
Long-term outcomes of balloon-expandable bare stent as chimney stent in thoracic endovascular aortic repair for supra-aortic branches reconstruction
Introduction
Thoracic endovascular aortic repair (TEVAR) is characterized with less invasion and promising prognosis, and the published European Society for Vascular Surgery (ESVS) guideline has recommended TEVAR as the first-line option when the aortic repair is indicated (1). Despite emerging as a prevalent modality, TEVAR may be limited under some situations due to the inadequate proximal landing zone. Multiple solutions have been put forward to extend the proximal landing zone, including hybrid approach, fenestrated/branched endografts and chimney technique. Hybrid approach, in which aortic arch debranching is in conjunction with endovascular aortic repair, demonstrates high early- and long-term mortality (2,3). Fenestrated/branched endografts are mostly limited in elective settings since they must be customized with high cost and long waiting time (4,5). Chimney technique has become an availability at the emergent settings, in which a bare or covered stent is deployed in the supra-aortic branch with part paralleled with the endograft in the aorta.
Type Ia endoleak has been the major concern of chimney technique because of the gutter between the main endograft and chimney stent (CS) (6,7). Several factors about stents were considered related to the incidence of type Ia endoleak, such as the option of self-expandable stent or balloon-expandable stent (BES), covered or bare stent and so on. Generally, BES were used more often in supra-aortic branches reconstruction because of its greater radial strength (8-10). Furthermore, the correlation of covered or bare BES with endoleak has not been specifically discussed. Therefore, a cohort of patients who underwent TEVAR with supra-aortic branches reconstruction using balloon-expandable bare stent (BEBS) was followed up in terms of endoleak and other complications in this study. The present study will provide evidence to chimney technique using BEBS for supra-aortic branches reconstruction.
Methods
From February 2010 to April 2015, chimney technique was applied to extend the proximal landing zone in 40 patients, who received TEVAR for thoracic aortic dilation diseases in our center. Of them, 33 patients were implanted BEBS as CSs. Data including demographics, peri-operative complications and follow-up consequences were prospectively collected and retrospectively analyzed. This study has received the approval of the institutional research ethics committee review board. Written informed consents were obtained from all patients. Of them, planned chimney technique was carried out for 32 patients while 1 was applied as a bailout approach for the intraoperative inadvertent coverage of the left common carotid artery (LCCA). The diseases treated included 18 Stanford type B aortic dissections (TBAD), 3 type Ia endoleaks after initial TEVAR, 10 thoracic aortic aneurysms (TAA), and 2 pseudoaneurysms. The mean age was 61±14 (ranged from 34 to 86) years old, and the proportion of male was 87.9% (29/33). The comorbidities and other demographics were listed in Table 1.
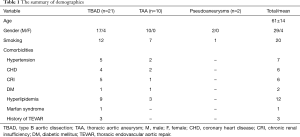
Full table
Indications for TEVAR with chimney technique included: the proximal landing zone was shorter than 1.5 cm; patients with high risk were not suitable for hybrid surgery or bypass surgery; intraoperative bailout for the unintentional coverage of the vital aortic arch branches.
All patients were treated with general anesthesia in a hybrid suite. Supra-aortic branches as well as the dominant vertebral artery were evaluated through preoperative enhanced computed tomography angiogram (CTA). After the systemic heparinization, the thoracic aorta endograft was delivered from the common femoral artery access. CSs implanted in the left subclavian artery (LSA) and innominate artery (IA) were introduced from the left and right brachial artery access with cut down exposing, respectively. Whereas the CS implanted in the LCCA was delivered from the LCCA access. After a 5 F pigtail catheter (Cordis Corporation, Miami Lakes, FL, USA) advanced through the right femoral artery sheath to the thoracic ascending aorta, the angiography was performed to determine the location of the proximal end of the aortic dilatation lesion and the orifice of the supra-arch branches. In addition, the cephalic artery angiography was also performed to confirm which side of the vertebra artery was the dominant one. LSA originating from the dominant vertebral artery was implanted with a CS. The main endograft was released firstly, then the BES was following deployed. CS was placed with the proximal end extending 5–10 mm over the aorta endograft, and with the distal end approximate 20 mm extending into the supra-arch branches. The completion angiogram was performed to confirm the position and shape of stents as well as immediate endoleaks.
All patients were followed up at 1, 3, 6 months and every 1 year thereafter. The CTA was performed to evaluate the endoleaks and the patency of CS during the follow-up session.
