X-ray indices of chest drain malposition after insertion for drainage of pneumothorax in mechanically ventilated critically ill patients
Introduction
Therapeutic chest drainage of a pneumothorax (PNO) is usually performed through the safe triangle just under the lateral border of the pectoralis muscle (1,2). Successful drainage of a PNO results in full re-expansion of the lung confirmed by chest ultrasound or less reliably by plain chest radiograph (CXR) (3-8).
Ideally, a CD inserted through the safe triangle runs anteriorly apically between the anterior chest wall and the anterior visceral pleura. This position guarantees an effective evacuation of PNO and offers optimal CD function in a supine patient. However, even a correctly placed CD may migrate in the pleural cavity ultimately assuming a more dependent position. Since the lung cross-section tapers in the cranial direction, a CD may turn laterally and ultimately fall dorsally.
The position of a CD introduced in the pleural cavity is usually assessed on a CXR, less commonly by chest ultrasound or by computed tomography (CT). A CXR provides a superimposed image where the anteroposterior dimension is not apparent. Only indirect signs based on projection and mutual relationship between structures are mostly used to estimate the position of objects in the anteroposterior dimension (9). To our best knowledge, there has not been published a single study concerning the correct position of a CD for the drainage of PNO on CXR.
In this pilot study, we hypothesized that several simple CXR parameters might be helpful in detecting whether a CD introduced in the pleural space for PNO drainage from the safe triangle changed its location and slipped laterally or posteriorly.
Methods
The study was performed between May 2015 and June 2017 in a 20-bed intensive care unit in a tertiary non-trauma center. It was approved by the Institutional Review Board and informed consent was waived due to retrospective design of the study.
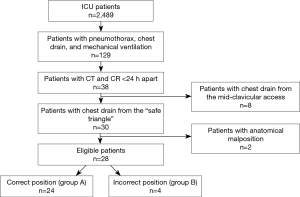
The inclusion criteria were: (I) the presence of a CD in the pleural space inserted for PNO drainage from the safe triangle; (II) mechanical ventilation; (III) CXR and CT scan with the drain performed less than 24 hours apart. The exclusion criteria were: (I) all anatomical malpositions (extrathoracic, intraparenchymal, interlobar) as they are inherently malpositioned; (II) mid-clavicular access.
All CDs were 16–20 F in size and they were inserted using blunt dissection with forceps above the upper rib edge. Trocar assistance was strictly limited to facilitating the passage of the CD only through the chest wall, i.e., the CD was pulled off the trocar after reaching the pleural space with its soft tip directed under the anterior chest wall towards the sternoclavicular joint. All drains were connected to a closed suction system providing a pressure of −20 mbar (Aqua-Seal, Covidien, Mansfield, MA, USA).
Twenty-eight patients were finally included in the study (for study flowchart see Figure 1). The patients were divided according to the position of the CD on the CT in two groups: group A with the tip of the CD anteriorly to the mid-axillary line (correct placement, n=24) and group B with the tip of the CD at the level of or posteriorly to the mid-axillary line (incorrect placement, n=4).
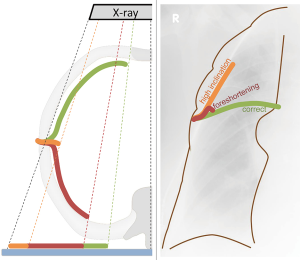
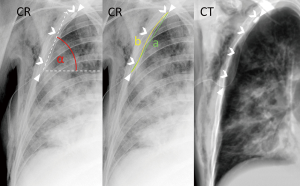
The parameters measured on the CT and CXR are listed in Table 1. Foreshortening of the CD was calculated as the distance from chest entry to its tip in the coronal plane (ignoring the anteroposterior dimension) divided by its true length obtained by three-dimensional measurement. The angle of inclination of the CD above the horizontal line at chest entry was measured on CXR (Figures 2,3). CD tortuosity was determined as the ratio of a straight distance of from chest entry of the CD to its tip and the length of the CD in the patient measured on CXR (Figures 2,3). The measurements on CT and CXR images were performed by a board-certified radiologist with >10 years’ experience in thoracic imaging.

