
Risk factors for obstructive sleep apnea-related hypertension in police officers in Southern China
Introduction
Obstructive sleep apnea (OSA) is a highly prevalent sleep disorder characterized by recurrent episodes of apnea and hypopnea during sleep (1). Studies have shown that OSA is independent of other known risk factors for hypertension (2-4). The prevalence of hypertension in OSA patients ranges from 30% to 70%, while the prevalence of OSA ranges from 40% to 50% in hypertensive patients (3,4) and 70% to 83% in patients with resistant hypertension (4). Hypertension that is primarily caused or exacerbated by OSA is called OSA-related hypertension after excluding other definite secondary etiologies (e.g., renal parenchymal disease, renal artery stenosis, primary aldosteronism, pheochromocytoma) (5).
Over the past decade, the prevalence of OSA has gradually increased in the adult population (6,7), affecting 15% of men and 5% of women. Similarly, the prevalence of OSA-related hypertension is also on the rise. Previous studies of OSA-related hypertension have focused on its pathogenesis (8,9), effectiveness of treatment (10,11), or other aspects of the health condition. However, few studies have focused on its diagnosis, treatment, and risk factors in occupational groups.
In China, hypertension is the main risk factor for the morbidity and mortality of cardiovascular and cerebrovascular disease. The main purpose of treating hypertension is to minimize the occurrence of cardiovascular and cerebrovascular complications and reduce the overall risk of death. Effective blood pressure (BP) control should be within 140/90 mmHg (12). The treatment of hypertension patients with kidney disease, diabetes mellitus, or stable coronary heart disease should be more individualized, and BP control should be less than 130/80 mmHg. However, only 23% of hypertensive patients can achieve effective BP control level in the general population (12). In great part, this is because the treatment of hypertension is based solely on traditional drug treatment, ignoring secondary causes such as OSA (12).
From years of experience in our sleep centers, we found that many patients with hypertension also had snoring and sleep apnea, but the patients paid less attention to OSA problems. There were also many OSA patients with abnormal BP, but they had not been treated. In the population, especially in the occupational population, the prevalence and risk factors of OSA-related hypertension were unclear. OSA can lead to daytime sleepiness, increases in motor vehicle accidents (13), impaired work performance, and increased risk of work absenteeism (14). The policeman’s working life is irregular: It is common for them to stay up late and work overtime (15,16), they are under great pressure at work, and their sleep problems are more serious (17). In addition, they need to perform important and high-risk tasks. More attention should be paid to this group’s sleep problems. Therefore, we investigated the prevalence and risk factors of OSA-related hypertension in a group of Chinese police officers to establish a scientific basis for diagnosis and treatment.
Methods
This study was approved by the Ethics Committee of Guangdong General Hospital (Ethical code: 2017244H) and was registered with the United States of America Clinical Trials Registry, registry number NCT03280914.
The participants were a group of police officers who took part in the annual health examination from July to December 2017 in Guangzhou, Guangdong Province, China. All participants signed an informed consent form before completing the survey.
The study consisted of sleep-related questionnaires, a home sleep apnea test (HSAT), BP measurement, and a physical examination. The sleep-related questionnaires were distributed to participants during the annual health check-up and included items such as medical history, smoking and alcohol consumption, exercise frequency, the Epworth sleepiness scale (ESS) (18), the Berlin questionnaire (19), the hospital anxiety and depression scale (HADS) (20), and the insomnia severity index (ISI) (21). The physical examinations included measurements of height, weight, neck circumference, waist circumference, and hip circumference, and an assessment of hypertension.
OSA diagnosis
OSA diagnosis was carried out in two steps (Figure 1). Participants were first screened with the Berlin Questionnaire (19) to determine if they were at high risk for OSA. The Berlin Questionnaire is a validated screening tool that classifies people into high- and low-risk categories and covers three areas: snoring behavior (category 1), daytime sleepiness (category 2), and hypertension or obesity (category 3). The criterion for being at high risk is a positive score in at least two categories.
