Unidirectionally thoracoscopic resection of lingual segment of the left upper pulmonary lobe
Introduction
Lung cancer is the leading cause of cancer-related mortality worldwide, more than 85% of which is non-small cell lung cancer (NSCLC) (1). Lung adenocarcinoma is not only one of the common types of non-small cell lung cancer, but also a pathological type with the highest incidence rate in non-smoking patients (2,3). Various histological subtypes of lung adenocarcinoma greatly differ in clinics, imaging, pathology, genetics, treatment and prognosis. In 2011, the International Association for the Study of Lung Cancer (IASLC), the American Thoracic Society (ATS) and the European Respiratory Society (ERS) jointly released the latest classification of lung adenocarcinoma (4), removed the concept of bronchioloalveolar carcinoma (BAC) that had been used for a long time, added the concepts of both adenocarcinoma in situ (AIS): a solitary pulmonary adenocarcinoma with tumor of ≤3 cm in diameter and tumor cell growth along the alveolar wall, but no stromal, vascular, or pleural invasion, and minimally invasive adenocarcinoma (MIA): a small adenocarcinoma with tumor of ≤3 cm in diameter, tumor cell growth along the alveolar wall and local infiltration lesion of ≤5 cm. Present studies have shown that the disease-free survival (DFS) in patients with AIS or MIA can achieve 100% if the tumor is completely removed (5). A patient with AIS undergoing unidirectionally thoracoscopic resection of lingual segment of the left upper pulmonary lobe and lymphadenectomy was reported in this video (Figure 1).

Case report
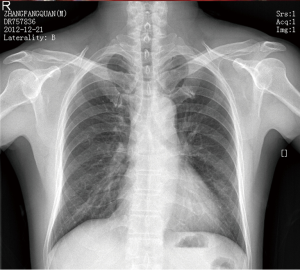
The 54-year-old male patient was admitted into the hospital due to repeated coughing for more than a month as well as chest tightness for more than 10 days. He had no history of smoking. At a local hospital before admission, he underwent enhanced chest CT scan, which showed a nodule of 18 mm × 15 mm in size on the lower lingual segment of the left upper pulmonary lobe, possibly considered as peripheral lung cancer. After admission, the patient underwent head MRI, bone scan, ultrasound examination of abdominal organs and other associated auxiliary examinations, and no distant metastases were found. Figure 2 shows the preoperative chest radiographs. Figure 3 shows the lesions in the lingual segment of the left upper pulmonary lobe. Considering preoperative auxiliary examinations, unidirectionally thoracoscopic resection of lingual segment of the left upper pulmonary lobe and lymphadenectomy was finally performed in the order of the lingual segmental vein, the lingual segmental bronchus, the lingual segmental artery, and the pulmonary tissues of the lingual segment in turn. The intraoperative frozen section pathological diagnosis was AIS.
Video description
Under combined intravenous general anesthesia, double-lumen endotracheal intubation and contralateral one-lung ventilation were performed. Double-lumen endotracheal intubation is essential for smooth thoracoscopic surgery. The lung tissue at the operating side should be fully collapsed so that the surgery can be successfully performed.
The patient is placed in a 90-degree position lying on the contralateral side.
The incision was designed appropriately for unidirectionally thoracoscopic resection of the left pulmonary lobe. An incision of about 1.5 cm was made as the thoracoscopic observation hole in the 7th intercostal space between the anterior axillary line and the median axillary line; then an incision of about 4 cm was made as the main operation hole in the 4th intercostal space between the anterior axillary line and the median clavicular line; and finally an incision of about 1.5 cm was made as the auxiliary operation role in the 9th intercostal space between the posterior axillary line and the inferior subscapular line. The surgeon stood at the ventral side of the patient and performed the surgery with a thoracoscopy device under the monitor.
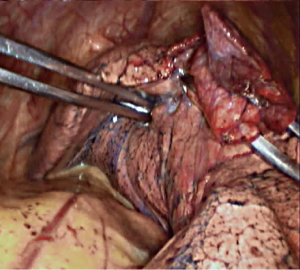
The surgery was performed unidirectionally, that is, the resection was performed right under the operation hole by first dissecting tissues to be resected from the most superficial structure, then exposing it, and finally cutting it off only in an unidirectionally gradual deepening way, and by disposing the pulmonary fissure without overturning the pulmonary lobe back and forth, up and down (7,8). In terms of the technical essentials, unidirectionally thoracoscopic resection of lingual segment of the left upper pulmonary lobe was basically the same as the right middle pulmonary lobectomy, with a resection direction of the lingual segmental vein, the lingual segmental bronchus, the lingual segmental artery, and the pulmonary tissues of the lingual segment in turn (9). The tissues were freed mainly with a coagulation hook and supportively with an aspirator. Vascular, bronchial and pulmonary parenchymas were handled with the endoscopic linear stapler or hem-o-lok clip applicator.
