
Prognostic value of neutrophil-to-lymphocyte, platelet-to-lymphocyte and lymphocyte-to-monocyte ratio ratios in patients operated on due to non-small cell lung cancer
Introduction
Lung cancer is the most frequently diagnosed malignant neoplasm (20% in males and 10% in females). Every year, about 22,000 Poles are affected by this disease. Of this number, 15,000 refer to males (1). In 2012, lung cancer claimed more than 1.6 million lives worldwide. It is estimated that in the year 2035, the disease will contribute to as many as 3 million deaths (2.1 million males and 0.9 million females) (2). The prognosis is bad. In Poland, the 5-year survival does not exceed 13.5%. A particular kind of neoplasm, its advancement, involvement of lymph nodes and presence of distant metastases are the most important prognostic factors (1).
Oncological studies are focused on searching for factors that, as early as in the diagnostics stage, would enable to determine a preliminary prognosis, which would in turn, allow to implement proper treatment, adjusted to a particular case (3-5). The search is multidirectional and includes cancer markers, tumour growth factors as well as any laboratory parameters. Their elevated values imply a body response to an ongoing neoplastic process. One of body reactions to neoplasia is an inflammatory response, particularly when the tumour is extensive and its breakdown products are absorbed into the bloodstream. On the one hand, inflammation stimulates immunity processes so it can inhibit the development of tumour. On the other hand, however, it favours carcinogenesis, growth of the tumour and its spread. The mechanism of the last process is related to suppression of immune response, inhibition of apoptosis and activation of angiogenesis. The inflammatory response of a body is reflected in various biochemical and haematological indices, available in a laboratory test panel (6,7). The neutrophil to lymphocyte ratio (NLR), the platelet to lymphocyte ratio (PLR) and the lymphocyte to monocyte ratio (LMR) are regarded as reliable markers of inflammatory processes, ongoing oncological patients. An elevated level of these ratios is a consequence of increased numbers of neutrophils, monocytes and platelets and/or a decreased number of lymphocytes, accompanying the neoplastic process (8-10).
The aim of the study is to evaluate of NLR, PLR and LMR ratios, being potential prognostic factors in patients surgically treated due to non-small cell lung cancer (NSCLC).
Methods
Studied population
In 2007–2014, 1,013 patients were treated surgically due to lung malignant neoplasms. Prior to the operation, all the patients had undergone chest CT and bronchoscopy. In the event of lymphadenopathy, the patients underwent mediastinoscopy and from 2008, also endobronchial ultrasound (EBUS) examinations. The patients who did not demonstrate metastases to lymph nodes or in whom the neoplasm involved only one group of nodes and the nodes were not conglomerated (PET) were qualified for the therapy. Upon admission to hospital, all the patients underwent basic laboratory tests, required before the operation. NLR, being a ratio of the absolute number of neutrophils and the absolute number of lymphocytes, PLR, being a ratio of the absolute number of platelets and the absolute number of lymphocytes and LMR, identified as a ratio of the absolute number of lymphocytes and the absolute number of monocytes, were calculated in all the studied patients.
Patient characteristics
A total of 532 patients, operated on in 2007–2014 due to NSCLC, were subject to an analysis. The Bioethics Committee of the Medical University of Lodz gave its consent for the study to be conducted (No. RNN/83/19/KE). Patients who had undergone wedge resections or segmentectomies were excluded from analyses. There were 174 females and 358 males, aged 36–84 years (the mean age: 63.6 years). Among them, 55% of patients admitted to nicotine addiction. Squamous cell carcinoma (269 cases), adenocarcinoma (204 cases), large cell carcinoma (43 cases) and mixed carcinoma (adenosquamous carcinoma: 16 cases) were indications for a surgical procedure.
The tumour was localized the right lung in 312 patients and in the left lung in 220 patients. The majority of patients (375 people), on the base of the CCI index, were categorized into grades 4–6. One hundred patients were categorized into grades 2 and 3 (Table 1).
