A rapid shallow breathing index threshold of 85 best predicts extubation success in chronic obstructive pulmonary disease patients with hypercapnic respiratory failure
Introduction
Chronic obstructive pulmonary disease (COPD) accounts for >3 million (6%) of all deaths globally (1), making it the fourth leading cause of death and the sixth leading cause of years lived with disability (2-4). Furthermore, its global burden is projected to increase in the coming decades due to continued risk factor exposure and population ageing (1,5).
COPD may be punctuated by periods of acute symptom worsening (exacerbations), with some requiring invasive mechanical ventilation (IMV) (1). Despite its advantages, ventilator-associated complications may include barotrauma, volutrauma, infections and others (6-8). Whereas it is widely agreed that expeditious liberation from IMV may limit unnecessary complications, the optimal method or technique to accomplish this remains debatable (4). For COPD patients, more than half of the time spent on the ventilator may be during the discontinuation process (4). Failed extubation may be associated with increased mortality, prolonged IMV, longer intensive care unit (ICU) and hospital length-of-stay (LOS), and higher rates of transfer to long-term-care facilities (9).
Several indices and predictors for extubation success have been reported in the literature (10-13), with the rapid shallow breathing index (RSBI) being among the most used clinically (12,14-16). The RSBI (breaths/min/L) is calculated by dividing the respiratory rate (RR) (f) by the spontaneous tidal volume (VT) in Liters (f/VT). For patients who otherwise meet criteria for IMV liberation, a RSBI <105 breaths/min/L suggests an increased likelihood of successful extubation (12). In a meta-analysis, the pooled sensitivity and specificity was 84% and 44% respectively, with a pooled positive likelihood ratio (LR+) of 1.49 and a pooled negative likelihood ratio (LR−) of 0.39 (17).
The reliability of the RSBI has been called into question for COPD patients, as some COPD patients have been noted to fail extubation despite a favorable RSBI (<105 breaths/min/L) (18). This has prompted some to call for more sophisticated parameters, or illness specific discriminatory thresholds for COPD patients (14,15,19). Some studies have advocated that a lower RSBI threshold may perform more optimally (20-27).
Moreover, both the timing and technique of measurement may influence outcomes. In one trial of 64 mechanically ventilated COPD patients, the RSBI [measured early during the spontaneous breathing trial (SBT)] did not accurately predict the successful outcome of a T-piece trial (18). It has been reported that in COPD patients, the RSBI measured through spirometry at the end of the SBT may be a better predictor of weaning outcome than when measured through pressure support (PS) (28). In this study we aimed to identify the highest performing RSBI threshold value for predicting successful mechanical ventilation (MV) liberation in COPD patients with hypercapnic respiratory failure.
Methods
This was a prospective observational multicenter study of COPD patients admitted to the Medical ICUs of eight academic medical centers from Jan 1, 2013 to Feb 28, 2017. The study was approved by institutional review boards at Baqiyatallah University of the Medical Sciences (340/5/5904) and Shahid Beheshti University of the Medical Sciences (SBMU1/REC/1393/89). Each participating hospital accepted these IRBs. Consent was required and covered both study participation and publication of findings. Informed consent was required prior to cardiac arrest event and could be provided by the patient, legal guardian, or healthcare surrogate. The informed consent included permission to present and publish de-identified results. All data generated or analyzed during this study are included in this article.
COPD diagnosis was in accordance with the global initiative for chronic obstructive lung disease (GOLD) criteria (1); a post-bronchodilator forced expiratory volume in one second (FEV1) to forced vital capacity (FVC) ratio <0.70 in patients with appropriate symptoms and significant exposures to noxious stimuli (1). All patients were intubated for hypercapnic respiratory failure [partial pressure of carbon dioxide (PaCO2) ≥50 mmHg and pH ≤7.35] and met the American Thoracic Society/European Respiratory Society guidelines to participate in a weaning trial (29). Ventilator weaning was conducted according to the defined protocol. Pre-defined weaning criteria included: (I) patent upper airway; (II) ability to protect airway (defined by mental status and presence of adequate gag and cough reflexes); (III) ability to clear secretions; (IV) decreasing secretion burden requiring suction not more frequently than every 2 hours; (V) level of support [fraction of inspired oxygen (FiO2) <50%; positive end-expiratory pressure (PEEP) =5]; and (6) hemodynamic stability not requiring chemical (vasopressors, inotropes) or mechanical (e.g., intra-aortic balloon pump, extracorporeal life support) circulatory support.