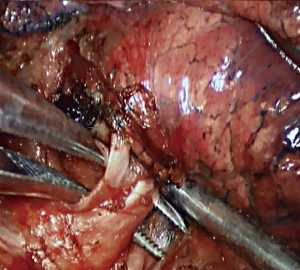
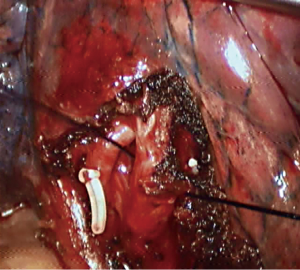
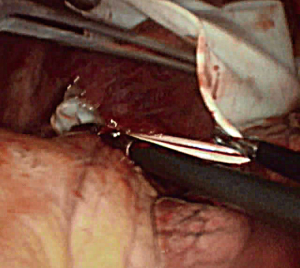
Step 1: Amputate the vein (Figures 4-6). Lingual segmental vein is the most subordinate branch of the left superior pulmonary vein. Pull back the pulmonary lobes, expose the lingual segmental vein, free it, and cut it off.
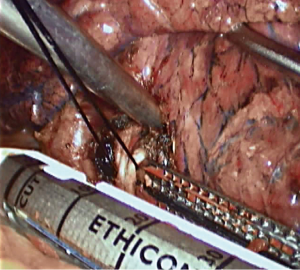
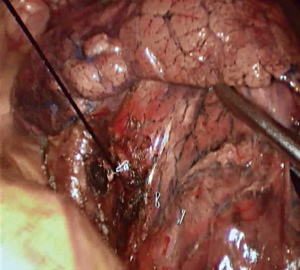
Step 2: Amputate the bronchus (Figures 7,8). Lingual segmental bronchus starts from the bronchial bifurcation of the upper pulmonary lobe. Free it, and cut it off.
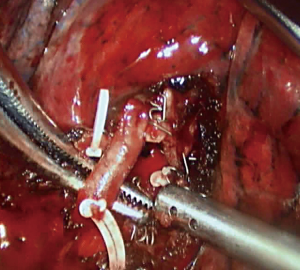
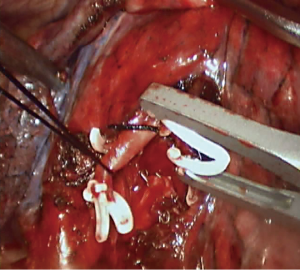
Step 3: Amputate the artery (Figures 9,10). Lingual segmental truncus arteriosus mainly supplies the blood for the lingual segment, which is the most anterior branches of the posterior lingual segmental artery. Lingual segmental artery starts from the front side of the pulmonary artery in the pulmonary fissure, and divides into two pulmonary segmental branches. During the surgery, the frontal segment of the oblique fissure was first opened, the start of the lingual segmental artery was found, then the oblique fissure was fully opened to the hilum of the lung, the trunk of the pulmonary artery between the lobes was exposed, the vascular sheath was opened and the lingual segmental artery was cut off (10).
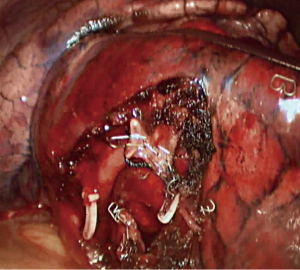
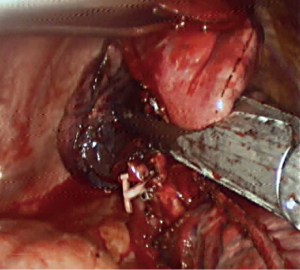
Step 4: Resect the pulmonary tissues of the lingual segment (Figure 11). Ventilate at the operational side to confirm the boundary of the lingual segment, and resect the pulmonary tissues of the lingual segment with the endoscopic linear stapler. Remove the specimen with a size 8 glove (Figure 12).
Comments
Thoracoscopy, also known as video-assisted thoracic surgery (VATS), is one of most attractive new technologies in the field of thoracic surgery in recent 30 years. By full thoracoscopic resection of lung cancer, the incision is small, only 3-5 cm in size, which avoids applying the rib spreader and cutting the chest muscles in large areas, thus better protecting the neuromuscular system, and significantly reducing postoperative pain, intraoperative hemorrhage and incidence rate of postoperative complications for patients. All these advantages are greatly appealing to both doctors and patients.
In the past, lobectomy with systematic lymphadenectomy was considered as the standard surgical procedure for treatment of early lung cancer (11). With the increasing popularity of lung cancer screening and gradual development of imaging technology, especially the emergence of multi-slice spiral CT scan technique, some small lung lesions can be found at an earlier stage; and clinical application of PET-CT allows more accurate early diagnosis and clinical staging of lung cancer than ever before; all of which have greatly improved the resection rate of early lung cancer. The establishment of a new classification of lung adenocarcinoma, especially the introduction of the concepts of AIS and MIA, has presented a new problem in the treatment of thoracic surgery: since the complete tumor removal can achieve complete healing of AIS and MIA, can pulmonary segmentectomy substitute pulmonary lobectomy in the treatment of AIS and MIA?
