Mechanical circulatory support for refractory cardiogenic shock post-acute myocardial infarction—a decade of lessons
Introduction
In the preceding decades, Scotland has drastically reduced the mortality from coronary heart disease (72% reduction in 2009 compared to 1950) (1). Despite this, post-MI mortality remains among the highest in Western Europe (1), branding Scotland as the ‘sick man of Europe’ (2). The inequalities in Scottish morbidity and mortality has resulted in an overall increase in health inequalities across the United Kingdom (3). Ischaemic heart disease is associated with a higher level of disability-adjusted life years (DALY) than any other condition in Scotland, mirroring not just the UK, but also DALY in the Global Burden of Disease Survey (4).
There are 0.9 catheterization labs per 100,000 inhabitants in Scotland for percutaneous coronary intervention (PCI) for acute myocardial infarction (AMI) (5), which are much less accessible to patients in remote and rural areas. An uncommon but sinister sequalae following AMI is cardiogenic shock (CS) that could be refractory to inotropic support. CS complicates 5–15% of AMIs occurring in ST-segment elevation myocardial infarctions (STEMIs) (6-8). Outcomes of CS are poor with mortalities of up to 90% reported in the literature in the absence of experienced care (9).
Initial management of CS consists of identifying incidental complications e.g., acute left ventricular rupture or mitral regurgitation, assessing haemodynamics, and optimising the reperfusion in the culprit coronary artery. Clinical trials of therapeutic interventions have not led to changes in practice. The results of the Intra-Aortic Balloon Pump in Cardiogenic Shock II (IABP-SHOCK II) trial highlighted the lack of survival benefit from the routine use of IABP therapy for this condition (10). The only available option for patients with refractory, life-threatening illness would be the institution of mechanical circulatory support (MCS), involving either extracorporeal membranous oxygenation (ECMO) or ventricular assist devices (VADs). MCS can potentially improve survival, however, evidence is lacking. EURO-SHOCK (ID754946-2), which is a clinical trial funded by the EU-Horizons 2020 7th Framework programme, will address this gap. EURO-SHOCK is a multicentre, randomised, controlled trial of management involving ECMO vs. standard care without ECMO in patient with CS post-MI. Given the current gap in knowledge, we studied the outcomes following use of MCS for treatment of CS post-AMI in Scotland during an 8-year period.
Methods
Patients
All patients who were referred to the MCS service in the Golden Jubilee National Hospital from January 2009 to August 2017 following primary PCI-treated STEMI with refractory CS were included in this study.
A retrospective review of prospectively collected data was undertaken on all patients registered to the MCS service. The database was interrogated for patient demographics, type of MCS [veno-arterial ECMO (VA-ECMO)/VAD] and duration of MCS support, PCI-outcomes and survival to 30 days. A time-to-event analysis was performed using patient survival as the primary outcome measure. Kaplan-Meier curves were used to graphically display data of 30-day survival. Students t-tests and Mann-Whitney U tests were used to analyse data for 30-day survival for continuous data with Fisher’s exact test used for categorical data. The study was registered with the Clinical Governance and Audit Department at the Golden Jubilee National Hospital with a data protection registration number Z7996020.
Results
Twenty-three patients (16 male, 7 females) were included. The median age of the patients as 50 years (range, 45–56 years). VA-ECMO was the initial MCS of choice in 17 (73.9%) patients with BIVAD for 4 (17.4%) patients and LVAD for 2 (8.7%) patients. 30-day mortality was 21.8% in this cohort, however survival to discharge was 52.2%.
Eleven (47.8%) patients recovered without the need for any further support, however only 9 (81.8%) patients in this subgroup survived to discharge. Three (13.0%) patients received a durable LVAD. In this subgroup, one patient was transplanted whereas two patients died due to complications while on support (VAD thrombus, disseminated intravascular coagulation).
The median length of in-hospital MCS support was 4 days (range, 4–43 days). Median in-hospital stay was 27 days (range, 9–41 days).
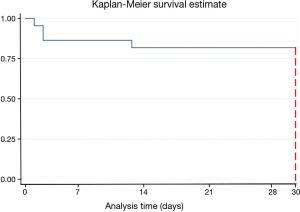
The 30-day mortality data of survivors vs. non-survivors are as follows (Figure 1, Tables 1-4).