The inclusion criteria were: (I) MV ≥24 hours and (II) no sedatives or low dose sedation (midazolam 1–2 mg/hour or fentanyl 25–100 mcg/hour). Patients were excluded if: (I) did not meet pre-defined weaning criteria, (II) Glasgow Coma Score (GCS) <10, (III) inadequate cough reflex, (IV) self-extubation, (V) neuromuscular disease, (VI) acute coronary syndrome, (VII) cardiac failure, (VIII) end-stage renal disease, or (IX) for hemodynamic instability or signs of systemic infection/reinfection during the weaning process.
All patients were ventilated using Dräger Evita® XL or Evita® 4 ventilators (Dräeger Medical, Inc., Germany). Weaning and extubation readiness was determined by the respiratory therapist (RT) using pre-defined criteria and the result of a T-piece SBT. Before the T-piece trial, medical therapy was optimized, and patients’ airway secretions were suctioned. All patients were initially placed on PS =0 cmH2O, PEEP =0 cmH2O, and FiO2 ≤0.4 for 3 minutes, and assessed for any signs of distress: RR >30 breaths/minute, arterial oxygen saturation (SaO2) <90%, heart rate (HR) >140 breaths/minute, a sustained increase or decrease of HR of >20%, systolic blood pressure (BP) >200 mmHg or <80 mmHg, agitation, anxiety, or diaphoresis without other identified cause. If the patients maintained adequate mentation, hemodynamic stability, and adequate oxygenation and cough reflex without signs of respiratory distress while on no/low-dose sedation, a 120 min T-piece SBT was initiated (30). The decision to return to MV was made by the RT and attending intensivist (both independent of research team and blinded to results) based on signs of poor tolerance: SaO2 < 90% with FiO2 > 0.4; PaCO2 > 55 mmHg or increased by ≥10 mmHg; arterial pH <7.33; RR >35 breaths/min or increased by 50% for ≥5 minutes; HR >140 beats/min or increased/decreased by >20%; mean arterial pressure (MAP) >130 or <70 mmHg; or the presence of agitation, diaphoresis, disorientation, or depressed mental status (31). Patients demonstrating one of these signs during the SBT were considered a failed wean and returned to ventilatory support (32). For those completing the 120-minute SBT, the RSBI was measured through the ventilator without PS; continuous positive airway pressure (CPAP) =0 cmH2O; PS =0 cmH2O) for one minute. Patients tolerating the 120 min SBT without signs of distress or hemodynamic instability were extubated (33). Successful weaning from MV was defined as complete respiratory autonomy for at least 48 hours (34).
Data collection
The data collection tool was a two-part checklist including demographic variables (age, sex, smoking history) and ventilation parameters (intubation hours, hospitalization hours, spontaneous RR and VT).
Statistical analysis
The sample size calculation was performed using STATA®14 (StataCorp LLC, College Station, USA). The sample size was based upon data from the pilot study where the sensitivity and specificity of RSBI to predict extubation success were 0.97 and 0.94 respectively, and the LR+ was 16.6. Assuming an Alpha of 0.05 and a power of 0.9, the necessary sample size for the unsuccessful and successful extubation groups was 44 and 43 respectively.