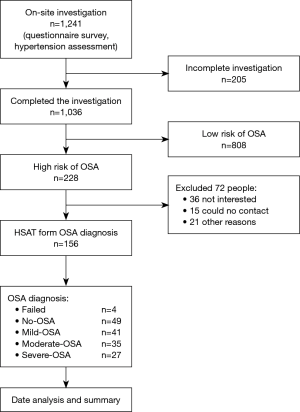
Individuals found to be at high risk of OSA based on the Berlin Questionnaire were administered an HSAT. Individuals were instructed not to drink alcoholic beverages, take drugs that affect sleep (including sleeping pills and analeptic drugs, but not including antihypertensive drugs), or perform any activities that might influence their sleep the night before the test. The participants were given a type 3 portable sleep monitor (ApneaLink Air, ResMed, Australia) with instructions on how to use it at home. The monitoring parameters included nasal airflow, blood oxygen saturation, heart rate, and chest and abdominal movement. Simultaneous recording of oxygen saturation and nasal airflow should take more than 4 hours; if the first monitoring fails, the measurement is repeated once.
Sleep monitor records were scored manually. According to the standard defined by the American Society of Sleep Medicine (22), apnea was defined as a drop of at least 90% in airflow from baseline lasting 10 seconds or longer. Hypopnea was defined as at least a 30% drop in airflow lasting at least 10 seconds with either an arousal or a 4% or greater drop in oxygen saturation. The apnea hypopnea index (AHI) (22) was used to define the total number of apnea and hypopnea events in an average hour of sleep. A diagnosis of OSA was defined as having an AHI of at least 5 events per hour with 5 to 14.9 events per hour considered mild OSA, 15 to 29.9 per hour moderate OSA, and 30 or more per hour severe OSA.
Physical examination and hypertension assessment
A physical examination was carried out on all participants as part of their annual health examination, including BP, height, weight, neck circumference, waist circumference, and hip circumference.
All participants were asked to sit quietly for 10 minutes before the initial BP measurement, then for an additional 1–2 minutes before a second measurement. BP was measured three times on the right arm and the average of the last two readings was used. BP was measured on three different days: on the days of start and end of HSAT, and the day of the physical examination. In line with the Chinese Hypertension Prevention Guide [2010] (11), hypertension was defined as either a systolic BP (SBP) ≥140 mmHg or a diastolic BP (DBP) ≥90 mmHg on at least 2 different days, or current use of antihypertensive drugs.
Definition of overweight and obesity
The subjects of this study were Chinese people, among whom the normal body mass index (BMI) ranges from 18.5 to 23.9 kg/m2, overweight is defined as BMI between 24 and 28 kg/m2, and obesity as BMI greater than 28 kg/m2 (23).
Definition of OSA-related hypertension
Hypertension that is primarily caused or exacerbated by OSA is called OSA-related hypertension (5). In this study, OSA-related hypertension was defined as meeting the diagnostic criteria for OSA and hypertension after excluding other definite secondary etiologies (e.g., renal parenchymal disease, renal artery stenosis, primary aldosteronism, pheochromocytoma) (5). Exclusion was performed based on patients’ medical history in this study.
Statistical analysis
All statistical analyses were performed using SPSS version 23.0 (IBM Corporation, Armonk, NY, USA). Continuous variables were presented as the mean with standard deviation (SD), and categorical variables as proportions (%). An independent-samples t-test was used to compare the continuous variables, and Pearson’s χ2 test was used to compare categorical variables. To determine the potential factors of OSA-related hypertension, bivariate correlation analysis was performed. Variables that were significant at a value of P<0.05 were then entered into a binary logistic regression analysis. Odds ratios (ORs) and 95% confidence intervals (CIs) for OSA-related hypertension in relation to potential risk factors were calculated. All statistical tests were two-sided and were considered statistically significant at a value of P<0.05.