Results
There were 33 patients undergoing chimney technique in TEVAR to preserve the flow of supra-aortic branches. The technical success rate was 100%. All procedures were elective and one LCCA was reconstructed by CS due to the intraoperative unintentional coverage. Single LCCA in 22 cases and single LSA in 8 cases were reconstructed (Figure 1). LCCA and LSA were stented simultaneously for 1 patient while LCCA and IA were stented simultaneously for 2 patients. Thoracic aorta endografts used in our group included 14 of Zenith endografts (Cook Inc., Bloomington, Ind, USA), 10 of Valiant thoracic stent grafts (Medtronic Vascular, Santa Rosa, Calif, USA), 5 of Valiant Captivia stent grafts (Medtronic Vascular, Santa Rosa, Calif, USA), 3 of Ankura thoracic stent grafts (Lifetech, Shenzhen, China), and 1 of Relay thoracic stent graft (Bolton Medical, Sunrise, Fla, USA). CSs used in this group were all BEBSs, including 25 of Express stents (Boston Scientific Corporation, Natick, MA, USA), 9 of Scuba stents (Medtronic, Minneapolis, MN, USA), 1 of Genesis stent (Cordis, J&J Medical, Miami Lakes, Florida, USA), and 1 of Omnilink stent (Abbott Corp., Santa Clara, CA, USA).
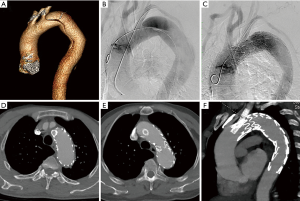
All CSs were expanded successfully by the balloon dilation with accurate position during the procedure. Fourteen immediate endoleaks (42.4%, 14/33) were found in the completion angiogram, including 12 (36.4%) type Ia endoleaks and 2 (6.1%) type II endoleaks. Two type II endoleaks appeared in two patients whose LCCAs were reconstructed, and the endoleaks both originated from LSA. One LSA was embolized by coils (Nester, Cook, Bloomington, USA) from the left brachial artery access, and the other LSA was excluded by a Plug (Lifetech, Shenzhen, China). For patients with type Ia endoleaks, 2 of them were managed by the balloon dilation and the endoleaks disappeared; the rest had been left with close surveillance. No other complications were found in the completion angiogram. One patient (3.0%) died 2 days after the operation owing to the acute rupture of aortic dissection.
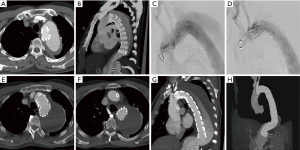
The mean follow-up time was 61.8 (ranged from 12 to 102) months, and the follow-up rate was 84.8% (28/33). The unmanaged 10 type Ia endoleaks were closely observed during the follow-up, of which 7 disappeared at 1 year and 1 disappeared at 2 years. The rest 2 patients with aortic dissections still had endoleaks but without further enlargement. During the follow-up, one patient was re-intervened for the increased false lumen due to the distal residual tears. This patient had LCCA reconstructed by chimney technique previously and another C-TAG stent graft (W.L. Gore and Assoc. Inc., Flagstaff, AZ, USA) was placed next to the first endograft, with the distal end landed above the celiac orifice (Figure 2). The long-term mortality was 9.1% (3/33). One patient, who had LCCA and LSA reconstructed simultaneously, died of cardiac infarction at 4 years. One died of renal failure at 6 years and the rest one died of unknown reason at 5 years. All CSs kept patent during the follow-up. One patient with sole LCCA reconstructed complained about coldness of the left upper limb without stent compression, and the symptom was relieved by medicine. No other complications, such as late endoleak, stroke, stent occlusion or migration, and so on, were found during the follow-up (Table 2).
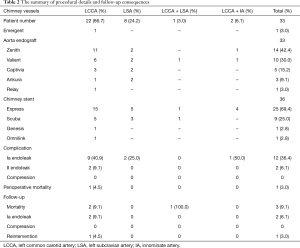
Full table
Discussion
Multiple approaches have been developed to extend the proximal landing zone for TEVAR. Chimney technique is effective to extend the proximal landing zone, especially in the emergent settings. This technique is also utilized in elective procedures increasingly because of its less demands on the devices and technology.
Compared with the hybrid surgery, the incidence of stroke is lower after the chimney (2.6% vs. 7.6%) (9). Besides, the calculated 30-day mortality also seemed a little bit lower for the chimney (7.9% vs. 11.9%) in the meta-analysis by Ahmad et al. and Moulakakis et al. (6,9). Fenestrated/branched stents are also developed to preserve the blood flow of supra-aortic branches with lower procedural mortality (2.4%), as well as low incidence of type I endoleak (11). However, this technique demands highly on the technology and requires custom-made stents, which are costly and often time consuming (12,13). Therefore, their applications have been restrained in emergent settings as well as occasions where the aorta is tortuous or the access is compromised. Chimney technique is initially developed for emergent or accidental situations, where a CS can be implanted in branch from the peripheral access. It has also been used in elective settings due to the easily achievable devices and comparable results. Nevertheless, type I endoleak is the major concern for CS due to the gutter, which is the channel between the CS and the aortic endograft and difficult to seal. Several factors of CSs have been considered related to the rate of endoleak.