Statistical analyses were performed using Statistical Package for Social Sciences software (SPSS for Windows, version 22.0; SPSS Inc., Chicago, USA), med calculator 13.1.0.0 (MedCalc Software bvba, Seoul, Republic of Korea) and 2×2 tables. Data were summarized using mean ± standard deviation (SD) for quantitative variables and frequency (%) for qualitative variables. Demographic data including gender, smoking, COPD severity, use of home oxygen therapy, rates of treatment with non-invasive ventilation (NIV), admission brain natriuretic peptide (BNP), high sensitivity C-reactive protein (hsCRP), ventilator associated pneumonia, and post-hypercapnic alkalosis were compared with Chi-square test. Variables including age, intubation duration, hospital LOS, RR, VT, partial pressure of oxygen (PaO2)/FiO2 ratio, PaO2/partial oxygen pressure of mixed alveolar gas (PALVO2) ratio, movement index, FVC, FEV1, FEV1/FVC, and forced expiratory flow at 25–75% (FEF25–75) were compared using the student T-test. APACHE II scores were compared using the likelihood ration test. Duration of midazolam infusion was compared using the t-test for equality of means.
To assess the prognostic value of the RSBI, receiver operating characteristic (ROC) curves was drawn. For each ROC curve analysis, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), diagnostic accuracy, LR+, and LR−, probability for weaning success when test is positive and probability for weaning success when test is negative of the index were used to predict the weaning outcome in the data set. Hanley et al. method was used to calculate the area under curve (AUC) for index and then the methods developed by the same authors were used to compare the AUCs (35). Bayes’ theorem was used to compute the weaning outcome the probability for weaning success when test is positive and negative (post-test probability) in the data set to assess the performance of test in predicting success weaning (36). In all analyses, P<0.05 were considered significant.
Results
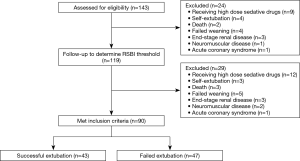
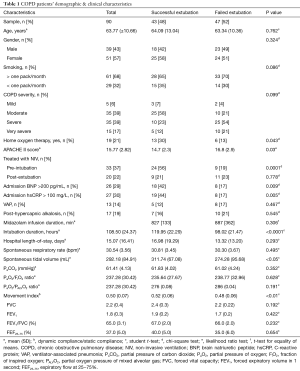
Out of 143 eligible patients, 90 met inclusion criteria. The patient flow diagram is shown in Figure 1. The data were normally distributed as determined by the one-sample Kolmogorov Simonov test (P>0.05). Table 1 details the demographic and clinical characteristics of all included patients. A slight female predominance was observed (57%) (Table 1). The mean (± SD) patient age was 63.77 (±10.66) years. Tobacco use, and COPD severity did not differ between groups. Slightly more than half of all patients (52.2%) failed extubation. Those successfully extubated on first attempt were more likely to be on home oxygen, treated with NIV pre-intubation, and to have an elevated C-reactive peptide (CRP) and BNP, Table 1. Patients failing extubation were more likely to have experienced ventilator-associated pneumonia (VAP). Neither the incidence of post-hypercapnic alkalosis nor duration of midazolam infusion differed between groups. The non-parametric comparison between patients extubated successfully versus those that failed extubation showed that only DoI, spontaneous VT (mL), and the ratio of dynamic-to-static compliance (movement index) showed significant differences (Table 1) (P<0.0001, P=0.03 and P=0.005, respectively), whereas RR, PaO2/FiO2 ratio, FVC, FEV1, FVC/FEV1, and FEF25–75 did not differ between groups (Table 1).
Full table
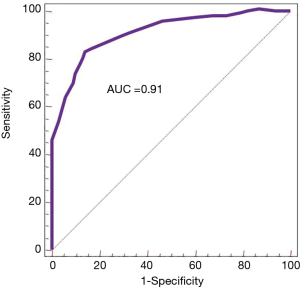
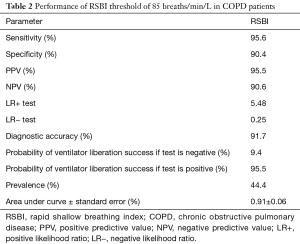
For RSBI, the area under the ROC curves was 0.91 (Figure 2). The point of optimal performance was ≤85 breaths/min/L, and performance characteristics are shown in Table 2. This threshold yielded a sensitivity (95.6%), specificity (90.4%), PPV (95.5%), NPV (90.6%), LR+ (5.48), LR− (0.25), and the diagnostic accuracy (91.7%). No differences were found between the DoI or hospital LOS on the performance of the RSBI ≤85 breaths/min/L.
