Shape of concentration-response curves between long-term particulate matter exposure and morbidities of chronic bronchitis: a review of epidemiological evidence
Introduction
Chronic exposure to particulate matter (PM) has been associated with increased morbidity and mortality of cardiopulmonary diseases, loss of lung function, increased risk of lung cancer, and reduced life expectancy (1-17). Understanding the shape of the concentration-response (C-R) function between the extent of air pollution and various adverse health effects, grouped by susceptibility to disease, has played a critical role in efforts to evaluate the impact of various levels of PM pollution on health, assess the adequacy of regulatory standards and determine the health consequences associated with changes in policy and pollution levels (1-4,18-29). Many epidemiological studies have analyzed the C-R relationship between ambient PM and cardiopulmonary mortality using various statistical models (24-29). Although the shapes of the estimated C-R curves did not differ significantly from linear and were not consistent with well-defined thresholds, some studies found that the slope of the C-R function was steeper at lower than at higher concentrations (28,29).
Most previous studies assessing the effects of long-term exposure to PM on cardiopulmonary mortality have been performed in areas with annual average PM less than 2.5 µm in diameter (PM2.5) concentrations ranging from 5 to 35 µg/m3, with fewer conducted in areas with much higher concentrations of ambient PM, such as China and India (30). A recent prospective study of 1.2 million American adults found that the C-R relationships associated with PM2.5 were qualitatively different for lung cancer and cardiovascular disease (CVD) mortality. For lung cancer, the C-R curve was almost linear, whereas for CVD the C-R curve was nonlinear, with a steep increase in risk at low exposures and flattening out at higher exposures. These results suggest that inappropriate extrapolations of linear exposure-response functions for CVD may substantially overestimate the disease burden of CVD attributable to PM in areas of very high exposure and may underestimate disease burden in areas of relatively low exposure (31). Ambient PM pollution was estimated to account for 3.1% of the deaths and disability-adjusted life years (DALYs) in the Global Burden of Disease Study 2010 (GBD2010), a sharp increase from the 0.4% DALY observed in GBD2000 (32). Much of this increase was due to the use of an integrated exposure-response curve to estimate a continuous and nonlinear risk function between PM2.5 and CVD mortality across the full range of PM concentrations (30-32).
Increased morbidities, including chronic bronchitis and respiratory symptoms such as persistent cough and phlegm, are among the leading deleterious health consequences of long-term PM exposure (1-8). Although these outcomes are not as serious as mortality from and hospitalization for acute cardiopulmonary diseases, they may have greater effects on public health and quality of life, since these chronic health effects involve a greater proportion of the population (33). Although many studies have assessed the detailed characteristics of the C-R curves showing the relationships between PM levels and mortality, fewer have examined the shapes of the C-R curves showing the relationships between PM and chronic respiratory diseases or symptoms, with most employing simple straight lines or scatter graphs (5-8). Even less is known about the C-R curves in Asia and Africa because fewer studies have been performed at higher PM ranges (29-33). Data on multicity morbidity may be more difficult to obtain than data on multicity mortality, with no review or meta-analysis to date systematically exploring the shapes of the C-R curves relating long-term PM exposure and chronic bronchitis symptoms.
Multicity studies on the C-R relationships between long-term PM exposure and symptoms of chronic bronchitis
Use of the keyword combinations of “PM” and/or “air pollution”, “bronchitis”, and “respiratory symptoms” to screen all available papers in Medline identified a total of ten studies showing the C-R curves or scatter plots relating PM concentrations and morbidity of chronic bronchitis symptoms (Table 1). Of these ten papers published from 1989-2012, nine were performed in the United States and European countries with low to moderate PM pollution (5,7,34-40), with one study performed in China with high PM pollution (TSP =188-689 µg/m3) (41). Six of these ten studies were cross-sectional surveys showing the C-R relationships in multiple cities with different PM levels, three were repeated cross-sectional surveys over periods of time, and one was a prospective cohort study analyzing the C-R relationships between decreasing PM pollution and possible reductions in the prevalence of respiratory symptoms. Seven of the ten studies were performed in children and adolescents, and three in adults. Chronic bronchitis was assessed in four studies, and chronic cough and/or phlegm as the health endpoints in six studies. Five showed the C-R curves or straight lines, and five just showed scatter plots. Two studies showed the C-R curves associated with the susceptibility status of the subjects, whereas the other eight did not. Among the five studies showing the C-R curves, along with the adjusted prevalence and PM levels, three used simple linear regression to draw the straight lines or curves, one used natural splines to draw C-R curves, and one used cubic splines and adjusted for the co-exposure to the air pollutants SO2 and NO2 as confounders.