Full table
The data was analyzed in GraphPad Prism (GraphPad Software, La Jolla, USA). The normality of the data was tested using the D'Agostino & Pearson omnibus test and statistical significance between the groups was tested by the t-test or the Mann-Whitney test as appropriate with subsequent ROC analysis. A P value below 0.05 was considered significant.
Results
The patients were 59.5±15 years old and 78% were males. All patients were mechanically ventilated with APACHE II 23±6, SOFA 8.9±2.5. The etiology of PNO included postsurgical (n=10), ventilator related in pneumonia (n=7), ventilator related in ARDS requiring ECMO (n=6), postprocedural (n=3), and spontaneous on intermittent positive pressure ventilation (IPPV, n=2).
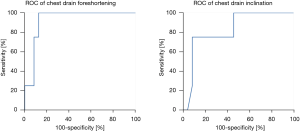
The parameters measured on CT and CXR are presented in Table 1. Greater CD foreshortening was the best clue of a misplaced CD with AUC of 0.93 (95% CI: 0.83–1.0, P=0.0071), 100% sensitivity and 88% specificity for a cut-off value of 82% (Table 1, Figure 4). The angle of CD inclination was greater in patients with misplaced CD with AUC of 0.83 (95% CI: 0.63–1.0, P=0.039), 75% sensitivity and 92% specificity for a cut-off value of 50 degrees (Table 1, Figure 4). There was no significant difference in CD tortuosity on CXR between the groups with AUC of 0.69 (95% CI: 0.34–1.0, P=0.22).
Three of the four patients with migrated CDs had a residual ventral PNO on the CT scan. The median time between the CXR and CT examinations was 5.4 hours (IQR 3.1).
Discussion
This pilot study evaluated simple parameters readily available from a plain CXR that would raise suspicion of CD migration after insertion for PNO drainage through the safe triangle in non-trauma critically ill mechanically ventilated patients. To our best knowledge, this is the first study to systematically evaluate malposition of a chest drain (CD) for PNO on a plain CXR.
In this pilot study, three of the four patients with malpositioned CDs had a residual ventral PNOs confirmed on CT. The data on how to proceed with an occult or residual PNO in mechanically ventilated patients with primary respiratory failure are scarce and conflicting. While some suggested that hemodynamically stable mechanically ventilated patients should be only observed (10,11), other studies were inconclusive (12). In contrast to occult PNOs in trauma that do not always require chest drainage (10-12), the rates of PNO progression may be higher in non-trauma patients and those with respiratory failure on aggressive IPPV in particular. Even a small PNO is still important on IPPV because it may easily enlarge due to positive inspiratory pressure (8). A secondary occult or small residual ventral PNO may limit lung recruitment, increase requirements for mechanical ventilation, prevent weaning, and even progress to a life-threatening emergency in the form of a tension PNO with its consequences (13-17).
The method used for the insertion of the CD combined a surgical and a modified trocar technique. Its main objective was to reduce the incidence of functional malposition due to intrapleural angulations commonly encountered with a pure surgical technique, and anatomical intraparenchymal or fissural malpositions related to the advancement of the trocar into the chest (18,19). Most indications for chest drainage are based on a combination of clinical signs, ultrasound assessment (8,20,21), and CXR. CT scans in our patients were not requested for the evaluation of PNO of the CD (19). These may be the reasons for a small number of patients included in the study and even smaller number of patients with CD malposition. By no means this pilot study attempts to quantify functional malpositions of CDs inserted for PNOs but rather explores potential ways of their diagnosis.
