Descriptive review of patent ductus arteriosus ligation by video-assisted thoracoscopy in pediatric population: 7-year experience
Introduction
Patent ductus arteriosus (PDA) is relatively common and reported to constitute 10% of all congenital heart disease (1). It is frequently seen in females and more prevalent in preterm babies possibly due to developmental immaturity (2). Persistent PDA if remain untreated, may results in ventricular hypertrophy, heart failure, reversal of shunt, infective endocarditis, aneurysmal dilation, failure to thrive and even early death (3). The initial treatment is usually pharmacologic, the surgical option is considered when conservative treatments are contraindicated (necrotizing enterocolitis, renal injury, intraventricular hemorrhage) or treatment failure is observed (4,5).
Previous work indicates that open thoracotomy is safe and reliable procedure (6). Recent observational data also suggests that open procedures are associated with poor neurodevelopmental outcomes (7). Less invasive, percutaneous catheter-based closure using coil embolization or such devices seems to be a favorable option in the older children and infants >4 kg only, and is associated with higher residual shunt (8-10). Notably, arterial injury seems to be consistently reported in preterm cohort who underwent percutaneous procedures (11). Nevertheless, the type of clinical settings, morphology of ductus, and patient’s weight are important determinants of choosing the type procedure.
Over the past two decades, other less invasive procedures such as video-assisted thoracoscopy (VATS) have gained immense popularity. VATS-PDA ligation offers several advantages including preservation of muscular architecture of spine (12). Studies done on VATS-PDA ligation showed reduced surgical times, length of stay, better cosmetics when compared to open thoracotomies however complication rates remain conflicting among the centers (13-17). Learning curve may be a postulated reason (18). Nonetheless, limited data also precludes the acceptability of VATS procedure for PDA ligation among the centers. Very few studies evaluated the immediate clinical outcomes, complications and explored the long-term survival in patients undergoing PDA closure by VATS. We therefore sought to report our single centered 7-year experience of PDA closure with VATS.
Methods
With Institutional Review Board of Poznan University of Medical Sciences approval and waived consent we conducted this retrospective study between February 2012 and October 2018. Study data were collected and managed per protocol hosted at our institution. Patients’ demographics, age, race, gender, body mass index (BMI), surgical history, length of hospital stay, the presence of complications, and long-term outcomes were collected.
Patient selection
We included single centered patients who underwent VATS-PDA closure in whom conservative treatment for PDA either failed or was contraindicated were enrolled in the study. None of them was an appropriate candidate for percutaneous closure. VATS was introduced in our center in 2012, we believed that there was a learning curve initially, hence we divided our included patients in two groups, i.e., 2012–2014 and 2015–2018.
Transthoracic echocardiography was performed in all patients before the surgery to confirm the presence of hemodynamically significant PDA. The exclusion criteria for VATS procedure were: diameter of PDA >9 mm, previous thoracotomy, previous history of left thoracic surgery, preexisting cardiac anomalies requiring simultaneous surgical intervention, endocarditis, calcified or aneurysmal PDA. Such criteria were observed in 6 patients (4.7%) who underwent surgical ligation of PDA through a posterolateral thoracotomy.
Technique of VATS closure
All procedures were performed by the same surgical team that consisted of two cardiac surgeons. All infants were operated in an infant incubator. The patients were anesthetized applying single-lumen, endotracheal intubation with isolated right lung ventilation. Endotracheal tube was adjusted immediately after the procedure to the supracarinal position. Midazolam, fentanyl, atracurium or pancuronium were used for induction and sevoflurane for anesthesia maintenance. Standard monitoring during the surgery included electrocardiogram, non-invasive blood pressure, capnography, pulse oximetry, central venous pressure.