Results
General characteristics and high risk of OSA screening
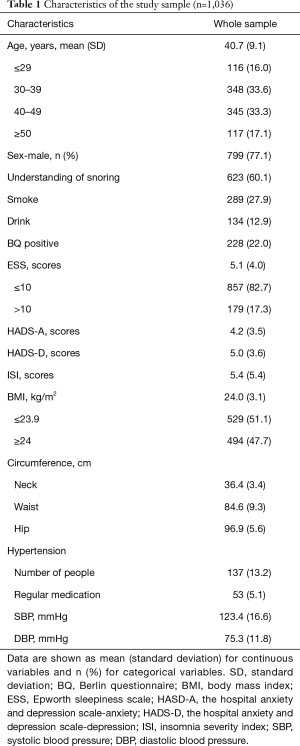
A total of 1,241 police officers were examined, but 205 were excluded because of incomplete information. Thus, 1,036 (799 males and 237 females) were included in the analysis. The mean age of the participants was 40.7±9.1 years (range, 22–63 years) with the age range of 30–49 years accounting for 66.9% of the study population. The mean BMI was 24.0±3.1 kg/m2, and 47.7% were overweight or obese. The Berlin Questionnaire showed that 22.0% (228/1,036) were at high risk for OSA (Table 1).
Full table
Prevalence of OSA
Among the group at high risk for OSA, 156 were willing to perform an HSAT. Nine people repeated HSAT monitoring because of insufficient time for the first monitoring, and four of them failed. Forty-nine had an AHI of lower than 5 events per hour, which did not conform to the diagnosis of OSA; 103 had an AHI of at least 5 events per hour, thereby meeting the diagnostic criteria for OSA used in this study. The prevalence of OSA in the police population was 9.9% (103/1,036).
Characteristics of OSA and OSA-related hypertension
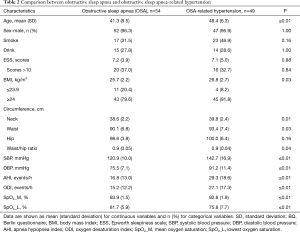
Of the 103 patients diagnosed with OSA, 54 OSA patients were without hypertension and 49 had both OSA and hypertension, which was considered OSA-related hypertension. Participants in the OSA-related hypertension group were older than in the OSA without hypertension group (P<0.001). The OSA-related hypertension group had a higher neck and waist circumference, AHI, BMI, oxygen desaturation index (ODI), and lower mean oxygen saturation and lowest oxygen saturation values than the OSA without hypertension group (Table 2).
Full table
Risk factors for OSA-related hypertension
To determine the influence of potential factors on OSA-related hypertension, bivariate correlation analysis was performed. Variables that were significant at a value of P<0.05 were then entered into a binary logistic regression analysis.
The bivariate correlation analysis included the following variables: age, BMI, neck circumference, waist circumference, hip circumference, smoking, family history of hypertension, AHI, ODI, mean oxygen saturation, lowest oxygen saturation, HADS-Anxiety, HADS-Depression, and ISI. The results showed that compared to OSA patients, the risk factors for OSA-related hypertension were: age (correlation coefficient r=0.42, P<0.001), BMI (r=0.22, P=0.03), neck circumference (r=0.26, P=0.01), waist circumference (r=0.22, P=0.03), AHI (r=0.36, P<0.001), ODI (r=0.37, P<0.001), mean oxygen saturation (r=−0.30, P=0.003), and lowest oxygen saturation (r=−0.40, P<0.001). Considering the collinearity between AHI and ODI, and mean oxygen saturation and lowest oxygen saturation, and given that in the diagnosis of OSA, AHI is the main diagnostic criterion (18), mean oxygen saturation can better describe the overall blood oxygen situation at night, so AHI and mean oxygen saturation were chosen for binary logistic regression analysis.