Full table
Discussion
COPD patients requiring MV have a high mortality, with up to one-third dying despite MV support (37-41). It has been estimated that for COPD patients, more than half of MV time is spent during the discontinuation process (4). Successful ventilator liberation remains a critical component of patient care in the ICU, yet clinical judgment alone is insufficient to predict success (36,42,43).
Extubation success
Neuromuscular drive has been previously associated with successful ventilator liberation. This may be measured by airway occlusion pressure (P0.1), negative inspiratory force (NIF), maximal inspiratory pressure MIP, or the RSBI (34). The RSBI is among the most used clinical indices for predicting extubation success from a SBT (11,12,14-16,36,42,44), however its accuracy for identifying COPD patients likely to successfully extubate has been questioned as some COPD patients with a favorable RSBI (<105 breaths/min/L) have been noted to fail extubation (18). This has prompted some to call for either more sophisticated parameters or illness specific discriminatory thresholds for COPD patients (14,15). Our findings similarly call into question the appropriateness of the RSBI <105 breaths/min/L threshold for COPD patients. We found that the threshold of ≤85 breaths/min/L yielded the optimal sensitivity, specificity, PPV, NPV, LR+ and LR− for extubation success, with an AUC of 0.91. Elsewhere it has been reported that the percentage of successful weaning COPD patients rose from 34% to 60% by applying the RSBI threshold of <85 rather than <105 breaths/min/L (20). Similarly, a Taiwanese study reported the optimal RSBI threshold for COPD to be <79 breaths/min/L (21). Moreover, multiple non-randomized studies from China, Egypt, and Iran have further suggested that RSBI thresholds <85 breaths/min/L may better identify COPD patients likely to succeed with extubation (19-28). In another study of 152 COPD patients, El Khoury et al. reported that use of RSBI ≥70 (when the PaO2/FiO2 was ≥200) identified patients at a higher risk of reintubation (70% sensitivity, 56% specificity; AUC 0.69) (45). For those COPD patients that fail extubation despite a favorable RSBI, Purro et al. reported that most showed ineffective inspiratory efforts which artificially lowered the RSBI, or did not increase breathing frequency, but P0.1 and P0.1/VT/inspiratory time were as high as in other ventilator dependent patients (46).
Further validation is needed, however the COPD-specific RSBI threshold of ≤85 breaths/min/L may be a higher-performing tool for determining success of ventilator liberation in patients with COPD in hypercapnic respiratory failure.
Duration of MV
Many variables have been assessed to aid prognostication of outcomes in patients with acute exacerbations of COPD on MV, but few have withstood the test of time. Longer DoI has been reported with concurrent sepsis, VAP, advanced age, post-hypercapnic alkalosis and higher APACHE II score (47,48). Each of these was similar between groups in this study, except APACHE II score, which was slightly higher in the failed extubation group. That said, the APACHE II score has not been widely reported to correlate with weaning success or DoI.
The longer DoI observed in the successfully extubated group in is study was likely due to a confluence of factors including higher rates of initial high sensitivity hs-CRP >100 mg/L, BNP >200 pg/mL, and higher rates of pre-morbid home oxygen therapy in the successfully extubated group (49,50). Additionally, those in the successfully extubated group were more often treated with NIV prior to intubation. Whereas NIV may reduce intubation rates in COPD (51), patients saved from intubation were not included in this study. It remains unclear whether selecting out such patients significantly impacted outcomes in this study. Additionally, higher rates of chronic hypoxia may have lengthened the time needed to fulfill extubation requirements or tempered the aggression with which clinicians pushed such patients for ventilator liberation. Lastly, the longer hospital LOS in the successfully extubated group should be interpreted considering the system from whence the data arises. In Iran, systems for outpatient rehabilitation remain underdeveloped, thus many of those activities are undertaken in the inpatient setting and patients with significant outpatient needs may remain in the hospital longer.