The surgery was performed in right lateral decubitus positioning, the initial 5-mm incision was made along the 4th or 5th intercostal space (ICS) in anterior axillary line. The first port was used for the video-camera (Karl Storz Endoskope GmbH, Germany) and carbon dioxide insufflation. The thorax cavity was insufflated with CO2 under pressure of 5–7 mmHg, to compress the left lung and improve the visualization of the surgical field. The second and third 3-mm incisions in the 3rd and 6th ICSs in the posterior axillary line were made to place the instruments and clip applicator. The anatomical structures such as PDA, left pulmonary artery, descending aorta, recurrent laryngeal and vagus nerve were identified. Thereafter, carefully opening of the mediastinal pleura overlying the PDA was done, and the duct was gently and slowly dissected free from the surrounding tissues. After obtaining optimal visualization and magnification, an appropriate size of titanium clips (5, 8 or 10 mm) were chosen and were placed across to occlude the arterial duct. One or two titanium clips were enough to completely close the PDA. After adequate hemostasis was achieved, the CO2 was aspirated from the thorax and the lung was re-expanded. The chest tubes were not routinely placed unless bleeding was expected or there was suspected lung injury. The chest tube, if necessary, was inserted through the first incision whereas the other holes for trocars were closed with single absorbable sutures.
Conversion to posterolateral thoracotomy, if necessary, was done through the anterior axillary line at 4th or 5th ICS. The mediastinal pleura was opened, PDA visualized and then dissected free from the surrounding tissues. The PDA was closed using titanium vascular clips. The chest tube was placed in all patients after conversion. Finally, the chest was closed in layers.
Early postoperative period and follow-up protocol
The postoperative echocardiography was routinely performed in the operating room to confirm the absence of ductal flow. Post-surgery, all patients were transferred to the pediatric cardiothoracic intensive care unit (P-CICU). Preterm infants requiring further treatment were treated in the neonatal ICU whereas the care was continued in pediatric cardiothoracic unit for other children. Chest X-ray was performed immediately after all surgeries to exclude hemothorax or pneumothorax and to confirm the proper position of the clip.
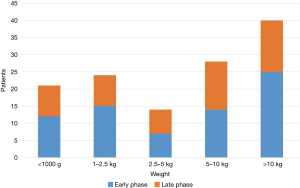
Outcomes were measured included not only mortality and morbidity but also the procedure time, need for intraoperative conversion, chest tubes insertion, length of hospital stay, blood transfusion, residual shunt and parents/patient’s satisfaction with their surgical scars. Learning curve was ascertained and aforementioned variables were compared between two equal-in-length periods: early phase—from 2012 to 2014 (n=73); and late phase—from 2015–2018 (n=54). Clinical characteristics were comparable between both groups. The patients were followed up after discharge either by family physicians in majority and few by phone interview.
Statistical analysis
Continuous data variables were checked for normality with Shapiro-Wilk test. Normally distributed data was expressed as the means ± standard deviations and further compared with unpaired student t-test. Otherwise, they are presented as median with the range (min to max) and differences were calculated by means of the Mann-Whitney U test. Survival rate was stratified with the use of Kaplan-Meier method. Statistical significance was assumed at P<0.05. Statistical analysis was computed with SPSS Statistics 24 (IBM, USA) software.
Ethics approval
This study was approved by the Bioethics Committee at Poznan University of Medical Sciences (No. KB 40/18).
Results
One hundred twenty-seven patients with hemodynamically significant PDA underwent clipping via VATS. Median age at the time of surgery was 1.7 years, ranged from 10 days to 15.8 years. Forty-six patients weighed <2.5 kg at the time of the procedure constitute almost 36% of total population, not a trivial fraction (Figure 1). Other preoperative demographics selected clinical data and overall complication rates are summarized in Table 1.

