Overview of paediatric heart-lung transplantation: a global perspective
In this issue of The Journal of Thoracic Disease (JTD) Jonathon Spahr and Shawn West from the Children’s Hospital of Pittsburgh and the University of Pittsburg School of Medicine have presented a perspective on paediatric heart-lung transplantation based on data from their own institution, the Organ Procurement and Transplant Network and supported with data from the International Society for Heart and Lung Transplantation (ISHLT). They have provided a detailed and succinct overview of progress to the current era including indications, contraindications, outcomes and potential future possibilities largely based on the US experience. This editorial aims to extend the review by elaborating on the global perspective of paediatric heart-lung transplantation, particularly in relation to changes in volume and indications over the past 30 years and the influence of organ allocation policies internationally.
The era of human heart-lung transplantation commenced with the first successful adult heart-lung transplant in March 1981 at Stanford University Medical Center (1). This was soon followed by the first successful paediatric heart-lung transplant at Stanford in a 15-year-old girl in 1986 (2). The introduction of cyclosporine in the early 1980s and favourable airway healing rates resulted in good outcomes and heart-lung transplantation rapidly became a popular option for both adult and paediatric patients with end-stage cardiopulmonary disease. Moreover donor organ access was devoid of the current issues related to volume and organ allocation policies. Heart-lung transplant numbers peaked in 1989 with 223 adult (3) and 61 paediatric transplants (4) performed internationally that year (Figure 1). Subsequently several changes resulted in a shift away from heart-lung transplantation. Technical issues with isolated lung transplantation were resolved (5) resulting in improved airway healing rates and better survival, and isolated lung transplantation became a viable treatment option for end-stage pulmonary disease in the late 1980s. It also became apparent that survival following combined heart-lung transplant is essentially identical to that for isolated lung transplant with outcome primarily determined by the lung allograft. Moreover, competition for the scarce resource of donor organs and the need for utilitarian distribution of organs also influenced the shift from heart-lung transplantation to isolated lung transplantation in those with structurally normal hearts with preserved function. Consequently isolated single or bilateral lung transplantation became a preferred option for several end-stage pulmonary diseases for which heart-lung transplantation had previously been employed. In children undergoing isolated lung transplantation survival is significantly better with double compared with single lung transplant (6) and consequently bilateral lung transplantation is preferred. Lung transplantation combined with concurrent intra-cardiac repair of congenital heart disease in paediatric patients with Eisenmenger-related end stage pulmonary hypertension has also become an option as experience with lung transplantation has evolved (7). This obviates the need for combined heart-lung transplant but is of course dependent on clinical judgement of the capacity for recovery of ventricular systolic function following lung transplantation and cardiac repair. Finally, medical therapies for pulmonary hypertension have dramatically improved and diversified over the last 30 years thereby delaying or avoiding the need for lung or heart-lung transplantation. In children with end-stage idiopathic pulmonary hypertension and preserved right ventricular function creation of a non-restrictive Potts shunt may also avoid or delay the need for lung or heart-lung transplantation (8).