RSBI technique
It remains unclear to what degree the technique of RSBI measurement impacts the accuracy of the measurement. For example, in the original work by Yang and Tobin, the RSBI was measured one-minute before the SBT with a hand-held spirometer attached to the end of the endotracheal tube (12). However, in common clinical practice the RSBI is calculated on PS without disconnecting the patient from the ventilator. Some have argued that this may influence results, whereas others have found no significant change (32,52). In one study, the AUC values (for successful liberation) of the RSBI measured through the ventilator on PS =5 cmH2O and CPAP =5 cmH2O was similar to measurements using the handheld spirometer (0.80 vs. 0.81) (43). However, another study reported that RSBI values measured on CPAP =0 cmH2O were significantly lower with a handheld spirometer (52). This difference was attributable to the base flow delivered by some ventilators. Some have concluded that RSBI measured through spirometry at the end of SBT is a better predictor of weaning outcome than RSBI measured under pressure support (28). Although we used the ventilator to measure the RSBI, we did so at the end of the SBT without pressure support like Youssef et al. in a way that most accurately reflects routine clinical practice.
RSBI validity
Literature has been controversial surrounding the utility of the SBT to predict successful ventilator liberation. In some studies, the SBT served as a high-performance predictor in weaning success (53,54), while in others it failed to account or accurately predict the consequences of tube removal in terms of upper airway patency, lower airway protection, secretion clearance, and continued stimulation of spontaneous respiration (55). Furthermore, some patients who tolerate a SBT under spontaneous or supported breathing scenarios (CPAP =5 cmH2O and PS =5 cmH2O) may develop respiratory dysfunction post-extubation that necessitates intervention (non-invasive or invasive ventilation) (43). Extubation failure rates in patients who passed an SBT have been reported at 16–20% (56,57). Thus, it should be noted that addition of the RSBI to the SBT, with all the listed challenges in the literatures, is a more precise tool than SBT alone.
The RSBI has also had its detractors. A meta-analysis conducted by the American College of Chest Physicians/American Association for Respiratory Care (AARC)/American College of Critical Care Medicine Task Force concluded that the RSBI has low accuracy as a weaning predictor (55). However, Tobin and Jubran raised significant criticism on these results. The authors indicated several methodological biases such as the inclusion of studies that were conducted in non-homogenous patient populations, and the evaluation of RSBI both as a predictor of the SBT or as a predictor of extubation (44).
Ineffective triggering (ITI) ≥10% or ITI <10% is a common problem early during MV and has been associated with increased morbidity, including longer MV duration, shorter ventilator-free survival, longer LOS, and lower likelihood of home discharge. One limitation of this study is that it was not designed to assess for rates of ITI (58). Additionally, we do not have echocardiogram data to accompany the BNP data to aid in risk stratification. Lastly, data on comorbid malignancy was not collected, and is thus not available to aid data interpretation.
Conclusions
The traditionally used RSBI threshold of <105 breath/min/L may not be appropriate for COPD patients on MV. In COPD patients intubated with hypercapnia, RSBI ≤85 breaths/min/L yields a 95.5% probability of ventilator liberation success, independent of ventilation duration or hospital LOS. This COPD-specific threshold improves the performance of the RSBI for predicting successful ventilator liberation.
Acknowledgements
The authors thank the Loghman Clinical Research Development Center, Loghman Hakim Hospital, and Shahid Beheshti University of Medical Sciences (Tehran, Iran) for methodological, logistical, and financial support. We also thank the Clinical Research Development Center of Baqiyatallah Hospital, Baqiyatallah University of Medical Sciences (Tehran, Iran) for its methodological support. These entities had no role in the collection, analysis, or interpretation of data and in writing the manuscript.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by institutional review boards at Baqiyatallah University of the Medical Sciences (340/5/5904) and Shahid Beheshti University of the Medical Sciences (SBMU1/REC/1393/89). Each participating hospital accepted these IRBs. Consent was required and covered both study participation and publication of findings. Informed consent was required prior to cardiac arrest event and could be provided by the patient, legal guardian, or healthcare surrogate. The informed consent included permission to present and publish de-identified results.
References
- Global Initiative for Chronic Obstructive Lung Disease. P Pocket Guide to COPD Diagnosis, Management and Prevention: A Guide for Healthcar. Scotts Valley: CreateSpace, 2017:37.
- Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012;380:2095-128. [Crossref] [PubMed]
- Kochanek K, Murphy S, Xu J, et al. Mortality in the United States, 2016. NCHS Data Brief, no 293. Hyattsville, MD: Centers for Disease Control and Prevention, 2017.
- Elgazzar AE, Walaa M, Salah A, et al. Evaluation of the minute ventilation recovery time as a predictor of weaning in mechanically ventilated COPD patients in respiratory failure. Egyp J Chest Dis Tuberc 2013;62:287-92. [Crossref]
- Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med 2006;3:e442. [Crossref] [PubMed]
- Koulenti D, Blot S, Dulhunty J, et al. COPD patients with ventilator-associated pneumonia: implications for management. Eur J Clin Microbiol Infect Dis 2015;34:2403-11. [Crossref] [PubMed]
- Ioannidis G, Lazaridis G, Baka S, et al. Barotrauma and pneumothorax. J Thorac Dis 2015;7:S38-43. [PubMed]
- Talwar D, Dogra V. Weaning from mechanical ventilation in chronic obstructive pulmonary disease: Keys to success. J Assoc Chest Physicians 2016;4:43-9. [Crossref]
- Blackwood B, Burns KE, Cardwell CR, et al. Protocolized versus non-protocolized weaning for reducing the duration of mechanical ventilation in critically ill adult patients. Cochrane Database Syst Rev 2014;6:CD006904. [PubMed]
- Boles JM, Bion J, Connors A, et al. Weaning from mechanical ventilation. Eur Respir J 2007;29:1033-56. [Crossref] [PubMed]
- Yang KL, Tobin MJ. A prospective study of indexes predicting the outcome of trials of weaning from mechanical ventilation. Surv Anesthesiol 1992;36:83-4. [Crossref]
- Yang KL, Tobin MJ. A prospective study of indexes predicting the outcome of trials of weaning from mechanical ventilation. New Engl J Med 1991;324:1445-50. [Crossref] [PubMed]
- Hajiesmaeili M, Goharimoghaddam K, Vahedian-Azimi A. New weaning indices in chronic obstructive pulmonary disease population. Egypt J Chest Dis Tuberc 2015;65:47-8. [Crossref]
- Chaiwat O, Sarima N, Niyompanitpattana K, et al. Protocol-directed vs. physician-directed weaning from ventilator in intra-abdominal surgical patients. J Med Assoc Thai 2010;93:930-6. [PubMed]
- Jacob B, Chatila W, Manthous CA. The unassisted respiratory rate/tidal volume ratio accurately predicts weaning outcome in postoperative patients. Critical Care Medicine 1997;25:253-7. [Crossref] [PubMed]
- Thiagarajan RR, Bratton SL, Martin LD, et al. Predictors of successful extubation in children. Am J Resp Crit Care Med 1999;160:1562-6. [Crossref] [PubMed]
- Meade M, Guyatt G, Cook D, et al. Predicting success in weaning from mechanical ventilation. Chest 2001;120:400S-24S. [Crossref] [PubMed]
- Boutou AK, Abatzidou F, Tryfon S, et al. Diagnostic accuracy of the rapid shallow breathing index to predict a successful spontaneous breathing trial outcome in mechanically ventilated patients with chronic obstructive pulmonary disease. Heart Lung 2011;40:105-10. [Crossref] [PubMed]
- Fadaii A, Amini SS, Bagheri B, et al. Assessment of rapid shallow breathing index as a predictor for weaning in respiratory care unit. Tanaffos 2012;11:28-31. [PubMed]
- Ghoneim AHA, El-Komy HMA, Gad DM, et al. Assessment of weaning failure in chronic obstructive pulmonary disease patients under mechanical ventilation in Zagazig University Hospitals. Egypt J Chest Dis Tuberc 2017;66:65-74. [Crossref]
- Lu YC, Tan C. Predictors of weaning failure in patients with chronic obstructive pulmonary disease. E-Da Med J 2018;5:11-5.