Full table
Operative data
Intraoperative mortality was none. The overall mean surgery time was 56.1±33.1 minutes and was found to be comparable in both periods [57.5±34.6 vs. 54.4±31.2 minutes (ns)], however the mean surgery time of the last 15 procedures was 38 minutes. Although, intraoperative conversion rate from VATS to thoracotomy was more frequent by at least 50% in the early phase 13/73 (17.8%) vs. 6/54 (11.1%) statistical analysis revealed no significant difference (ns). Of note, only one conversion was noted during the last 25 procedures (4.0%). The most prevalent reason for conversion was noted to be incomplete PDA closure (n=7) followed by post-clipping ductal bleeding (n=5), inadequate visualization (n=5) cardiopulmonary instability post-insufflation (n=2) and injury to pulmonary vein during preparation (n=1). Conversion was associated with significantly longer mean surgery time (100.1±47.1 vs. 47.3±20.5 minutes) and higher rate of blood transfusion (52.4% vs. 24.5%). Chest tube was placed in 27 patients (21.3%) and removed after 30.1±18.3 hours with median drainage of 27.4 mL.
In-hospital outcomes
Overall in-hospital mortality was 4.7% (n=6). Two preterm infants, weighting 700 and 2,000 g, died at the 40th and 48th hours after the surgery. The first infant underwent a complete PDA closure using VATS and died due to massive cerebral bleeding and the second one needed emergency conversion to thoracotomy and died due to cardiopulmonary failure. Four patients died during the treatment on the neonatal ICU at 3rd, 4th, 18th days after the procedure due to acute cardiopulmonary failure and at 20th day postoperatively due to cerebral bleeding.
Higher overall postoperative total complications rate was observed in the early phase (P=0.03, Table 2). Thoracotomy was performed in the early postoperative period in two children. One preterm infant weighing 890 g required open surgery 8 days post procedure due to lung injury and pneumothorax and another 7-year-old child 5-day post-procedure for surgical repair of chylothorax.

Full table
Seventy-five patients (59.1%) [median age: 2.8 years, median weight: 13.7 kg] were discharged home after mean in-hospital stay of 2.2±1.6 days. Fifty patients (39.4%) [median age: 32.6 days, median weight: 1.55 kg] were transferred to the neonatal ICU for continuity of care. Twenty-nine of them needed respiratory support with median time of mechanical ventilation 17.7 days. The mean length of stay at neonatology unit was 49.9±33.0 days.
Follow-up period
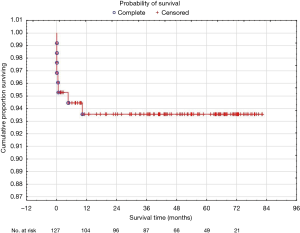
All patients completed the follow-up period for a median of (min to max) 4.0±1.9 years (1 month to 6.7 years). All but two patients discharged home survived follow-up period without any adverse events. Two children (preterm infants) who were discharged after 13- and 26-day treatment on neonatology unit died at home 139 and 310 days post-procedure, due to sudden infant death syndrome and cerebral disorders. A 5-year probability of survival after VATS closure of PDA stratified with the use of Kaplan-Meier method was 93.6% (Figure 2).
Residual ductal patency was detected in 2 children (1.6%) post procedure during the follow-up period. The pre-procedure diameter of the duct was 5 and 6 mm. In both cases, the residual shunts were noted to be hemodynamically insignificant not requiring any further treatment.
Among 106 patients (83.5%) who underwent successful VATS, 97.2% patients and parents were satisfied with the surgical scar. None of our patients had high-degree scoliosis or other musculoskeletal deformation requiring further treatment.
Preterm infants less than 1,000 g
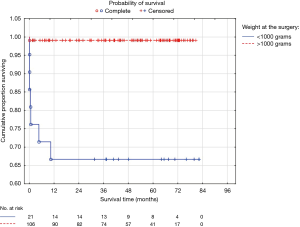
Patients weighing at the time of surgery less than 1,000 g make up to 16.5% of all operated children. These infants were born before the 29th gestational week (22–29 weeks) with mean birth weight 702.9±128.2 g and presented significantly more symptoms of heart failure, respiratory failure, renal failure and intraventricular hemorrhage (47.6% vs. 18.8%; 100% vs. 28.3%; 9.5% vs. 3.8%; 52.4% vs. 16.0%) than patients weighing over 1,000 g. Patients weight did not affect the surgery related complications or duration of the procedure, however, the total complication rate was significantly higher in the smaller children <1,000 g (66.7% vs. 27.4%). The conversion rate was regardless of weight and remains at 14.3% in children weighing less than 1,000 g. One patient died at P-CICU (4.8%) and other 3 at neonatology unit. Overall mortality in this group of patients was 28.6% (n=6). A 5-year probability of survival estimated according to the Kaplan-Meier curve was 66.7% and it was significantly lower than calculated probability among children weighting over 1,000 g at the time of surgery (Figure 3).