The detection of malposition of CD inserted for PNO in supine CXR is difficult, because it is not readily obvious and there are no specific signs. In this study, we assumed that a migrating CD inserted from the safe triangle would turn upwards and laterally and later dorsally and that this trajectory would result in the CD pointing steeply up after entering the pleural cavity and later foreshortening on CXR (Figure 5). The angle of inclination which can be directly measured on CXR had only a moderate sensitivity of CD malposition. Foreshortening of CD was a better performing parameter with AUC of 0.93. Foreshortening is based on the elementary principle of projection in plain radiography (9) and the knowledge of the length of CD inside the pleural cavity that can be read on the rule printed on the CD. CD tortuosity can be directly measured on CXR, but its performance in detecting CD malposition is poor. Compared to the inclination of the CD, both CD tortuosity and foreshortening have a greater risk of bias resulting from radiographic and measurement techniques. Moreover, foreshortening requires the knowledge of the distance of insertion.
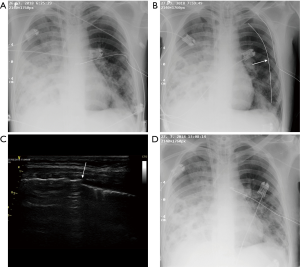
The CD inserted through the safe triangle for a PNO should resemble “bird wings” when viewed on bedside CXR. Different configurations and kinks should raise suspicion of its malposition. Although one may observe the change of CD position between two sequential radiographs, the extent to which the CD has migrated is difficult to assess. Furthermore, performing routine daily radiographs is no longer a recommended practice (22).
The limitations of this study are multiple. Firstly, the authors’ practice adhered to current recommendations (1,2) and excluded anterior insertions from the midclavicular line. Secondly, the parameters measured on CXR may have been influenced by the radiographic technique (center, deviation from the anterio-posterior axis, distance). Thirdly, the number of patients especially those with malposition of the CD was small. Fourthly, only patients with CT and CXR less than 24 hours apart were included which constitutes a selection bias. Lastly, this pilot study was conducted retrospectively. Nevertheless, its main findings, the utility of CD foreshortening and CD inclination angle, warrant further investigation.
Conclusions
This pilot study evaluated simple parameters measured or derived from bedside CXR that could raise suspicion of CD malposition after insertion for PNO drainage through the safe triangle in non-trauma mechanically ventilated critically ill patients. Greater foreshortening of the CD and a steep angle of inclination of the CD above the horizontal at chest entry should raise suspicion of CD migration and mandate further investigation by chest ultrasound to rule out residual pneumothorax occult on CXR.
Acknowledgements
Funding: This study was supported in part from project reg. no. CZ.2.16/3.1.00/21565 from OP Prague Competitiveness. The supporting source had no involvement in the preparation of this paper nor study execution.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved (2370/15 S-IV) by the General University Hospital Institutional Review Board and written informed consent was waived for the collection and analysis of anonymous data for the purpose of this retrospective study.
References
- Havelock T, Teoh R, Laws D, et al. Pleural procedures and thoracic ultrasound: British Thoracic Society pleural disease guideline 2010. Thorax 2010;65 Suppl 2:ii61-76. [Crossref] [PubMed]
- Elsayed H, Roberts R, Emadi M, et al. Chest drain insertion is not a harmless procedure - are we doing it safely? Interact Cardiovasc Thorac Surg 2010;11:745-8. [Crossref] [PubMed]
- Martinez T, Pasquier P, Swiech A, et al. Lung ultrasound for chest tube insertion. Am J Emerg Med 2015;33:1095-6. [Crossref] [PubMed]
- Alrajab S, Youssef AM, Akkus NI, et al. Pleural ultrasonography versus chest radiography for the diagnosis of pneumothorax: review of the literature and meta-analysis. Crit Care 2013;17:R208. [Crossref] [PubMed]
- Roberts DJ, Niven DJ, James MT, et al. Thoracic ultrasonography versus chest radiography for detection of pneumothoraces: challenges in deriving and interpreting summary diagnostic accuracy estimates. Crit Care 2014;18:416. [Crossref] [PubMed]
- Soldati G, Testa A, Sher S, et al. Occult traumatic pneumothorax: diagnostic accuracy of lung ultrasonography in the emergency department. Chest 2008;133:204-11. [Crossref] [PubMed]
- Blaivas M, Lyon M, Duggal S. A prospective comparison of supine chest radiography and bedside ultrasound for the diagnosis of traumatic pneumothorax. Acad Emerg Med 2005;12:844-9. [Crossref] [PubMed]
- Brook OR, Beck-Razi N, Abadi S, et al. Sonographic Detection of Pneumothorax by Radiology Residents as Part of Extended Focused Assessment With Sonography for Trauma. J Ultrasound Med 2009;28:749-55. [Crossref] [PubMed]
- Lacey G de, Morley S, Berman L. The Chest X-Ray: A Survival Guide. Elsevier Saunders; 2008. ISBN: 9780702030468.
