Pulmonary alveolar proteinosis
Introduction
Pulmonary alveolar proteinosis (PAP) is an uncommon pulmonary disease characterised by abnormal excessive alveolar accumulation of surfactant. We present a case of PAP in an elderly female that was successfully treated with whole lung lavage.
Case report
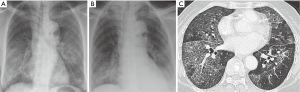
A 68-year-old female presented to the Respiratory Clinic for chronic cough and dyspnoea of six months duration. There was no fever or weight loss. Her symptoms did not improve despite previous courses of antibiotics and oral steroids. She had a medical history of ischemic heart disease for which she was on clopidogrel, telmisartan, metoprolol and rosuvastatin. There was otherwise no significant family or contact history. She denied exposure to cigarette smoke, occupational hazards or toxic fumes. Respiratory examination revealed scattered lung crepitations. Cyanosis and clubbing were not noted. Pulse oximetry on room air was 93%. Her leucocyte count at presentation was 9.59×109/L (normal range 4×109-10×109/L) and serum LDH was elevated at 1,282 U/L (normal range 180-380 U/L). A chest radiograph was performed which showed bilateral alveolar opacities (Figure 1). A CT thorax demonstrated bilateral ground-glass opacities and septal reticulations forming a “crazy paving pattern” (Figure 1C).
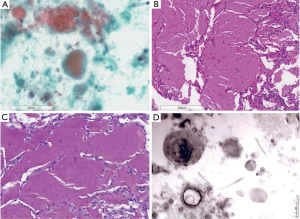
Bronchoalveolar lavage revealed grainy eosinophilic debris (Figure 2) whilst transbronchial lung biopsy of the left lower lobe demonstrated preserved alveolar spaces filled with amorphous grainy material (Figure 2B,C). Lavage cultures were negative. A diagnosis of PAP was made.
The bronchoscopy was complicated by hemoptysis which resolved spontaneously and a left pneumothorax which necessitated placement of a left chest tube. She subsequently underwent two sequential whole lung lavages under general anesthesia, with the aim of treating the left lung first followed by the right lung during the second lavage 48 hours later. It was anticipated that the second lavage would pose a ventilator challenge as single lung ventilation would be performed on the lung affected by the previous hemorrhage and pneumothorax. Apart from frequent bronchoscopy to aspirate any spillover lavage fluid, we opted to reduce the volume of each aliquot as guided by oxygen saturations. The lowest oxygen saturation recorded was 82% (on 100% inspired oxygen fraction) which was transient, and this improved to 100% at the end of the lavage. In total, 16.1 L of saline was used for the first procedure, and 13 L was used for the second lavage.
The patient was monitored in the intensive care unit for 24 hours after each lavage. Post procedural chest X-rays did not demonstrate pneumothoraces, allowing removal of the chest tube. Her symptoms improved and at time of discharge 48 hours after the second lavage, her pulse oximetry was 96% on room air.
Discussion
PAP is an uncommon condition that was first described in a 27-patient case-series by Rosen et al. in 1958 (1). This condition arises from defective clearance and accumulation of surfactant in pulmonary alveoli, for which granulocyte macrophage-colony stimulating factor (GM-CSF) plays a critical role in enabling surfactant catabolism by alveolar macrophages. Understanding the pathogenesis of this disease was made possible in 1994 when it was reported that knockout mice that had GM-CSF deficiency developed similar disease (2). This knowledge was enhanced by the finding of a neutralizing IgG antibody against GM-CSF (3,4), and this autoantibody was subsequently noted to also impair neutrophil function, predisposing patients to increased risk of infections (5). Based on the etiology, three forms are currently recognized (6): genetic (arising from mutations in surfactant proteins or GM-CSF receptor genes), autoimmune, and secondary to toxic inhalation or haematological disorders. Autoimmune PAP is the most common form (representing about 90% of all cases) (7).
Common clinical manifestations are cough and dyspnea occurring in the third to sixth decades, although fever, chest pain and hemoptysis may also occur. The diagnosis is suspected by a CT thorax demonstrating the characteristic “crazy paving” pattern of reticulations superimposed on ground glass opacities, the latter arising from the accumulation of lipoproteinaceous material in the alveolar spaces (8). Bronchoalveolar lavage confirms the diagnosis by demonstrating foamy macrophages and eosinophilic granules that are positive for periodic acid-Schiff (PAS) staining (9).