The results of the binary logistic regression analysis showed that age (OR =5.81, 95% CI: 1.78–18.98, P<0.01) and severity of OSA (OR =5.07, 95% CI: 1.72–14.89, P<0.01) were associated with increased risk of OSA-related hypertension. Mood is closely related to hypertension, so mood (depression and anxiety) status is calculated as a variable of adjustment. The results showed that age (adjusted OR =5.81, 95% CI: 1.80–18.80, P<0.01) and severity of OSA (adjusted OR =4.56, 95% CI: 1.49–13.93, P<0.01) were also associated with increased risk of OSA-related hypertension (Table 3).

Full table
Current status of diagnosis and treatment of OSA-related hypertension
Of the 103 people diagnosed with OSA, 47.6% (49/103) of them were also diagnosed with hypertension. In the OSA without hypertension group, all patients were newly diagnosed with OSA and none had ever received OSA treatment. In the hypertension without OSA group, 92% of the patients had previously been diagnosed with hypertension, but 65% had not received treatment. In the OSA-related hypertension group, none of the participants had previously been diagnosed with OSA, 30.6% (15/49) had not previously been diagnosed with hypertension, 55.1% (27/49) had never been treated for hypertension, and 100% had never been treated for OSA (Table 4).

Full table
Discussion
It is well-known that OSA is one of the most common causes of hypertension (2,3). The underlying mechanisms are generally related to sympathoexcitation (24), a proinflammatory state, and endothelial dysfunction (25). OSA induces intermittent hypoxemia and causes oxidative stress, leading to dysfunction of the vascular endothelium (24-27). Intermittent hypoxia triggers the chemo-reflex receptors that stimulate the sympathetic nervous system, leading to vasoconstriction, and may therefore result in hypertension. Another mechanism involves excess aldosterone (28), a hormone that regulates circulating blood volume and potassium concentration by regulating the feedback loop within the adrenal cortex. The prevalence of hypertension in patients with OSA ranges from 30% to 70% (3,4). An estimated 40% to 50% of hypertensive patients also have OSA, but these patients are often undiagnosed and untreated (29).
The prevalence of OSA varies among different occupational groups. For example, the prevalence of OSA in World Trade Center-exposed rescue/recovery workers was reported to be 36.5% (30), in firefighters 28.4% (31), in North American police officers 33.6% (32), and in trucking-sector operators 15–30% (33). Our study shows that the prevalence of OSA in Chinese police officers was about 10%, which is comparable to that of the general population (1), but lower than that in other occupational groups (30-32). One possible reason is a difference in weight. For instance, obesity is considered a major risk factor for OSA. The rate of overweight/obese individuals in our study was 32.7%, which was lower than that of firemen (79.7%) (31) or rescue/recovery workers (87.6%) (30). Further, HSAT was only offered to participants identified as being at high risk for OSA by the Berlin Questionnaire; it was not offered to all personnel.
Previous studies focused only on the prevalence of OSA and work hazards associated with OSA (30-33), and no attention was paid to the content of OSA-related hypertension, such as diagnostic rate and treatment rate. From years of experience in our sleep centers, we have found that most patients with OSA-related hypertension never had an HSAT or polysomnography (PSG) testing. These patients were typically only treated with one or more traditional antihypertensive drugs, but their efficacy was not good. Current guidelines recommend that patients with hypertension, particularly those with difficult-to-treat hypertension, should be screened for OSA (34,35); however, it is difficult to implement this in clinical practice. In order to draw people’s attention to OSA and OSA-related hypertension, we selected a group of police officers to investigate OSA and hypertension. Our aims were to clarify the prevalence of OSA and OSA-related hypertension in this population and to explore related risk factors. Our results show that the prevalence of OSA in police officers in southern China is comparable to that of the general population, and that half of the OSA patients also have hypertension, which is considered OSA-related hypertension. Further, age and severity of OSA were identified as risk factors for OSA-related hypertension. Previous studies have shown (36,37) that patients with OSA have a 1.96-fold higher risk of hypertension than those without OSA, patients with an AHI of 15 or higher have a 3.2-fold increase for developing hypertension than those without OSA. Therefore, in addition to actively treating moderate to severe OSA patients, advocating for a change in the therapeutic model for young snorers and patients with mild OSA to reduce their weight may help reduce the risk of OSA-related hypertension.