- Ball CG, Dente CJ, Kirkpatrick AW, et al. Occult pneumothoraces in patients with penetrating trauma: Does mechanism matter? Can J Surg 2010;53:251-5. [PubMed]
- Kirkpatrick AW, Rizoli S, Ouellet JF, et al. Occult pneumothoraces in critical care: a prospective multicenter randomized controlled trial of pleural drainage for mechanically ventilated trauma patients with occult pneumothoraces. J Trauma Acute Care Surg 2013;74:747-54; discussion 754-5. [Crossref] [PubMed]
- Ouellet JF, Trottier V, Kmet L, et al. The OPTICC trial: a multi-institutional study of occult pneumothoraces in critical care. Am J Surg 2009;197:581-6. [Crossref] [PubMed]
- Baldt MM, Bankier AA, Germann PS, et al. Complications after emergency tube thoracostomy: assessment with CT. Radiology 1995;195:539-43. [Crossref] [PubMed]
- Lim KE, Tai SC, Chan CY, et al. Diagnosis of malpositioned chest tubes after emergency tube thoracostomy Is computed tomography more accurate than chest radiograph? Clin Imaging 2005;29:401-5. [Crossref] [PubMed]
- Ball CG, Hameed SM, Evans D, et al. The Canadian Trauma Trials Collaborative. Occult pneumothorax in the mechanically ventilated trauma patient. Can J Surg 2003;46:373-9. [PubMed]
- Enderson BL, Abdalla R, Frame SB, et al. Tube thoracostomy for occult pneumothorax: a prospective randomized study of its use. J Trauma 1993;35:726-9; discussion 729-30. [Crossref] [PubMed]
- Karnik AM, Khan FA. Pneumothorax and barotrauma. In: Parillo JE, Dellinger RP. editors. Critical care medicine: principles of diagnosis and management in the adult. 2nd ed. St. Louis: Mosby, 2001:930-48.
- Dural K, Gulbahar G, Kocer B, et al. A novel and safe technique in closed tube thoracostomy. J Cardiothorac Surg 2010;5:21. [Crossref] [PubMed]
- Remérand F, Luce V, Badachi Y, et al. Incidence of Chest Tube Malposition in the Critically Ill. A Prospective Computed Tomography Study. Anesthesiology 2007;106:1112-9. [Crossref] [PubMed]
- Volpicelli G, Elbarbary M, Blaivas M, et al. International Liaison Committee on Lung Ultrasound for International Consensus Conference on Lung Ultrasound (2012) International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med 2012;38:577-91. [Crossref] [PubMed]
- Lichtenstein D, Mezière G, Biderman P, et al. The ‘lung point’: an ultrasound sign specific to pneumothorax. Intensive Care Med 2000;26:1434-40. [Crossref] [PubMed]
- Hejblum G, Chalumeau-Lemoine L, Ioos V, et al. Comparison of routine and on-demand prescription of chest radiographs in mechanically ventilated adults: a multicentre, cluster-randomised, two-period crossover study. Lancet 2009;374:1687-93. [Crossref] [PubMed]