Discussion
Overall during our study, the mortality rate was 6.3%. All who died were preterm infants. None death was related directly to the procedure and we demonstrated no intraoperative mortality. Postoperative morality of PDA ligation through posterolateral thoracotomy in the literature is described as minimal in older children and can reach up to 21% mortality among preterm infants (13,19-21). Several investigations that compared VATS with conventional thoracotomy revealed that both techniques were equal to one another in terms of their efficiency and safety (13,14,22).
In the present cohort of wide range of pediatric age and body weight, VATS-PDA ligation was carried out successfully and safely. The noted post-operative deaths (6.3%) seems relatively high, however mostly ostensible to premature consequences than surgical complications per se (23). Further analysis of these deaths revealed that those patients already had some degree of heart failure, respiratory failure, bronchopulmonary dysplasia, infection, anemia of prematurity or renal failure preoperatively. Additionally, our cohort represented 38.6% of premature infants—not a trivial fraction. We also observed extended mechanical ventilation and length of hospital stay possibly due to aforementioned reasons. The incidence of PDA is more common in females, our study also depicted more incidence in females (62%). The most difficult group of patients are preterm infants less than 1,000 g. This group presented both the highest overall complication rate and mortality with survival rate after 5 years only 66.7%. More attention must undoubtedly be focused on transport to operating room. All our patients were operated in the operating room, which is located next to the P-CICU. All Infants were operated in an infant incubator to reduce the potential risks of transportation such as hypothermia, dislocation of the tracheal tube or vascular line. Some investigations describe even surgery on bedside, to reduce the risk of transport of the smallest babies (24).
The rate of conversion from VATS to posterolateral thoracotomy in our study was 16.5%, however, 90% of these conversions were performed intra procedure whereas deemed necessary in 10% during the initial post-procedure days. Our conversion rate was relatively high from the previously observed rates of 0.3–12% in the literature (22). It is noteworthy that the intraprocedural conversion rate during the initial 2 years of early phase was 20.4%, which decreased significantly to less than 5% in the last 2 years of late phase. The reduction of conversion rate in the late phase of our study suggests the effect of learning curve—a known phenomenon in VATS procedures (25). This effect was also translated to reduction of the total postoperative complications (42.5% vs. 22.2%, P=0.03) in the late phase. All procedures were always performed by two same surgeons what enabled the standardization and the improvement of the technical skills that resulted in significant reduction of surgery time to 38 minutes in last 15 cases. We strongly believe that gaining surgical experience allowed us to avoid many technical and medical pitfalls. Lower prevalence of pulmonary complications in the late phase could be related to the meticulous PDA preparation and less compression of the lung. Nonetheless, this learning curve was observed without any effect on mortality.
Despite routine post procedural echocardiography, two of our patients had hemodynamically insignificant residual shunts during the initial post-operative days. The residual shunt after surgical closure are mostly rare and hemodynamically insignificant. However, none of them required any further treatment. Previous two large numbered studies including 700 and 2,000 patients after thoracoscopic clipping of PDA reported late residual shunt rate of 0.6% and 0.2% (15,16). Nevertheless, investigators reported nil redo-procedure rate for residual shunts (26,27).