Five-year survival without treatment has been reported to be 85% (10). Whole lung lavage is the first-line therapy, with the purpose of washing out the lipoproteinacous material from the alveoli (11) (Figure 3). It improves symptoms 5-year survival to 94% (8). Symptoms, radiologic abnormalities and physiologic parameters are also improved. The duration of response is currently unclear, with the median duration of benefit reported to be 15-36 months (10,12). Up to 66% of patients may require a repeat lavage (10). Given the rarity of PAP, proper prospective studies evaluating the optimal technique, safety and duration of benefit have not been performed. Complications of whole lung lavage include low oxygen saturation, hemodynamic instability, convulsions, pneumothorax, pleural effusion and fever have been reported. Whilst generally 15-20 L of saline (based on aliquots of 1 L) is required for each lavage to produce clinical improvement, this must be weighed against the potential risk of peri-procedural desaturations. We suggest adjusting the volumes of aliquots to avoid precipitous drops in oxygen saturations.

Corticosteroids are not recommended for treating PAP (10). Other promising therapies for autoimmune PAP include GM-CSF supplemental therapy (13) or aiming to reduce levels of anti-GM-CSF antibodies by using plasmapheresis (14) or rituximab (15), although these therapeutic options require further studies.
Acknowledgements
The authors contributed equally on this report. The authors would also like to thank Antonio de Guzman Jr., histotechnologist, for his help in the production of the electron microscopy photograph.
Disclosure: The authors declare no conflict of interest.
References
- Rosen SH, Castleman B, Liebow AA. Pulmonary alveolar proteinosis. N Engl J Med 1958;258:1123-42. [PubMed]
- Dranoff G, Crawford AD, Sadelain M, et al. Involvement of granulocyte-macrophage colony-stimulating factor in pulmonary homeostasis. Science 1994;264:713-6. [PubMed]
- Tanaka N, Watanabe J, Kitamura T, et al. Lungs of patients with idiopathic pulmonary alveolar proteinosis express a factor which neutralizes granulocyte-macrophage colony stimulating factor. FEBS Lett 1999;442:246-50. [PubMed]
- Kitamura T, Tanaka N, Watanabe J, et al. Idiopathic pulmonary alveolar proteinosis as an autoimmune disease with neutralizing antibody against granulocyte/macrophage colony-stimulating factor. J Exp Med 1999;190:875-80. [PubMed]
- Uchida K, Beck DC, Yamamoto T, et al. GM-CSF autoantibodies and neutrophil dysfunction in pulmonary alveolar proteinosis. N Engl J Med 2007;356:567-79. [PubMed]
- Trapnell BC, Whitsett JA, Nakata K. Pulmonary alveolar proteinosis. N Engl J Med 2003;349:2527-39. [PubMed]
- Inoue Y, Trapnell BC, Tazawa R, et al. Characteristics of a large cohort of patients with autoimmune pulmonary alveolar proteinosis in Japan. Am J Respir Crit Care Med 2008;177:752-62. [PubMed]
- Holbert JM, Costello P, Li W, et al. CT features of pulmonary alveolar proteinosis. AJR Am J Roentgenol 2001;176:1287-94. [PubMed]
- Wells AU. The clinical utility of bronchoalveolar lavage in diffuse parenchymal lung disease. Eur Respir Rev 2010;19:237-41. [PubMed]
- Seymour JF, Presneill JJ. Pulmonary alveolar proteinosis: progress in the first 44 years. Am J Respir Crit Care Med 2002;166:215-35. [PubMed]
- Michaud G, Reddy C, Ernst A. Whole-lung lavage for pulmonary alveolar proteinosis. Chest 2009;136:1678-81. [PubMed]
- Brach BB, Harrell JH, Moser KM. Alveolar proteinosis. Lobar lavage by fiberoptic bronchoscopic technique. Chest 1976;69:224-7. [PubMed]
- Kavuru MS, Sullivan EJ, Piccin R, et al. Exogenous granulocyte-macrophage colony-stimulating factor administration for pulmonary alveolar proteinosis. Am J Respir Crit Care Med 2000;161:1143-8. [PubMed]
- Luisetti M, Rodi G, Perotti C, et al. Plasmapheresis for treatment of pulmonary alveolar proteinosis. Eur Respir J 2009;33:1220-2. [PubMed]
- Borie R, Debray MP, Laine C, et al. Rituximab therapy in autoimmune pulmonary alveolar proteinosis. Eur Respir J 2009;33:1503-6. [PubMed]