Full table
Surgical treatment
Anatomical procedures, i.e., lobectomies, bilobectomies and pneumonectomies, were performed with a standard technique, through anterolateral thoracotomy under general anaesthesia with the use of a double-lumen tube. In the event of involvement of the right lung, a lymphadenectomy procedure was performed for lymph nodes of groups: 2R, 3A, 4R, 7, 8, 9, 10, 11. Patients were removed lymph nodes of groups: 3A, 4L, 5, 6, 7, 8, 9, 10, 11 if the left lung was involved in the neoplastic process. Minimum 6 node groups of N1 and N2 categories were dissected. The degree of tumour advancement was determined on the base of the TNM Classification of Malignant Tumours [Union for International Cancer Control (UICC) 2017, 8thedition], whereas the collected lymph nodes were classified in compliance with the Naruke map.
Statistical methods
The following factors were subject to a statistical analysis, conducted for determination of potential prognostic values of NLR, PLR and LMR ratios: age, sex, nicotinism, the number of leukocytes, neutrophils, monocytes, platelets, histopathological diagnosis, T category, N category, the Charlson comorbidity index (CCI), kind of surgery, patient survival.
- Continuous variables: verified with the Shapiro-Wilk test; due to lack of normal distribution, they were presented as medians and values of the lower and upper quartiles (25–75%) and further analyses—with the application of non-parametric tests;
- Comparisons of two groups—the U Mann-Whitney test. Comparisons of more groups—the Kruskall-Walis test with post-hoc comparisons, with the application of the Dunn-Bonferroni test;
- Nominal variables—as a number of observations and percent values, calculated for the study and control group. Comparisons—with the application of the Chi square test;
- Correlations—rank, non-parametric—the Spearman rank correlation;
- Survival analysis—the Kaplan-Meyer curves test, the Log-rank test, single-factor analysis of nominal parameters;
- Single-factor analysis of continuous parameters and multi-factor analysis—the Cox proportional hazard model.
Results
Surgical treatment and post-operative degree of advancement
A total of 220 patients (41%) were in stage I, 180 patients (34%) were in stage II and 132 were in stage III. The most common procedure was lobectomy—400 cases (75%). Pneumonectomy was performed in 94 patients (18%), whereas—bilobectomy—in 37 patients (7%). R0 resection was obtained in all patients. Three patients died postoperatively (one due to an infarct, one due perforation of gastric ulcer and one due to a stroke). Within 30 days, following the operation, another 5 patients died. Complications were observed in 116 patients (22%). In most cases, they were prolonged air leak (49 patients), 15 reoperation due to bleeding and bronchial fistula. Atelectasis in the site of the lung operation was observed in 13 cases. Thirteen patients required postoperative transfusion of packed red blood cells and 11 patients were treated due to a paroxysm of atrial fibrillation.
Overall survival (OS) of patients
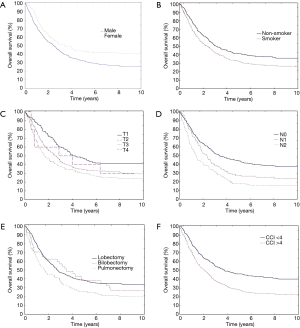
The mean OS was 50 months; 76% of patients survived 1 year, 57% of patients survived 2 years, 46% survived 3 years and 35% of patients survived 4 years. An analysis of OS revealed a significantly longer survival in females (median 3.42 vs. 2.46; P=0.00117) and non-smokers (median 3.09 vs. 2.36; P=0.006). Patients with T1 and T2 categories lived longer than those with T3 and T4 categories (P=0.0009); besides, the survival was longer in those with N0 category than in those with N1 and N2 categories (median respectively: 3.38 vs. 2.28 vs. 1.28; P=0.0000). Patients who underwent lobectomy or bilobectomy lived significantly longer than patients after pneumonectomy (median respectively 2.73 and 3.49 vs. 1.67; P=0.0046) (Figure 1A,B,C,D,E). Tumour localization (right lung/left lung; a particular lobe) did not contribute to statistical significance and did not significantly affect survival (respectively P=0.048; P=0.01). A kind of histopathological diagnosis did not have a significant effect on the lifespan (P=0.7) (Table 2). For CCI, being an index of comorbidity, the authors calculated the receiver operating characteristic (ROC) and determined the maximum values of sensitivity and specificity of the studied parameter. The optimal CCI limit value was 4. The lifespan of patients with CCI value above 4 was significantly shorter (3.76 vs. 2.13; P=0.0000) (Figures 1F,2A).