Full table
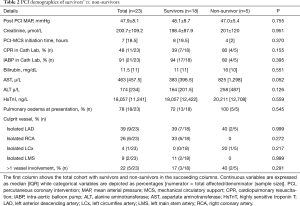
Full table

Full table

Full table
Removal of MCS
Death post-removal of MCS device were caused by malignant arrhythmia (n=2).
Post 30-day survival
Three (13.0%) of patients underwent heart transplantation and are well at up to 6 years post-operatively. Patients who were successfully weaned had a mean ejection fraction of 35.6%±10.2% and received ongoing heart failure medical therapy.
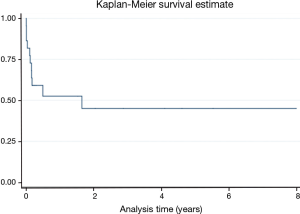
Long-term follow up of up to 8 years is depicted in Figure 2. The curve demonstrates a high mortality beyond 30-day up to the first 6-month post MCS support.
Discussion
For the first time, we have documented MCS therapy and related outcomes in a contemporary Scottish population of patients with AMI complicated by CS. More than 79.2% of patients survived to 30 days. This result compares favourably to other studies investigating outcomes of patients receiving MCS therapy in AMI/CS. In the ENCOURAGE study, approximately half of the cohort survived to 30 days (11). One other North American study reported a 30-day mortality rate of 41% (12). Most other publications report a 30-day survival rate of 23–76% survival rate in this specific patient cohort (13-15). Among the survivors, 16.7% subsequently underwent heart transplantation.
There are challenges to comparisons of outcomes in post AMI-CS patients between studies, not least because of the heterogeneity in patient populations and practice. comparing outcomes of existing studies however is complicated by the variability of the cohorts as CS comprises a wide spectrum of clinical and haemodynamic instability. There is substantial heterogeneity with presentations of the patients with several factors being predictors of poor outcome in larger studies. A literature review revealed older age (6,16), signs of end-organ hypo-perfusion (17), involvement of the LAD artery (16-18), severity of disease (triple vessel disease) (16) and renal failure (identified by elevated creatinine) (16,18). Our cohort was limited in size. Nonetheless, our findings indicate 30-day mortality results are similar to or potentially better than prior cohort studies.
We concentrated on the presenting pathology (AMI with CS) and not the device (VAD vs. ECMO) as we felt most patients would receive a strategy that was either escalated or de-escalated based on recovery. Patients who were improving for example were stepped down from ECMO to a VAD (short term or long term). ECMO was the treatment of choice in most patients as in the acute phase, almost all the patients presented with acute pulmonary oedema.
Almost half (47.8%) of the patients had their support successfully weaned from ECMO and/or VAD without any further support device or transplantation. Myocardial recovery has been reported in previous publications (19-21). VA-ECMO is readily available and can be rapidly instituted percutaneously negating the need for operating theatre resources. Some limitations to ECMO have been reported in the literature. This includes inadequate left ventricular decompression as emptying depends on the native ejection function of the ventricle. Decreasing the flow rate on the ECMO circuit also reduces afterload alongside using inotropes such as dobutamine to improve contractility and decrease ejection. This may result in pulmonary hypertension, oedema and bleeding (22). The interaction between the tubing surfaces causes activation of monocytes and release of interleukins 1 and 6 (23). Some of the decompression can be attenuated by IABP insertion. It is associated with a smaller left ventricular dimension and a lower pulmonary artery pressure by restoring pulsatility and decreasing left ventricular afterload (24). IABP may also reduce the mean of cerebral blood flow during myocardial stunning, and increases the mean flow during cardiac recovery (25). Activation of clotting cascades is the predominant reason for bleeding complications. Frequent echocardiograms are done at our unit to ensure there is adequate decompression of the right and left ventricles. Another deleterious effect of VA ECMO is the neurological morbidity. Brain death has been reported in up to 21% in adults treated in ECMO centres. Up to 50% of patients have evidence of cerebral injury (25). In our cohort, 26.1% of patients had evidence of a cerebral injury. The same deleterious effects of ECMO are also noted in VADs (about 20%) (26).
Myocardial ischaemia is commonly the preceding event in CS (9,27). It impairs myocardial contractility which in turn reduces stroke volume. An impeded cardiac index causes tissue hypoperfusion, which includes coronary hypoperfusion causing worsening myocardial ischaemia, resulting in a vicious cycle. Serum lactate, creatinine and AST are used as surrogates of organ hypoperfusion in our study. Initial compensatory vasoconstriction arises from catecholamine release to increase blood pressure but systemic inflammatory response syndrome (SIRS) mediated pathological release of vasodilatory agents results in a net reduction in cardiac index. This acts in conjunction with the reduction in left ventricular function as a result of myocardial stunning from the primary insult. There is a small window of reversibility afforded during myocardial stunning by reperfusion which is facilitated by early reperfusion (28). Capillary leakage from SIRS causes tissue oedema and a reduction in circulating volume.