We found that the prevalence of OSA-related hypertension was high, but the diagnostic and therapeutic rates were very low. All patients with OSA or OSA-related hypertension had never been diagnosed or treated before this study, which is consistent with the current status of OSA clinical diagnosis and treatment (12). Therefore, the standardized diagnosis and treatment of OSA-related hypertension should receive adequate attention.
Our study also showed that the prevalence of OSA in police officers in southern China was close to that of the general population, but the prevalence of OSA-related hypertension was high and the diagnostic and therapeutic rates were low.
OSA often causes a special type of hypertension (2,4,26,27) with a “non-dipper” change in circadian rhythm of BP, and studies have shown that the fluctuations of BP over time are closely related to the development and progression of target organ disease and confer an increased risk of cardiovascular events, poor vascular outcome, neurocognitive dysfunction, and kidney disease. Continuous positive airway pressure (CPAP) is the first-line treatment for OSA. Effective CPAP treatment can reduce BP in OSA-related hypertension (10,11), improve effective BP control level, reduce the dosage or number of antihypertensive drugs, and improve nocturnal BP fluctuation patterns, which ultimately contributes to the prevention of cardiovascular disease (38) and other adverse outcomes (39). However, the multi-center SAVE study showed that CPAP treatment did not prevent secondary cardiovascular events in moderate-to-severe OSA patients with established cardiovascular disease (39), suggesting that early detection, diagnosis, and treatment of OSA may reduce the prevalence of hypertension caused by OSA and help prevent cardiovascular events and other adverse consequences. In recent years, annual occupational medical examinations have become an important part of health management, but OSA screening has never been included in such health management. We believe that early prevention and treatment of OSA may be achieved by screening for OSA among people undergoing annual occupational medical examinations; therefore, we recommend that these examinations include OSA screening.
Further intervention and follow-up are needed with the police officers in this study to determine whether CPAP treatment can reduce the incidence of hypertension in patients with only OSA, and whether it can effectively control the BP of OSA-related hypertension patients.
One limitation of this study was that HSAT was only performed in participants identified as being at high risk for OSA by the Berlin questionnaire, which may have resulted in a lower prevalence of OSA in our study population. Another potential limitation was that, unlike PSG, HSAT did not record arousals and hours of sleep, which may underestimate the severity of OSA and lead to misdiagnosis of some patients with mild OSA.
Conclusions
Among the policemen of southern China, a higher prevalence was detected not only in OSA but also in OSA-related hypertension. Age and severity of OSA are risk factors for OSA-related hypertension after mood (depression and anxiety) status adjustment. However, OSA-related hypertension has not received sufficient attention in Chinese police officers. OSA and hypertension screening should be included in routine physical examinations of the healthy occupational population to encourage early intervention and treatment.
Acknowledgments
We would like to thank Editage (www.editage.cn) for English language editing.
Funding: This work was supported by Science and Technology Planning Project of Guangdong Province, China (2016A020216030 to Q Ou), the National Natural Science Foundation of China (818870077 to Q Ou), and the National Natural Science Foundation of China (81771799 to H Liu).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was approved by the Ethics Committee of Guangdong General Hospital (Ethical code: 2017244H). Informed consent was obtained from all participants prior to inclusion in the study.