Video-assisted thoracoscopic segmentectomy is currently one of the most minimally invasive lung surgeries. In terms of pulmonary functions and anatomy, the lingual segmentectomy (S4 + S5) is equivalent to the right middle pulmonary lobectomy resection. From the technical perspective, the complexity of pulmonary segmental dissection and the individual variability of human anatomical structure are the greatest difficulties of pulmonary segmentectomy. First, surgeons should familiarize with the three-dimensional relationships among bronchus, artery and vein, and pay particular attention to the arterial anatomic variations that may occur. The anatomical structure of hilum of pulmonary segment has some certain regularities. In pulmonary segments, the arterial branch runs with the bronchi branch, while the pulmonary venous branch runs in the pulmonary segment receiving the venous blood from two adjacent pulmonary segments. The pulmonary segment artery may present with anatomical variations, of which the most common variation is that two corresponding arteries supply blood to the same pulmonary segment. Therefore, we should pay attention to whether there is another variant arterial branch when pulmonary segmental arteries are dissected. The most reliable way of determining pulmonary segmental variation is to determine the bronchus of appropriate pulmonary segment because pulmonary segmental bronchus seldom has variation. Second, we should determine the gap between pulmonary segments. It may be determined in accordance with inflatable pulmonary segments, by closing the pulmonary segmental bronchus in the collapsed pulmonary lobes and make it ventilated, the pulmonary segment to be resected is no longer inflated (12).
The literature has shown that pulmonary segmentectomy for early lung cancer, especially for tumors with a diameter of less than 2 cm can achieve a similar long-term survival rate as pulmonary lobectomy (13,14). However, the effectiveness and safety of pulmonary segmentectomy in treatment of AIS or MIA should be confirmed in further large-scale prospective studies.
Acknowledgements
Funding: Sub-subject of nationally supportive subject—Research and Application of Appropriate Technology in Control and Prevention of Common Diseases in Xinjiang Autonomous Region (Subject No. 2013 BAI05B02).
Disclosure: The authors declare no conflict of interest.
References
- NCCN clinical practice guidelines in oncology: non-small cell lung cancer Version 3, 2014. Fort Washington, PA: NCCN.
- Mindell JA, Maduke M. ClC chloride channels. Genome Biol 2001;2:REVIEWS3003.
- Schmidt-Rose T, Jentsch TJ. Transmembrane topology of a CLC chloride channel. Proc Natl Acad Sci U S A 1997;94:7633-8. [PubMed]
- Travis WD, Brambilla E, Noguchi M, et al. International association for the study of lung cancer/american thoracic society/european respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 2011;6:244-85. [PubMed]
- Russell PA, Wainer Z, Wright GM, et al. Does lung adenocarcinoma subtype predict patient survival?: A clinicopathologic study based on the new International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary lung adenocarcinoma classification. J Thorac Oncol 2011;6:1496-504. [PubMed]
- Cai K, Feng S, Wu H, et al. Unidirectionally thoracoscopic resection of lingual segment of the left upper pulmonary lobe. Asvide 2014;1:270. Available online: http://www.asvide.com/articles/283
- Liu LX, Che WG, Pu Q, et al. Single-direction VATS lobectomy. Chinese Journal of Clinical Thoracic and Cardiovascular Surgery 2008;24:156-8.
- Cai K, Wu H, Ren P, et al. Unidirectionally progressive resection of lower right lung cancer under video-assisted thoracoscopy. J Thorac Dis 2013;5:S310-4. [PubMed]
- Zhang XL, Liu LX. Progress of Thoracoscopic Pulmonary Segmentectomy for Early-Stage Non-small Cell Lung Cancer. Chinese Journal of Clinical Thoracic and Cardiovascular Surgery 2012;19:177-180.
- Gossot D. Atlas of Endoscopic Major Pulmonary Resections. Springer, 2010.
- Yan TD, Black D, Bannon PG, et al. Systematic review and meta-analysis of randomized and nonrandomized trials on safety and efficacy of video-assisted thoracic surgery lobectomy for early-stage non-small-cell lung cancer. J Clin Oncol 2009;27:2553-62. [PubMed]
- Zhang GL. Practical Chest Surgery. Beijing: Chinese Medical Science and Technology Press, 2007:1317.
- Read RC, Yoder G, Schaeffer RC. Survival after conservative resection for T1 N0 M0 non-small cell lung cancer. Ann Thorac Surg 1990;49:391-8; discussion 399-400. [PubMed]
- Koike T, Togashi K, Shirato T, et al. Limited resection for noninvasive bronchioloalveolar carcinoma diagnosed by intraoperative pathologic examination. Ann Thorac Surg 2009;88:1106-11. [PubMed]