C, et al. A novel approach to cancer staging: application to esophageal cancer. Biostatistics 2009;10:603-20. [Crossref] [PubMed]
- Park SY, Kim DJ, Jung HS, et al. Relationship Between the Size of Metastatic Lymph Nodes and Positron Emission Tomographic/Computer Tomographic Findings in Patients with Esophageal Squamous Cell Carcinoma. World J Surg 2015;39:2948-54. [Crossref] [PubMed]
- Rice TW, Chen LQ, Hofstetter WL, et al. Worldwide esophageal cancer collaboration: pathologic staging data. Dis Esophagus 2016;29:724-33. [Crossref] [PubMed]
- Rice TW, Lerut TE, Orringer MB, et al. Worldwide Esophageal Cancer Collaboration. neoadjuvant pathologic staging data. Dis Esophagus 2016;29:715-23. [Crossref] [PubMed]