Firstly, the self-expandable stent is more flexible and causes less vascular trauma, whereas the BES is characterized with more radial strength and facilitates the accurate placement. The endoleak and other complications about self-expandable and BES were nearly the same with 20% and 16% by the meta-analysis of Hogendoorn et al. (8). But this result required further investigation due to its insufficient number in each type of stents for reliable comparisons. In contrast, the systematic study of chimney technique in supra-aortic branches reconstruction in China by Zhao et al. demonstrated that the balloon-expandable bare CSs were associated with good early-term outcomes and a lower rate of endoleaks (14). In addition, the PERICLES registry, which compared balloon-expandable covered stent (BECS) with other non-BECS, showed improved survival of BECS group though with a higher risk of type Ia endoleak in endovascular repair of juxtarenal and pararenal aortic diseases. This study suggested the preference of BES for chimney (10). What’s more, the technical success of self-expandable stent might be lower than the BES due to its less radial force. As for the high blood flow within the aorta arch and pulsatile thoracic aorta, the self-expandable stents might be compressed by the endograft, which can be bailed out by BESs (15). Therefore, BES might be preferred for supra-arch branches chimney. Nevertheless, more studies are still needed to compare the effectiveness of these two kinds of stents for supra-arch branches chimney. All CSs used in this study were BEBSs for their greater radial force, to improve the technical success and long-term outcomes.
Secondly, another related factor concerned is whether the covered or bared BES is related to less endoleak. We have retrospectively analyzed a cohort of patients who all have BEBS to extend the proximal landing zone. The rate of immediate endoleak reached to 42.5% (14/33), including 12 type Ia endoleaks and 2 type II endoleaks. This rate has apparently overpassed that of the overall stents in the meta-analysis 9.4% by Ahmad et al., and 11% by Lindblad et al. (6,7). What’s more, a single-center study by Zhu et al. demonstrated a rate of immediate type I endoleak as 15% (5/34), and the bare chimney-stent repair had been used in all five cases. In this study, 25 (74%) balloon-expandable and 9 (26%) self-expandable stents were used, of which 7 (21%) were covered stents and 27 (79%) were bare stents (15). In another single-center study of double chimney technique for aortic arch diseases, the rate of immediate endoleak reached to 13% (3/23), all of which utilized were covered CSs (16). Based on our results and other studies, bare stents might be related to a higher rate of endoleak when compared with the rate of overall stents used (7,8,17,18). This might result from the extra blood flow through the bare stent mesh besides that in the gutter. However, great heterogeneity existed among these different studies and no one has intentionally compared the rate of endoleak between bare and covered BES. Thus, further investigations are needed to elucidate the correlation of bare or covered BES with the incidence of endoleak. On the other hand, few covered stents have been developed intended for CSs. As a result, the radial force, shape, length etc. of stents utilized now might not fulfill the best clinical requirement. The short and long-term patency of CS was promising in our study, so was the rate of patency reported in other studies (13,15,17,19,20).
In conclusion, when the chimney technique is chosen for supra-aortic branches reconstruction during TEVAR, the BES stent may be more suitable with long-term patency, while the BEBS may be related to a higher incidence of early endoleak.
Limitations
There are two main limitations in this study. Firstly, it is a retrospective study only including patients who received BEBSs as CSs. The outcomes of other covered stents as CSs therapy cannot be compared. Secondly, the number of patients enrolled was only thirty-four. More cases undergoing TEVAR combined with chimney technique using different kinds of chimney stents in the future are required to be followed up to compare the clinical outcomes. Therefore, the selection of stents for chimney technique can be based on the evidence.
Conclusions
The BES may be more suitable for supra-aortic branches reconstruction with long-term patency when the chimney technique is employed during TEVAR. However, BEBS may be related to a higher rate of early endoleak.
Acknowledgements
Funding: This work was supported by the National Natural Science Foundation of China (Grant number: 81570438).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the institutional research ethics committee review board and written informed consents were obtained from all patients.