Full table
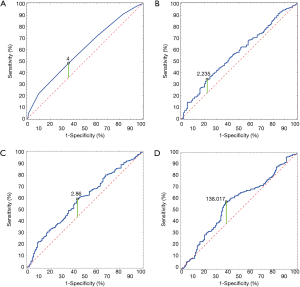
Morphological indices of inflammation as prognostic factors
In order to optimally determine limit values of NLR, PLR and LMR, the authors used ROC curves. The ROC curve enabled to calculate the area under the curve. The values for NLR, LMR and PLR were respectively: 0.587 (95% CI: 0.531–0.642, P=0.0024), 0.587 (95% CI: 0.531–0.642, P=0.0024) and 0.567 (95% CI: 0.509–0.624, P=0.0238). The optimal limit values for NLR, LMR and PLR were: 2.235, 2.86 and 138.017, respectively (Figure 2B,C,D).
A single-factor analysis revealed significant relationships between the values of NLR, PLR and LMR ratios, the CCI Index, the number of monocytes and OS of the patients (Table 3).
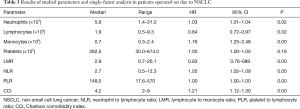
Full table
A multi-factor analysis of over 2-year survival showed that the stage of a neoplastic disease, the CCI value above 4 and the PLR value above 138 (P=0.0008) can be considered negative prognostic factors in patients operated on due to NSCLC. For over 5-year survival, none of the studied ratios (NLR, PLR and LMR) appeared to be an independent prognostic factor (Table 4).
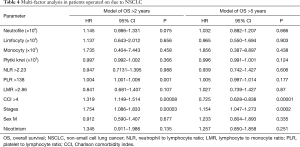
Full table
Discussion
Clinical studies definitely confirm that there is a close relationship between neoplasia and chronic inflammation. Leukocytosis, thrombocytosis, neutrophilia and lymphocytopenia, which occur in the inflammatory course, are considered negative prognostic factors (6). Inflammatory reaction which is ongoing in the tumour becomes generalised in the course of its growth, which leads to an increased number of leukocytes. The generalization of the inflammatory process results in weaker cell resistance, which is manifested with a reduced number of T lymphocytes (6,7). Cytokines which appear as a response to inflammation, stimulate megacariocytes, which in turn, increases the number of platelets in the course of neoplastic disease. Thrombocytaemia occurs in 39–57% of patients with NSCLC and is considered one of risk factors for metastasis (6). NLR, PLR and MLR ratios reflect mutual quantitative relationships of these haematocytes.
In our study, the single-factor analysis revealed a significant influence of preoperative values of NLR, PLR and LMR ratios on OS of patients after surgical treatment of NSCLC. Chen et al. confirmed preoperative values of the above ratios in patients with IB stage, operated on due to NSCLC. The multi-factor analysis revealed prognostic values of LMR and PLR ratios in patients with this disease stage (11). Results of our study indicate that the PLR ratio is characterised with the highest preoperative prognostic value. For PLR >138, over 2-year survival (OS >2) was significantly short (P=0.001). Such a relationship was not observed for OS >5. The multi-factor analysis confirmed that not only increased PLR values but also CCI >4 and IIA, IIB and IIIA stages are independent, negative prognostic factors.
Lee et al. made a similar observation. Apart from preoperative values of the PLR ratio, exceeding 180 and stages II and III according to the TNM Classification of Malignant Tumours, also age, male sex and postoperative radiotherapy appeared to be independent, negative prognostic factors of OS (12). Similarly, Xu et al. showed that PLR >135 constitutes an independent predictive factor for T category (13). A different study, conducted by Tod et al., revealed that the PLR ratio is a negative prognostic factor for patients treated surgically due to NSCLC and administered complementary chemotherapy (9). This observation corresponds to results of our study, in which we confirmed a prognostic relationship between the PLR ratio and the degree of advancement, i.e., stages II and III, according to the TNM Classification of Malignant Tumours, where a complementary therapy is usually implemented.