References
- Huang QR, Qin Z, Zhang S, et al. Clinical patterns of obstructive sleep apnea and its comorbid conditions: a data mining approach. J Clin Sleep Med 2008;4:543-50. [PubMed]
- Nieto FJ, Young TB, Lind BK, et al. Association of sleep-disordered breathing, sleep apnea, and hypertension in a large community-based study. Sleep Heart Health Study. JAMA 2000;283:1829-36. [Crossref] [PubMed]
- Shahar E, Whitney CW, Redline S, et al. Sleep-disordered breathing and cardiovascular disease: cross-sectional results of the Sleep Heart Health Study. Am J Respir Crit Care Med 2001;163:19-25. [Crossref] [PubMed]
- Martínez-García MA, Gómez-Aldaraví R, Gil-Martínez T, et al. Sleep-disordered breathing in patients with difficult-to-control hypertension. Arch Bronconeumol 2006;42:14-20. [Crossref] [PubMed]
- Hypertension Professional Committee of Chinese Medical Association. Expert consensus on clinical diagnosis and treatment of obstructive sleep apnea-related hypertension. Chin J Pract Intern Med 2013;12:435-41.
- Young T, Peppard PE, Gottlieb DJ. Epidemiology of obstructive sleep apnea: a population health perspective. Am J Respir Crit Care Med 2002;165:1217-39. [Crossref] [PubMed]
- Peppard PE, Young T, Barnet JH, et al. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol 2013;177:1006-14. [Crossref] [PubMed]
- Lavie L. Obstructive sleep apnoea syndrome—an oxidative stress disorder. Sleep Med Rev 2003;7:35-51. [Crossref] [PubMed]
- Dopp JM, Reichmuth KJ, Morgan BJ. Obstructive sleep apnea and hypertension: mechanisms, evaluation, and management. Curr Hypertens Rep 2007;9:529-34. [Crossref] [PubMed]
- Durán-Cantolla J, Aizpuru F, Montserrat JM, et al. Continuous positive airway pressure as treatment for systemic hypertension in people with obstructive sleep apnoea: randomised controlled trial. BMJ 2010;341:c5991. [Crossref] [PubMed]
- Bazzano LA, Khan Z, Reynolds K, et al. Effect of nocturnal nasal continuous positive airway pressure on blood pressure in obstructive sleep apnea. Hypertension 2007;50:417-23. [Crossref] [PubMed]
- China Hypertension Prevention and Treatment Guidelines Revision Committee. China hypertension prevention and treatment guidelines. Chin J Cardiol 2010;39:701-8.
- Tregear S, Reston J, Schoelles K, et al. Obstructive sleep apnea and risk of motor vehicle crash: systematic review and meta-analysis. J Clin Sleep Med 2009;5:573-81. [PubMed]
- Kessler RC, Berglund PA, Coulouvrat C, et al. Insomnia and the performance of US workers: results from the America Insomnia Survey. Sleep 2011;34:1161-71. [Crossref] [PubMed]
- Vila B. Impact of long work hours on police officers and the communities they serve. Am J Ind Med 2006;49:972-80. [Crossref] [PubMed]
- Garbarino S, De Carli F, Nobili L, et al. Sleepiness and sleep disorders in shift workers: a study on a group of Italian police officers. Sleep 2002;25:648-53. [PubMed]
- Neylan TC, Metzler TJ, Best SR, et al. Critical incident exposure and sleep quality in police officers. Psychosom Med 2002;64:345-52. [Crossref] [PubMed]
- Johns Murray W. A new method for measuring daytime sleepiness: the Epworth Sleepiness Scale. Sleep 1991;14:540-45. [Crossref] [PubMed]
- Netzer NC, Stoohs RA, Netzer CM, et al. Using the Berlin Questionnaire to identify patients at risk for the sleep apnea syndrome. Ann Intern Med 1999;131:485-91. [Crossref] [PubMed]
- Snaith RP. The Hospital Anxiety and Depression Scale. Health Qual Life Outcomes 2003;1:29. [Crossref] [PubMed]