The incidence of post thoracotomy scoliosis is predominantly found in premature infants. We did not find any post-thoracotomy scoliosis or non-healing surgical scars in our group during follow ups, even in patients who underwent intra- or post-operative conversion to thoracotomy. Open thoracotomy involves significant muscle division, rupture of intercostal ligaments and rib spreading which may lead to imbalance of forces in muscular architecture and further attributes to scoliosis (28,29). Lawal et al. (12) also observed reduction of chest asymmetry, scoliosis and relatively better cosmetics results with VATS when compared to open procedures. Similar observations were also made by Chen et al. (13).
Our study is unique in its attempt to assess both short- and long-term complications of VATS in a large numbered cohort which includes almost 40% of preterm population. We also recognized the implication of learning curve in VATS-PDA ligation which may be responsible for increased complication in the early phase and may precludes its widespread usage. Our study is limited in being a retrospective analysis and therefore suffers some degree of confounding and bias. This investigation was a single-center and the results were analysed objectively without any control group hence the results may not be generalizable. Regardless, our results alienate with previous work. High early phase complications attributed to learning curve may falsely increase the total complication rate of the study but in general, reflects the trend of any new technique when introduced.
Conclusions
Overall, our results imply that VATS is a safe as well as efficient method for PDA ligation that ensures satisfactory late cosmetic results. Postoperative mortality and extended hospital stay may be attributed to prematurity. Although learning curve exists it does not affect the safety and late outcomes.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by the Bioethics Committee at Poznan University of Medical Sciences (No. KB 40/18).
References
- Brickner ME, Hillis LD, Lange RA. Congenital heart disease in adults. First of two parts. N Engl J Med 2000;342:256-63. [Crossref] [PubMed]
- Schneider DJ, Moore JW. Patent ductus arteriosus. Circulation 2006;114:1873-82. [Crossref] [PubMed]
- Panagopoulos PG, Tatooles CJ, Aberdeen E. Patent ductus arteriosus in infants and children. A review of 936 operations (1946-69). Thorax 1971;26:137-44. [Crossref] [PubMed]
- Calder AL, Kirker JA, Neutze JM, et al. Pathology of the ductus arteriosus treated with prostaglandins: comparisons with untreated cases. Pediatr Cardiol 1984;5:85-92. [Crossref] [PubMed]
- Evans N. Current controversies in the diagnosis and treatment of patent ductus arteriosus in preterm infants. Adv Neonatal Care 2003;3:168-77. [Crossref] [PubMed]
- Mavroudis C, Backer CL, Gevitz M. Forty-six years of patient ductus arteriosus division at Children's Memorial Hospital of Chicago. Standards for comparison. Ann Surg 1994;220:402-9. [Crossref] [PubMed]
- Janz-Robinson EM, Badawi N, Walker K. Neurodevelopmental Outcomes of Premature Infants Treated for Patent Ductus Arteriosus: A Population-Based Cohort Study. J Pediatr 2015;167:1025-32.e3. [Crossref] [PubMed]
- Dimas VV, Takao C, Ing FF, et al. Outcomes of transcatheter occlusion of patent ductus arteriosus in infants weighing ≤ 6 kg. JACC Cardiovasc Interv 2010;3:1295-9. [Crossref] [PubMed]
- Wang K, Pan X, Tang Q. Catheterization therapy vs surgical closure in pediatric patients with patent ductus arteriosus: a meta-analysis. Clin Cardiol 2014;37:188-94. [Crossref] [PubMed]