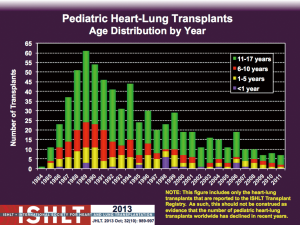
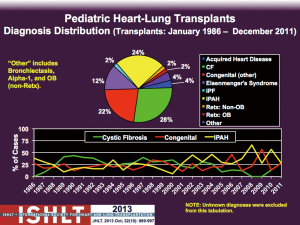
From a global perspective the most common underlying diagnoses in paediatric heart-lung transplant candidates over the past 30 years have been cystic fibrosis (28%), idiopathic pulmonary arterial hypertension (IPAH, 24%) and congenital heart disease (CHD, 22%) (4) (Figure 2). In the early era of heart-lung transplantation the most common diagnosis for paediatric candidates internationally was cystic fibrosis accounting for 40% of heart-lung transplants whereas CHD (15-25%) and IPAH (16%) were less substantial contributors (4) (Figure 2). The explanted hearts from those patients with structurally normal hearts and preserved ventricular function could then be used for domino heart transplantation with the advantages of a controlled ex vivo organ ischemic time and a donor right ventricle primed to work against elevated pulmonary vascular resistance (9). Airway healing is optimized in patients who undergo heart-lung transplantation as there is preserved bronchial blood supply provided by coronary-bronchial collaterals (1,10) that are not present when the lungs are harvested in isolation. However, increasing technical success with isolated lung transplantation led to this becoming the preferred option for patients with isolated lung disease and domino heart transplants are now rarely performed. Consequently from 1995 onwards the number of paediatric heart-lung transplants performed worldwide decreased substantially from previous highs of 31-61 transplants/year to about 20/yr on average until 2002 when numbers further declined to only 10-15 transplants/year (4) (Figure 1). Throughout this time the indications for paediatric heart-lung transplantation have also shifted with cystic fibrosis now only accounting for 0-15% of paediatric heart-lung transplants worldwide whereas IPAH accounts for 27-67% and congenital heart disease for 13-57% (4) (Figure 2). However, there are important geographic differences in the relative proportions of underlying diagnoses that likely relate to differences in organ allocation policies and organ availability throughout the world. IPAH and CHD each account for about 40% of paediatric heart-lung transplants in the USA currently whereas in Europe cystic fibrosis, IPAH and CHD are each responsible for about 30% of transplants (Figure 3).
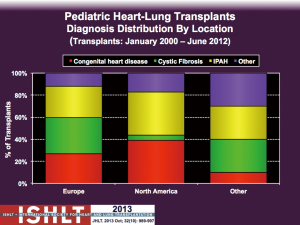
Intricacies of organ allocation policies in the USA (11) substantially influence the ability to achieve heart-lung transplantation in both adults and children. Heart-lung candidates must be listed on both heart and lung transplant wait lists as multi-organ candidates. Importantly individuals awaiting heart-lung transplant compete with UNOS status 1A heart-only candidates for the heart component of the organ block. It is rare that a candidate awaiting heart-lung transplant will fulfil criteria for status 1A heart listing thereby significantly limiting access to organs. This can be addressed by applying for an exemption. For paediatric candidates aged 12 years and older a lung allocation score (LAS) is required to define urgency for the lung component of the heart-lung block. Similar to heart urgency these candidates often do not fulfil criteria to achieve a high LAS and therefore may be disadvantaged. In contrast, the Eurotransplant policy (12) prioritizes heart-lung candidates above heart-only candidates with the same level of urgency in most European countries thereby optimizing access for the multi-organ candidates. Importantly, allocation policies in both Europe and the USA prioritize paediatric organs (donor <18 years of age) to paediatric recipients.
Infant heart-lung transplantation is essentially only performed in centres in the USA (Figure 4), a scenario that parallels the practice of infant lung transplantation. ISHLT data suggests that outcome following infant heart-lung transplantation is very poor with essentially no survivors beyond one year (13). However, under-reporting and incomplete reporting of outcomes likely influences this data. Moreover, it is not consistent with prior reports of acceptable early and medium term survival following infant heart-lung transplantation (14,15). Median graft survival following infant isolated lung transplantation in the USA is 4 years and is equivalent to other paediatric age groups (16). Median graft survival conditional on one-year survival is excellent at 7.4 years for infant lung transplantation (16). It is likely that survival following infant heart-lung transplant in the current era should be similar to that following infant lung transplant as heart-lung transplant outcome is defined by lung allograft survival. However, no infant heart-lung transplants have been reported to ISHLT since 2007 (4), likely for many of the general reasons mentioned previously, and most were performed in the decade from 1998-2007. The ability to repair or palliate complex CHD involving pulmonary vascular abnormalities has improved substantially over time thereby avoiding the need to perform heart-lung transplantation as primary intervention for CHD in infants. Finally, some patients may be amenable to intra-cardiac repair combined with infant lung transplantation given the difficulty in accessing a donor heart-lung block in the USA.