- Wu ZG, Li XB, Zhu JH, et al. Clinical value of shallow fast breathing index in predicting weaning of COPD patients in ICU. Xiuhong Clin Pulm J 2012;9:157-8.
- Luo B, Zhu XQ. The significance of shallow fast breathing index in weaning patients with chronic obstructive pulmonary disease. Jilin Med J 2009;30:2255-6.
- Lu ZH, Fang Q, Wang GB. The significance of shallow fast breathing index on weaning patients with COPD mechanical ventilation. Chinese J Crit Care Med 2005;3:12-3.
- Li YM, He GQ. Clinical study on long-term artificial ventilation and weaning index in patients with chronic obstructive pulmonary disease. Chinese J Tuberc Resp Dis 2000;4:217-20.
- Liu L, Wu WJ, Wang YX, et al. The value of superficial fast index combined with N-terminal pro-brain natriuretic peptide in guiding ventilator evacuation in patients with chronic obstructive pulmonary disease and respiratory failure. China Modern Med 2019;26:51-3.
- Guo Z, Zhou X, Shen C. Clinical analysis of long-term mechanical ventilation and withdrawal in patients with chronic obstructive pulmonary disease. Hebei Medical J 2003;25:326-7.
- Mahouri AR, Nourouzinia S, Farasatkish R, et al. Assessment of the rapid shallow breathing index as a predictor of weaning of patients with prolonged mechanical ventilation. Tanaffos 2007;6:30-5.
- Youssef HAA, Shalaby AEO, Abd El Hafiz AM, et al. Predictive value of rapid shallow breathing index in relation to the weaning outcome in ICU patients. Egypt J Chest Dis Tuberc 2016;65:465-72. [Crossref]
- Girard TD, Alhazzani W, Kress JP, et al. An Official American Thoracic Society/American College of Chest Physicians Clinical Practice Guideline: Liberation from Mechanical Ventilation in Critically Ill Adults. Rehabilitation Protocols, Ventilator Liberation Protocols, and Cuff Leak Tests. Am J Respir Crit Care Med 2017;195:120-33. [Crossref] [PubMed]
- Moosavinasab SM, Hosseini SM, Vahedi E, et al. Validation of combined index during weaning from a mechanical ventilator in chronic obstructive pulmonary patients: An observational prospective multi-center triple-blinded study in military hospitals in Iran. J Mil Med 2018;20:93-104.
- de Souza LC, Guimarães FS, Lugon JR. Evaluation of a new index of mechanical ventilation weaning: the timed inspiratory effort. J Intensive Care Med 2015;30:37-43. [Crossref] [PubMed]
- Souza LC, Lugon JR. The rapid shallow breathing index as a predictor of successful mechanical ventilation weaning: clinical utility when calculated from ventilator data. J Bras Pneumol 2015;41:530-35. [Crossref] [PubMed]
- Perren A, Domenighetti G, Mauri S, et al. Protocol-driven weaning from mechanical ventilation: clinical outcome in patients randomized for a 30-min or 120-min trial with pressure support ventilation. Intensive Care Med 2002;28:1058-63. [Crossref] [PubMed]
- Nava S, Rubini F, Zanotti E, et al. Survival and prediction of successful ventilator weaning in COPD patients requiring mechanical ventilation for more than 21 days. Eur Respir J 1994;7:1645-52. [Crossref] [PubMed]
- Hanley JA, McNeil BJ. A method of comparing the areas under receiver operating characteristic curves derived from the same cases. Radiology 1983;148:839-43. [Crossref] [PubMed]
- Nemer SN, Barbas CS, Caldeira JB, et al. A new integrative weaning index of discontinuation from mechanical ventilation. Crit Care 2009;13:R152. [Crossref] [PubMed]
- Bott J, Carroll MP, Conway JH, et al. Randomised controlled trial of nasal ventilation in acute ventilatory failure due to chronic obstructive airways disease. Lancet 1993;341:1555-7. [Crossref] [PubMed]
- Mador MJ, Sethi S. Systemic inflammation in predicting COPD exacerbations. JAMA 2013;309:2390-91. [Crossref] [PubMed]
- Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 1995;333:817-22. [Crossref] [PubMed]
- Foglio C, Vitacca M, Quadri A, et al. Acute exacerbations in severe COPD patients. Chest 1992;101:1533-38. [Crossref] [PubMed]
- Jeffrey AA, Warren PM, Flenley DC. Acute hypercapnic respiratory failure in patients with chronic obstructive lung disease: Risk factors and use of guidelines for management. Thorax 1992;47:34-40. [Crossref] [PubMed]
- Nemer SN, Barbas CS, Caldeira JB, et al. A new integrative weaning index of discontinuation from mechanical ventilation. Crit Care 2009;13:R152. [Crossref] [PubMed]
- Takaki S, Kadiman SB, Tahir SS, et al. Modified rapid shallow breathing index adjusted with anthropometric parameters increases predictive power for extubation failure compared with the unmodified index in postcardiac surgery patients. J Cardiothorac Vasc Anesth 2015;29:64-8. [Crossref] [PubMed]
- Tobin MJ, Jubran A. Meta-analysis under the spotlight: focused on a meta-analysis of ventilator weaning. Crit Care Med 2008;36:1-7. [Crossref] [PubMed]
- El Khoury MY, Panos RJ, Ying J, et al. Value of the PaO2:FiO2 ratio and Rapid Shallow Breathing Index in predicting successful extubation in hypoxemic respiratory failure. Heart Lung 2010;39:529-36. [Crossref] [PubMed]
- Purro A, Appendini L, De Gaetano A, et al. Physiologic determinants of ventilator dependence in long-term mechanically ventilated patients. Am J Resp Crit Care Med 2000;161:1115-23. [Crossref] [PubMed]
- Gursel G. Determinants of the length of mechanical ventilation in patients with COPD in the intensive care unit. Respiration 2005;72:61-7. [Crossref] [PubMed]
- Banga A, Khilnani GC. Post-hypercapnic alkalosis is associated with ventilator dependence and increased ICU stay. COPD 2009;6:437-40. [Crossref] [PubMed]
- Tofan F, Rahimi-Rad MH, Rasmi Y, et al. High sensitive C-reactive protein for prediction of adverse outcome in acute exacerbation of chronic obstructive pulmonary disease. Pneumologia 2012;61:160-2. [PubMed]
- Vallabhajosyula S, Haddad TM, Sundaragiri PR, et al. Role of B-Type Natriuretic Peptide in Predicting In-Hospital Outcomes in Acute Exacerbation of Chronic Obstructive Pulmonary Disease With Preserved Left Ventricular Function: A 5-Year Retrospective Analysis. J Intensive Care Med 2018;33:635-44. [Crossref] [PubMed]
- Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. New Engl J Med 1995;333:817-22. [Crossref] [PubMed]
- Desai NR, Myers L, Simeone F. Comparison of 3 different methods used to measure the rapid shallow breathing index. J Crit Care 2012;27:418.e1-6. [Crossref] [PubMed]
- Cappati KR, Tonella RM, Damascena AS, et al. Interobserver agreement rate of the spontaneous breathing trial. J Crit Care 2013;28:62-8. [Crossref] [PubMed]
- Su KC, Tsai CC, Chou KT, et al. Spontaneous breathing trial needs to be prolonged in critically ill and older patients requiring mechanical ventilation. J Crit Care 2012;27:324.e1-7. [Crossref] [PubMed]
- MacIntyre NR, Cook DJ, Ely J, et al. Evidence-based guidelines for weaning and discontinuing ventilatory support: A collective task force facilitated by the American College of Chest Physicians; the American Association for Respiratory Care; and the American College of Critical Care Medicine. Chest 2001;120:375S-95S. [Crossref] [PubMed]
- Maraghi SE, Hosny M, Samir M, et al. Usage of B-type natriuretic peptide for prediction of weaning outcome by spontaneous breathing trial. Egypt J Chest Dis Tuberc 2014;63:671-78. [Crossref]
- de Wit M, Miller KB, Green DA, et al. Ineffective triggering predicts increased duration of mechanical ventilation. Crit Care Med 2009;37:2740-5. [PubMed]