- Backes CH, Rivera BK, Bridge JA. Percutaneous Patent Ductus Arteriosus (PDA) Closure During Infancy: A Meta-analysis. Pediatrics 2017. [Crossref] [PubMed]
- Backes CH, Cheatham SL, Deyo GM. Percutaneous Patent Ductus Arteriosus (PDA) Closure in Very Preterm Infants: Feasibility and Complications. J Am Heart Assoc 2016. [Crossref] [PubMed]
- Lawal TA, Gosemann JH, Kuebler JF. Thoracoscopy versus thoracotomy improves midterm musculoskeletal status and cosmesis in infants and children. Ann Thorac Surg 2009;87:224-8. [Crossref] [PubMed]
- Chen H, Weng G, Chen Z. Comparison of posterolateral thoracotomy and video-assisted thoracoscopic clipping for the treatment of patent ductus arteriosus in neonates and infants. Pediatr Cardiol 2011;32:386-90. [Crossref] [PubMed]
- Vanamo K, Berg E, Kokki H. Video-assisted thoracoscopic versus open surgery for persistent ductus arteriosus. J Pediatr Surg 2006;41:1226-9. [Crossref] [PubMed]
- Nezafati MH, Soltani G, Mottaghi H. Video-assisted thoracoscopic patent ductus arteriosus closure in 2,000 patients. Asian Cardiovasc Thorac Ann 2011;19:393-8. [Crossref] [PubMed]
- Villa E, Vanden Eynden F. Paediatric video-assisted thoracoscopic clipping of patent ductus arteriosus: experience in more than 700 cases. Eur J Cardiothorac Surg 2004;25:387-93. [Crossref] [PubMed]
- Burke RP, Jacobs JP, Cheng W. Video-assisted thoracoscopic surgery for patent ductus arteriosus in low birth weight neonates and infants. Pediatrics 1999;104:227-30. [Crossref] [PubMed]
- Hopper AN, Jamison MH, Lewis GW. Learning curves in surgical practice. Postgrad Med J 2007;83:777-9. [Crossref] [PubMed]
- Monteiro AJ, Canale LS, Rosa RV, et al. Minimally invasive thoracotomy (muscle-sparing thoracotomy) for occlusion of ligamentum arteriosum (ductus arteriosus) in preterm infants. Rev Bras Cir Cardiovasc 2007;22:285-90. [Crossref] [PubMed]
- Valentík P, Omeje I, Poruban R, et al. Surgical closure of patent ductus arteriosus in pre-term babies. Images Paediatr Cardiol 2007;9:27-36. [PubMed]
- Cho J, Yoon YH, Kim JT, et al. Patent ductus arteriosus closure in prematurities weighing less than 1 kg by subaxillary mini-thoracotomy. J Korean Med Sci 2010;25:24-7. [Crossref] [PubMed]
- Stankowski T, Aboul-Hassan SS, Marczak J, et al. Is thoracoscopic patent ductus arteriosus closure superior to conventional surgery? Interact Cardiovasc Thorac Surg 2015;21:532-8. [Crossref] [PubMed]
- Hines MH, Raines KH, Payne RM. Video-assisted ductal ligation in premature infants. Ann Thorac Surg 2003;76:1417-20. [Crossref] [PubMed]
- Avsar MK, Demir T, Celiksular C, et al. Bedside PDA ligation in premature infants less than 28 weeks and 1000 grams. J Cardiothorac Surg 2016;11:146. [Crossref] [PubMed]
- Nachira D, Meacci E, Porziella V. Learning curve of uniportal video-assisted lobectomy: analysis of 15-month experience in a single center. J Thorac Dis 2018;10:S3662-9. [Crossref] [PubMed]
- Liem NT, Tung CV, Van Linh N. Outcomes of thoracoscopic clipping versus transcatheter occlusion of patent ductus arteriosus: randomized clinical trial. J Pediatr Surg 2014;49:363-6. [Crossref] [PubMed]
- Chen H, Weng G, Chen Z. Comparison of long-term clinical outcomes and costs between video-assisted thoracoscopic surgery and transcatheter amplatzer occlusion of the patent ductus arteriosus. Pediatr Cardiol 2012;33:316-21. [Crossref] [PubMed]
- Jaureguizar E, Vazquez J, Murcia J. Morbid musculoskeletal sequelae of thoracotomy for tracheoesophageal fistula. J Pediatr Surg 1985;20:511-4. [Crossref] [PubMed]
- Shelton JE, Julian R, Walburgh E. Functional scoliosis as a long-term complication of surgical ligation for patent ductus arteriosus in premature infants. J Pediatr Surg 1986;21:855-7. [Crossref] [PubMed]