Decision making for MCS is also an important part of the discussion. Traditional ethical principles are not straightforward when applied to ECMO patients as it is often seen as the ceiling of therapy available. A survey of self-reported physicians with vast experience in VA-ECMO revealed majority of physicians felt physicians should have the right to discontinue management over family’s objection (29). MCS is a costly intervention thereby complicating the decision-making process with finite resources available for clinicians in the National Health Service (NHS). In our unit, a multidisciplinary team is consulted to ensure an informed decision which takes into account all facets of care prior to initiating MCS support.
There are still some limitations in this study. The data presented represents the first reported series of patients in Scotland with AMI complicated by CS treated with MCS. However, as it is a retrospective study with a small cohort of patients, the reproducibility of the results may vary and may not capture the European or British population as a whole. There is a selection bias in the sample as only patients who were deemed potentially salvageable were included in the study, which may comparisons with other studies difficult.
Conclusions
MCS usage in these patients carries a high mortality in the early post-implantation period. However, there is a significant benefit to patients who survive the initial bridging period to recovery or destination therapy. Further prospective studies are needed to identify predictors of long-term survival.
Acknowledgements
The authors wish to thank Clare Connolly for providing the necessary administrative assistance for the procurement of data.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by Clinical Governance Department, Golden Jubilee National Hospital (IRB number 1507), and this was a retrospective study, so informed consent was waived.
References
- Lewsey JD, Lawson KD, Ford I, et al. A cardiovascular disease policy model that predicts life expectancy taking into account socioeconomic deprivation. Heart 2015;101:201-8. [Crossref] [PubMed]
- McCartney G, Walsh D, Whyte B, et al. Has Scotland always been the ‘sick man’ of Europe? An observational study from 1855 to 2006. Eur J Public Health 2012;22:756-60. [Crossref] [PubMed]
- Hanlon P, Lawder RS, Buchanan D, et al. Why is mortality higher in Scotland than in England and Wales? Decreasing influence of socioeconomic deprivation between 1981 and 2001 supports the existence of a 'Scottish Effect'. J Public Health (Oxf) 2005;27:199-204. [Crossref] [PubMed]
- GBD 2015 DALYs and HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016;388:1603-58. [Crossref] [PubMed]
- Hagen TP, Häkkinen U, Belicza E, et al. Acute Myocardial Infarction, Use of Percutaneous Coronary Intervention, and Mortality: A Comparative Effectiveness Analysis Covering Seven European Countries. Health Economics 2015;24:88-101. [Crossref] [PubMed]
- Babaev A, Frederick PD, Pasta DJ, et al. Trends in management and outcomes of patients with acute myocardial infarction complicated by cardiogenic shock. JAMA 2005;294:448-54. [Crossref] [PubMed]
- Goldberg RJ, Spencer FA, Gore JM, et al. Thirty-year trends (1975 to 2005) in the magnitude of, management of, and hospital death rates associated with cardiogenic shock in patients with acute myocardial infarction: a population-based perspective. Circulation 2009;119:1211-9. [Crossref] [PubMed]
- Aissaoui N, Puymirat E, Tabone X, et al. Improved outcome of cardiogenic shock at the acute stage of myocardial infarction: a report from the USIK 1995, USIC 2000, and FAST-MI French Nationwide Registries. Eur Heart J 2012;33:2535-43. [Crossref] [PubMed]
- Reynolds HR, Hochman JS. Cardiogenic shock: current concepts and improving outcomes. Circulation 2008;117:686-97. [Crossref] [PubMed]
- Thiele H, Zeymer U, Neumann F-J, et al. Intraaortic Balloon Support for Myocardial Infarction with Cardiogenic Shock. N Engl J Med 2012;367:1287-96. [Crossref] [PubMed]