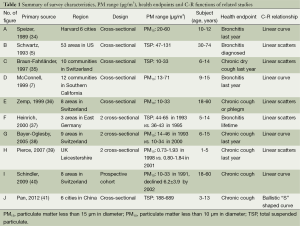
Full table
Shapes of the C-R curves and scatter plots
Figure 1 shows the shapes of the C-R curves and scatter plots in the ten papers listed chronologically. These included the five studies in which the crude or adjusted prevalence of bronchitis symptoms was plotted against PM concentrations, yielding apparently linear “C-R scatter plots”, but did not report C-R curves.
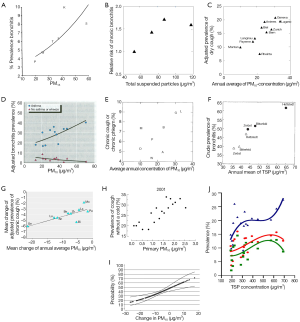
The earliest study, the Harvard Six Cities Study, showed a linear C-R relationship between PM15 level (range, 20-59 µg/m3) and the prevalence of bronchitis for the first time among children aged 10-12 years [(34) (Figure 1A)]. Further analysis showed that a linear C-R relationship existed only among children with asthma/wheeze, but not in other children (42). Two other studies showed a linear C-R scatter plot between TSP level (range, 47 to 131 µg/m3) and the relative risk of chronic bronchitis in adults from 53 urban areas of the United States [(5) (Figure 1B)], and between PM10 level (range, 10 to 33 µg/m3) and the adjusted prevalence of nocturnal dry cough among schoolchildren from ten communities in Switzerland [(35) (Figure 1C)]. A linear C-R relationship was observed when PM10 level (range, 13 to 71 µg/m3) was plotted against the adjusted prevalence of bronchitis among children with asthma, but not for children without asthma or wheeze, in 12 communities in Southern California [(7) (Figure 1D)]; these results confirmed the findings of the Harvard Six Cities Study (42). C-R scatter plots relating PM10 level (range, 10 to 33 µg/m3) and the prevalence of chronic cough or phlegm production in adults aged 18-60 years from eight regions of Switzerland [(36) (Figure 1E)], and between TSP concentration (range, 36 to 65 µg/m3) and the crude prevalence of bronchitis among schoolchildren in three regions of the former East Germany (Figure 1F), were also determined (37). A linear C-R relationship was observed between decrease in PM10 level (range, 0 to –20 µg/m3) and the temporal changes in the prevalence of chronic cough (range, –1% to –6%) among schoolchildren in nine areas of Switzerland [(38) (Figure 1G)]. The latter results confirmed that even a moderate decrease in PM exposure could contribute to a reduced prevalence of chronic cough in Swiss schoolchildren. Moreover, these findings did not suggest a threshold for the adverse effects of PM10, because the beneficial effects of rather small PM10 reductions could be observed in such a moderately polluted environment. A linear scatter plot related low PM10 levels (range, 0 to 2.5 µg/m3) with the prevalence of chronic cough among a random sample of children aged 1-5 years from the Leicestershire Health Authority Child Health Database in the UK [(39) (Figure 1H)].
A linear relationship was observed between reductions in PM10 levels (range, –30 to 20 µg/m3) and the reduced prevalence of chronic cough over an 11 year follow up period among adults in eight areas of Switzerland [(40) (Figure 1I)]. The Swiss cohort study on air pollution and lung disease in adults (SAPALDIA) confirmed the findings of a cross-sectional study of successive age cohorts of Swiss schoolchildren [(38) (Figure 1F)], with both studies indicating that reductions in ambient particle concentrations may have beneficial effects on the respiratory health of adults within a few years, even in areas with moderate to low levels of air pollution, providing further evidence for the linearity of the C-R relationship between PM10 levels and bronchitis symptoms. In contrast, ballistic “S”-shaped C-R curves were observed between TSP level and the adjusted prevalence of chronic cough among children in 18 districts of six cities in China [(41) (Figure 1J)]. Susceptible children had significantly higher and steeper C-R curves than did nonsusceptible children across the entire high TSP (range, 188 to 689 µg/m3), consistent with the findings of the Harvard Six City Study (34) and of a study of 12 communities in Southern California (7).
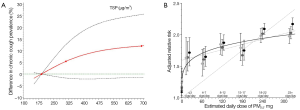
Figure 2 compares the shape of the C-R curves relating the prevalence of chronic cough and TSP level among Chinese children in six cities [(41), Figure 2A] with the shape of the C-R curves analyzing the adjusted relative risks of cardiovascular mortality and estimated average daily dose of PM2.5 from cigarette smoke and air pollution in a prospective cohort of 1.2 million American adults [(19), Figure 2B]. Both figures exhibited similar ballistic “S” shaped C-R curves, showing that these relationships were relatively steep in the low to moderate PM range and flattened out in the high PM range.