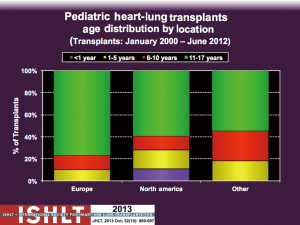
There are essentially two primary clinical indications for paediatric heart-lung transplantation in individuals with end-stage cardiopulmonary disease being limited life expectancy and poor quality of life. Those individuals with an estimated life expectancy of 1-2 years based on objective data such as FEV1, VO2(max), 6-minute-walk distance and supra-systemic pulmonary artery pressures should be considered for listing. The subgroup of infants is unique in that many of these candidates are dependent on invasive ventilation, inotropes and other supportive therapies, and therefore have a constant threat to life. Secondly, candidates with a poor quality of life as indicated by markedly limited physical activity, inability to attend school, inability to perform normal activities such as walking and playing with friends, dependency on inhaled oxygen therapy or non-invasive ventilation and frequent hospital admissions should also be considered for transplantation. Of course the standard requirements for transplant candidacy such as demonstrated compliance, good carer support and the absence of clinical contra-indications also need to be verified. In particular, adequate parental support, engagement and compliance is essential to a successful outcome following paediatric thoracic transplantation and its importance cannot be over-emphasized.
There are few absolute contraindications to heart-lung transplantation outside of the standard issues such as active or recent malignancy, active high-risk infection or multi-system organ failure. However many centres would be reluctant to offer heart-lung transplant to a patient dependent on veno-arterial extra-corporeal membrane oxygenation (VA-ECMO). This is particularly true for infants supported on VA-ECMO who have a very high pre-transplant mortality (14). This philosophy is changing somewhat in paediatric patients (17) as experience with lung and heart-lung transplantation in adults supported with awake, extubated VA-ECMO increases in high volume centres (18,19). It is important to emphasize that the need for VA-ECMO indicates dual organ failure and carries higher risk of complications and mortality than veno-venous ECMO for isolated respiratory failure with preserved cardiac function. Heart-lung transplantation may be utilized in situations where isolated lung transplant is not an option such as congenital pulmonary vein stenosis previously managed by sutureless repair or other techniques that prevent safe and adequate explantation of the native lungs with preservation of the phrenic nerves. Absence of native intra- and extra-pericardial pulmonary arteries is a contra-indication to isolated heart transplantation that has been addressed by using heart-lung transplantation in patients with lesions such as pulmonary atresia, ventricular septal defect and major aorto-pulmonary collaterals (PA/VSD/MAPCAs). Unfortunately, early outcomes in this patient population are poor due to a high risk of intra-operative exsanguination from extensive systemic to pulmonary collateral vessels, particularly in the setting of previous thoracotomies and sternotomies for palliative or corrective procedures. Consequently many experienced centres consider PA/VSD/MAPCAs to be an absolute contraindication to heart-lung transplantation (20).
It is important to clarify that heart-lung transplantation, like many solid organ transplants, is not a cure but is instead a form of palliation. This is particularly important to emphasize for anyone receiving a lung allograft as median survival following paediatric lung or heart-lung transplant still remains poor at approximately 5 years (6). Despite extensive investigation we remain limited in our ability to prevent or treat the inevitable development of bronchiolitis obliterans syndrome and chronic lung allograft dysfunction. Nevertheless combined heart-lung transplant continues to be a valuable therapeutic option for those with a poor quality of life or at high risk of dying as long as the recipient and their family accept its limitations.
Acknowledgements
Disclosure: The author declares no conflict of interest.