- Muller G, Flecher E, Lebreton G, et al. The ENCOURAGE mortality risk score and analysis of long-term outcomes after VA-ECMO for acute myocardial infarction with cardiogenic shock. Intensive Care Med 2016;42:370-8. [Crossref] [PubMed]
- Truby L, Naka Y, Kalesan B, et al. Important role of mechanical circulatory support in acute myocardial infarction complicated by cardiogenic shock. Eur J Cardiothorac Surg 2015;48:322-8. [Crossref] [PubMed]
- Tang GH, Malekan R, Kai M, et al. Peripheral venoarterial extracorporeal membrane oxygenation improves survival in myocardial infarction with cardiogenic shock. J Thorac Cardiovasc Surg 2013;145:e32-3. [Crossref] [PubMed]
- Kar B, Gregoric ID, Basra SS, et al. The percutaneous ventricular assist device in severe refractory cardiogenic shock. J Am Coll Cardiol 2011;57:688-96. [Crossref] [PubMed]
- Hendry PJ, Masters RG, Mussivand TV, et al. Circulatory support for cardiogenic shock due to acute myocardial infarction: a Canadian experience. Can J Cardiol 1999;15:1090-4. [PubMed]
- Zeymer U, Vogt A, Zahn R, et al. Predictors of in-hospital mortality in 1333 patients with acute myocardial infarction complicated by cardiogenic shock treated with primary percutaneous coronary intervention (PCI); Results of the primary PCI registry of the Arbeitsgemeinschaft Leitende Kardiologische Krankenhausärzte (ALKK). Eur Heart J 2004;25:322-8. [Crossref] [PubMed]
- Sleeper LA, Reynolds HR, White HD, et al. A severity scoring system for risk assessment of patients with cardiogenic shock: a report from the SHOCK Trial and Registry. Am Heart J 2010;160:443-50. [Crossref] [PubMed]
- Klein LW, Shaw RE, Krone RJ, et al. Mortality after emergent percutaneous coronary intervention in cardiogenic shock secondary to acute myocardial infarction and usefulness of a mortality prediction model. Am J Cardiol 2005;96:35-41. [Crossref] [PubMed]
- Abrams D, Combes A, Brodie D. Extracorporeal membrane oxygenation in cardiopulmonary disease in adults. J Am Coll Cardiol 2014;63:2769-78. [Crossref] [PubMed]
- Combes A, Leprince P, Luyt CE, et al. Outcomes and long-term quality-of-life of patients supported by extracorporeal membrane oxygenation for refractory cardiogenic shock. Crit Care Med 2008;36:1404-11. [Crossref] [PubMed]
- Brechot N, Luyt CE, Schmidt M, et al. Venoarterial extracorporeal membrane oxygenation support for refractory cardiovascular dysfunction during severe bacterial septic shock. Crit Care Med 2013;41:1616-26. [Crossref] [PubMed]
- Pagani FD, Lynch W, Swaniker F, et al. Extracorporeal life support to left ventricular assist device bridge to heart transplant: A strategy to optimize survival and resource utilization. Circulation 1999;100:II206-10. [Crossref] [PubMed]
- Pennington DG, Merjavy JP, Swartz MT, et al. The importance of biventricular failure in patients with postoperative cardiogenic shock. Ann Thorac Surg 1985;39:16-26. [Crossref] [PubMed]
- Petroni T, Harrois A, Amour J, et al. Intra-aortic balloon pump effects on macrocirculation and microcirculation in cardiogenic shock patients supported by venoarterial extracorporeal membrane oxygenation. Crit Care Med 2014;42:2075-82. [Crossref] [PubMed]
- Risnes I, Wagner K, Nome T, et al. Cerebral outcome in adult patients treated with extracorporeal membrane oxygenation. Ann Thorac Surg 2006;81:1401-6. [Crossref] [PubMed]
- Backes D, van den Bergh WM, van Duijn AL, et al. Cerebrovascular complications of left ventricular assist devices. Eur J Cardiothorac Surg 2012;42:612-20. [Crossref] [PubMed]
- Thiele H, Allam B, Chatellier G, et al. Shock in acute myocardial infarction: the Cape Horn for trials? Eur Heart J 2010;31:1828-35. [Crossref] [PubMed]
- Kajimoto M, O'Kelly Priddy CM, Ledee DR, et al. Myocardial Reloading After Extracorporeal Membrane Oxygenation Alters Substrate Metabolism While Promoting Protein Synthesis. J Am Heart Assoc 2013;2:e000106. [Crossref] [PubMed]
- Meltzer EC, Ivascu NS, Stark M, et al. A Survey of Physicians' Attitudes toward Decision-Making Authority for Initiating and Withdrawing VA-ECMO: Results and Ethical Implications for Shared Decision Making. J Clin Ethics 2016;27:281-9. [PubMed]