- Morin CM, Belleville G, Bélanger L, et al. The Insomnia Severity Index: psychometric indicators to detect insomnia cases and evaluate treatment response. Sleep 2011;34:601. [Crossref] [PubMed]
- Collop NA, Anderson WM, Boehlecke B, et al. Clinical guidelines for the use of unattended portable monitors in the diagnosis of obstructive sleep apnea in adult patients. J Clin Sleep Med 2007;3:737-47. [PubMed]
- Jia W. Obesity in China: its characteristics, diagnostic criteria, and implications. Front Med 2015;9:129-33. [Crossref] [PubMed]
- Lesske J, Fletcher EC, Bao G, et al. Hypertension caused by chronic intermittent hypoxia—influence of chemoreceptors and sympathetic nervous system. J Hypertens 1997;15:1593-603. [Crossref] [PubMed]
- Hoyos CM, Melehan KL, Liu PY, et al. Does obstructive sleep apnea cause endothelial dysfunction? A critical review of the literature. Sleep Med Rev 2015;20:15-26. [Crossref] [PubMed]
- Lattanzi S, Brigo F, Silvestrini M. Blood pressure profile and nocturnal oxygen desaturation. J Clin Hypertens (Greenwich) 2018;20:656-8. [Crossref] [PubMed]
- Lattanzi S, Brigo F, Silvestrini M. Obstructive sleep apnea syndrome and the nocturnal blood pressure profile. J Clin Hypertens (Greenwich) 2018;20:1036-8. [Crossref] [PubMed]
- Calhoun DA, Nishizaka MK, Zaman MA, et al. Aldosterone excretion among participants with resistant hypertension and symptoms of sleep apnea. Chest 2004;125:112-7. [Crossref] [PubMed]
- Logan A G, Perlikowski S M, Mente A, et al. High prevalence of unrecognized sleep apnoea in drug-resistant hypertension. J Hypertens 2001;19:2271-7. [Crossref] [PubMed]
- Webber MP, Lee R, Soo J, et al. Prevalence and incidence of high risk for obstructive sleep apnea in World Trade Center-exposed rescue/recovery workers. Sleep Breath 2011;15:283-94. [Crossref] [PubMed]
- Barger LK, Rajaratnam SM, Wang W, et al. Common sleep disorders increase risk of motor vehicle crashes and adverse health outcomes in firefighters. J Clin Sleep Med 2015;11:233-40. [PubMed]
- Rajaratnam SM, Barger LK, Lockley SW, et al. Sleep disorders, health, and safety in police officers. JAMA 2011;306:2567-78. [Crossref] [PubMed]
- Howard ME, Desai AV, Grunstein RR, et al. Sleepiness, sleep-disordered breathing, and accident risk factors in commercial vehicle drivers. Am J Respir Crit Care Med 2004;170:1014-21. [Crossref] [PubMed]
- Parati G, Lombardi C, Hedner J, et al. Recommendations for the management of patients with obstructive sleep apnoea and hypertension. Eur Respir J 2013;41:523-38. [Crossref] [PubMed]
- Parati G, Lombardi C, Hedner J, et al. Position paper on the management of patients with obstructive sleep apnea and hypertension: joint recommendations by the European Society of Hypertension, by the European Respiratory Society and by the members of European COST (COoperation in Scientific and Technological Research) ACTION B26 on obstructive sleep apnea. J Hypertens 2012;30:633-46. [Crossref] [PubMed]
- Pankow W, Lies A, Lohmann FW. Sleep-disordered breathing and hypertension. N Engl J Med 2000;343:966-author reply 967. [Crossref] [PubMed]
- Peppard PE, Young T, Palta M, et al. Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 2000;342:1378-84. [Crossref] [PubMed]
- MacMahon S, Peto R, Cutler J, et al. Blood pressure, stroke, and coronary heart disease. Part 1, Prolonged differences in blood pressure: prospective observational studies corrected for the regression dilution bias. Lancet 1990;335:765-74. [Crossref] [PubMed]
- McEvoy RD, Antic NA, Heeley E, et al. CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med 2016;375:919-31. [Crossref] [PubMed]