References
- Jamieson SW. Combined heart and lung transplantation. West J med 1985;143:829-33. [PubMed]
- Deuse T, Sista R, Weill D, et al. Review of heart-lung transplantation at Stanford. Ann thorac Surg 2010;90:329-37. [PubMed]
- Yusen RD, Christie JD, Edwards LB, et al. The Registry of the International Society for Heart and Lung Transplantation: Thirtieth Adult Lung and Heart-Lung Transplant Report--2013; focus theme: age. J Heart Lung Transplant 2013;32:965-78. [PubMed]
- Benden C, Edwards LB, Kucheryavaya AY, et al. The Registry of the International Society for Heart and Lung Transplantation: fifteenth pediatric lung and heart-lung transplantation report--2012. J Heart Lung Transplant 2012;31:1087-95. [PubMed]
- Cooper JD. The evolution of techniques and indications for lung transplantation. Ann Surg 1990;212:249-55. [PubMed]
- Benden C, Edwards LB, Kucheryavaya AY, et al. The Registry of the International Society for Heart and Lung Transplantation: Sixteenth Official Pediatric Lung and Heart-Lung Transplantation Report--2013; focus theme: age. J Heart Lung Transplant 2013;32:989-97. [PubMed]
- Choong CK, Sweet SC, Guthrie TJ, et al. Repair of congenital heart lesions combined with lung transplantation for the treatment of severe pulmonary hypertension: a 13-year experience. J Thorac Cardiovasc Surg 2005;129:661-9. [PubMed]
- Baruteau AE, Serraf A, Lévy M, et al. Potts shunt in children with idiopathic pulmonary arterial hypertension: long-term results. Ann Thorac Surg 2012;94:817-24. [PubMed]
- Klepetko W, Wollenek G, Laczkovics A, et al. Domino transplantation of heart-lung and heart: an approach to overcome the scarcity of donor organs. J Heart Lung Transplant 1991;10:129-31. [PubMed]
- Singh SP, Nath H, McGiffin D, et al. Coronary tracheal collaterals after heart-lung transplant. Am J Cardiol 2003;92:1490-2. [PubMed]
- Organ Procurement and Transplantation Network (OPTN) Policies. Policy 6: Allocation of Hearts and Heart-Lungs. 2014.
- Chapter 6: ET Thoracic Allocation System (EThAS). In: Eurotransplant manual version 3.3. 2014. Available online: https://www.eurotransplant.org/cms/index.php?page=et_manual
- Aurora P, Edwards LB, Christie JD, et al. Registry of the International Society for Heart and Lung Transplantation: Twelfth Official Pediatric Lung and Heart/Lung Transplantation Report-2009. J Heart Lung Transplant 2009;28:1023-30. [PubMed]
- Ro PS, Spray TL, Bridges ND. Outcome of infants listed for lung or heart/lung transplantation. J Heart Lung Transplant 1999;18:1232-7. [PubMed]
- Starnes VA, Oyer PE, Bernstein D, et al. Heart, heart-lung, and lung transplantation in the first year of life. Ann Thorac Surg 1992;53:306-10. [PubMed]
- Khan MS, Heinle JS, Samayoa AX, et al. Is lung transplantation survival better in infants? Analysis of over 80 infants. J Heart Lung Transplant 2013;32:44-9. [PubMed]
- Wong J, Buchholz H, Conradi A, et al. Successful Modified Semi-Ambulatory Veno-Arterial Extracorporeal Membrane Oxygenation (VA ECMO) Bridge To Heart-Lung Transplantation (HLTx) In A Four Year Old Child With Idiopathic Pulmonary Hypertension (IPAH). Am J Resp Crit Care 2012;185:A5824.
- Olsson KM, Simon A, Strueber M, et al. Extracorporeal membrane oxygenation in nonintubated patients as bridge to lung transplantation. Am J Transplant 2010;10:2173-8. [PubMed]
- Abrams DC, Brodie D, Rosenzweig EB, et al. Upper-body extracorporeal membrane oxygenation as a strategy in decompensated pulmonary arterial hypertension. Pulm Circ 2013;3:432-5. [PubMed]
- Goerler H, Simon A, Gohrbandt B, et al. Heart-lung and lung transplantation in grown-up congenital heart disease: long-term single centre experience. Eur J Cardiothorac Surg 2007;32:926-31. [PubMed]