A meta-analysis, carried out by Qiang et al. on 1,554 patients from 7 centres, confirmed a high negative prognostic value of the PLR ratio in patients with NSCLC, shortening OS and disease-free survival (DFS). The authors pointed out that PLR, being a potential biomarker, is easily available and cheap (14).
A mutual interaction of platelets and leukocytes, being a response to an ongoing inflammatory process, might in future appear to be a useful and cheap marker, which will enable to implement a proper treatment and foresee prognosis. The presented results correspond to those obtained by other authors.
Conclusions
Increased values of NLR, PLR and LMR as well as the number of monocytes affect the survival period of NSCLC patients. The PLR ratio, the stage of the neoplastic disease and CCI above 4 are an independent and significant prognostic factor for expected, over 2-year survival of patients operated on due to NSCLC. The stage of tumour advancement and the CCI value are independent prognostic factors for 5-year survival.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The Bioethics Committee of the Medical University of Lodz gave its consent for the study to be conducted (No. RNN/83/19/KE).
References
- Krzakowski M, Jassem J, Antczak A, et al. Cancer of the lung, pleura and mediastinum. Oncol Clin Pract 2019;15. [Crossref]
- Didkowska J, Wojciechowska U, Mańczuk M, et al. Lung cancer epidemiology: contemporary and future challenges worldwide. Ann Transl Med 2016;4:150. [Crossref] [PubMed]
- Kim SH, Lee HW, Go SI, et al. Clinical significance of preoperative count and platelet-to lymphocyte ratio (PLT-PLR) in patients with surgically non-small cell lung cancer. Oncotarget 2016;7:36198-206. [PubMed]
- Remon J, Gorham J, Besse B, et al. Circulating free DNA new dynamic biomarker in non small cell lung cancer patients. Eur Respir J 2015;46:1548-50. [Crossref] [PubMed]
- Ichinose J, Murakawa T, Kawashima M, et al. Prognostic significance of red cell distribution width in elderly patients undergoing resections for non-small cell lung cancer. J Thorac Dis 2016;8:3658-66. [Crossref] [PubMed]
- Wócik E, Stasik Z, Rychlik U, et al. NLR and PLR ratios in prognostic assessment of non-small cell lung cancer patients. Diagn Lab 2016;52:13-20.
- Multhoff G, Moll M, Radons J. Chronic inflammation in cancer development. Front Immunol 2012;2. [Crossref] [PubMed]
- Deng M, Ma X, Liang X, et al. Are pretreatment neutrophil-lymphocyte ratio and platelet-lymphocyte ratio useful in predicting the outcomes of patients with small-cell lung cancer. Oncotarget 2017;8:37200-7. [PubMed]
- Toda M, Tsukioka T, Isumi N, et al. Platelet-to-lymphocyte ratio predict the prognosis of patient with non-small cell lung cancer treated with surgery and postoperative adjuvant chemotherapy. Thoracic Cancer 2018;9:112-9. [Crossref] [PubMed]
- Li W, Ma G, Wu Q, et al. Prognostic value of lymphocyte –to-monocyte ratio among Asian lung cancer patients: a systemic review and meta-analysis. Oncotarget 2017;8:110606-13. [PubMed]
- Chen Y, Wang W, Zhang X, et al. Prognostic significance of combined preoperative platelet-to-lymphocyte ratio and lymphocyte-to-monocyte ratio in patients undergoing surgery with IB stage non-small-cell lung cancer. Cancer Manag Res 2018;10:5411-22. [Crossref] [PubMed]
- Lee BM, Rodrigeuz A, Mena G, et al. Platelet-to-lymphocyte ratio and use of NSIADs during perioperative period as prognostic indicators in patients with NSCLC undergoing surgery. Cancer Control 2016;23:284-94. [Crossref] [PubMed]
- Xu F, Xu P, Cui W, et al. Neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios may aid and identifying patients with non-small cell lung cancer and predicting tumor-node-metastases stage. Oncol Lett 2018;16:483-90. [PubMed]
- Qiang G, Liang C, Xiao F, et al. Prognostic significance platelet-to-lymphocyte ratio in non-small-cell lung cancer: a meta-analysis. Onco Targets Ther 2016;9:869-76. [Crossref] [PubMed]