References
- Schermerhorn ML, Jones DW. Management of Descending Thoracic Aorta Disease: Evolving Treatment Paradigms in the TEVAR Era. Eur J Vasc Endovasc Surg 2017;53:1-3. [Crossref] [PubMed]
- Riambau V. Application of the Bolton Relay Device for Thoracic Endografting In or Near the Aortic Arch. Aorta (Stamford) 2015;3:16-24. [Crossref] [PubMed]
- Zhao Y, Cui J, Yin H, et al. Hybrid operation versus the chimney technique for reconstruction of a single aortic arch branch: a single-centre experience. Interact Cardiovasc Thorac Surg 2017;25:575-81. [Crossref] [PubMed]
- Lu Q, Feng J, Zhou J, et al. Endovascular repair by customized branched stent-graft: A promising treatment for chronic aortic dissection involving the arch branches. J Thorac Cardiovasc Surg 2015;150:1631-8.e5. [Crossref] [PubMed]
- Caradu C, Berard X, Sassoust G, et al. Chimney versus fenestrated endovascular aortic repair for juxta-renal aneurysms. J Cardiovasc Surg (Torino) 2018;59:600-10. [PubMed]
- Ahmad W, Mylonas S, Majd P, et al. A current systematic evaluation and meta-analysis of chimney graft technology in aortic arch diseases. J Vasc Surg 2017;66:1602-10.e2. [Crossref] [PubMed]
- Lindblad B, Bin Jabr A, Holst J, et al. Chimney Grafts in Aortic Stent Grafting: Hazardous or Useful Technique? Systematic Review of Current Data. Eur J Vasc Endovasc Surg 2015;50:722-31. [Crossref] [PubMed]
- Hogendoorn W, Schlosser FJ, Moll FL, et al. Thoracic endovascular aortic repair with the chimney graft technique. J Vasc Surg 2013;58:502-11. [Crossref] [PubMed]
- Moulakakis KG, Mylonas SN, Markatis F, et al. A systematic review and meta-analysis of hybrid aortic arch replacement. Ann Cardiothorac Surg 2013;2:247-60. [PubMed]
- Scali ST, Beck AW, Torsello G, et al. Identification of optimal device combinations for the chimney endovascular aneurysm repair technique within the PERICLES registry. J Vasc Surg 2018;68:24-35. [Crossref] [PubMed]
- Katsargyris A, Oikonomou K, Klonaris C, et al. Comparison of outcomes with open, fenestrated, and chimney graft repair of juxtarenal aneurysms: are we ready for a paradigm shift? J Endovasc Ther 2013;20:159-69. [Crossref] [PubMed]
- XiaoHui M, Wei G, ZhongZhou H, et al. Endovascular repair with chimney technique for juxtarenal aortic aneurysm: a single center experience. Eur J Vasc Endovasc Surg 2015;49:271-6. [Crossref] [PubMed]
- Banno H, Cochennec F, Marzelle J, et al. Comparison of fenestrated endovascular aneurysm repair and chimney graft techniques for pararenal aortic aneurysm. J Vasc Surg 2014;60:31-9. [Crossref] [PubMed]
- Zhao Y, Shi Y, Wang M, et al. Chimney Technique in Supra-Aortic Branch Reconstruction in China: A Systematic and Critical Review of Chinese Published Experience. Vasc Endovascular Surg 2017;51:429-35. [Crossref] [PubMed]
- Zhu Y, Guo W, Liu X, et al. The single-centre experience of the supra-arch chimney technique in endovascular repair of type B aortic dissections. Eur J Vasc Endovasc Surg 2013;45:633-8. [Crossref] [PubMed]
- Wang T, Shu C, Li QM, et al. First experience with the double chimney technique in the treatment of aortic arch diseases. J Vasc Surg 2017;66:1018-27. [Crossref] [PubMed]
- Bin Jabr A, Lindblad B, Dias N, et al. Efficacy and durability of the chimney graft technique in urgent and complex thoracic endovascular aortic repair. J Vasc Surg 2015;61:886-94.e1. [Crossref] [PubMed]
- Pecoraro F, Lachat M, Cayne NS, et al. Mid-term Results of Chimney and Periscope Grafts in Supra-aortic Branches in High Risk Patients. Eur J Vasc Endovasc Surg 2017;54:295-302. [Crossref] [PubMed]
- Gehringhoff B, Torsello G, Pitoulias GA, et al. Use of chimney grafts in aortic arch pathologies involving the supra-aortic branches. J Endovasc Ther 2011;18:650-5. [Crossref] [PubMed]
- Wang T, Shu C, Li M, et al. Thoracic Endovascular Aortic Repair With Single/Double Chimney Technique for Aortic Arch Pathologies. J Endovasc Ther 2017;24:383-93. [Crossref] [PubMed]