Summary and discussion
To our knowledge, this is the first review to systematically evaluate the C-R curves relating long-term PM exposure and morbidity of chronic bronchitis symptoms. Similar to multicity studies on the C-R relationships between PM and cardiovascular mortality, most of the ten studies relating PM exposure and bronchitis symptoms were conducted in the low to moderate PM range and among children. All ten studies showed linear, non-threshold C-R relationships in the low to moderate PM range for both children and adults, findings observed even in the very low PM10 range (0.73-1.93 µg/m3) among UK children aged 1-5 years [(39), Figure 1H]. The linear C-R curves and scatter plots observed in the prospective cohort of Swiss adults [(40), Figure 1I] and the three repeated cross-sectional surveys in children from East Germany [(37), Figure 1F], Switzerland [(38), Figure 1G] and the UK [(39), Figure 1H] confirm the linear C-R relationships observed in the six cross-sectional studies, suggesting that the C-R relationships between PM reductions and beneficial effects on respiratory health may be attributable not only to the decreased incidence of respiratory symptoms, but to an increased recovery in persons symptomatic at baseline [(40), Figure 1I].
Nonlinear relationships in the entire PM range
Interestingly, similar ballistic “S” shaped C-R curves are observed for the relationship between TSP exposure and morbidity from chronic cough in Chinese children [(41), Figure 2A] and the relationship between PM2.5 and cardiovascular mortality in a prospective cohort of American adults [(19), Figure 2B], suggesting that the C-R curves relating PM exposure with both mortality and morbidity are relatively steep at very low to moderate levels of exposure and flatten out at high levels of exposure. Numerous hypotheses have sought to explain the mechanisms leading to a nonlinear C-R relationship; these include errors in calculating exposure levels at high levels of pollution, the existence of competing risks, preferential avoidance based on symptoms or sensitivity, decreased inhalation, differential toxicities, and biological saturation (19,41). Any increase in infiltration and/or clearance mechanisms resulting from elevated PM exposure may yield a nonparallel relationship between the PM doses to which subjects are exposed and inhaled tissue levels in the lower respiratory tract, particularly in the bronchial airways of the trachea (43).
Susceptibility
Only two studies showed that susceptible children had 2- to 3-fold elevated C-R curves compared with nonsusceptible children: children from Southern California [(7), Figure 1D] and China [(41), Figure 1J]. These findings suggest that genetic susceptibility is important in explaining the differences in baseline response levels among subjects exposed to the same levels of PM. Susceptible children have steeper C-R curves and exhibit greater prevalence changes across the entire TSP range, as determined by the prevalence of persistent cough and bronchitis for each 100 µg/m3 increment in TSP, than do nonsusceptible children, suggesting that susceptible children respond to a greater extent to changes in PM concentration than do nonsusceptible children (41). Additional studies are warranted to compare the characteristics of the C-R curves for susceptible and nonsusceptible populations across the entire PM pollution range, as well as to identify a biologically plausible mechanism that results in one population being more susceptible than another to PM-related health effects (44).
Conclusions
The C-R curve relating PM exposure and morbidity of chronic bronchitis symptoms is probably ballistic ‘S’ shaped, linear in the low to moderate PM range, and flattening out in the high PM range. Noticeable differences were observed in the shape and magnitude of the C-R curves for susceptible and nonsusceptible populations, based on the limited number of cross-sectional or prospective cohort studies published. Additional multi-area studies are required to assess the characteristics of the C-R functions between PM (specific size fractions, sources and components) and multiple chronic respective health end points (bronchitis and asthmatic symptoms), especially in areas with high levels of PM pollution. The advanced statistical models used in evaluating the C-R relationships between PM and mortality should be introduced to analyze the C-R relationships between PM and morbidity. More attention should be paid to susceptibility and confounding factors. New evidence, particularly from prospective cohort studies in developing areas with high PM pollution, will have important scientific and public health implications in assessing the DALY of PM exposure and related air quality standards.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Pope CA 3rd, Dockery DW. Health effects of fine particulate air pollution: lines that connect. J Air Waste Manag Assoc 2006;56:709-42. [PubMed]
- WHO. Effects of air pollution on children’s health and development: a review of the evidence. Special programme on health and development. European Center for environment and health. Bonn, Germany: World Health Organization, 2005.
- Rückerl R, Schneider A, Breitner S, et al. Health effects of particulate air pollution: A review of epidemiological evidence. Inhal Toxicol 2011;23:555-92. [PubMed]
- Anderson JO, Thundiyil JG, Stolbach A. Clearing the air: a review of the effects of particulate matter air pollution on human health. J Med Toxicol 2012;8:166-75. [PubMed]
- Schwartz J. Particulate air pollution and chronic respiratory disease. Environ Res 1993;62:7-13. [PubMed]
- Peters JM, Avol E, Navidi W, et al. A study of twelve Southern California communities with differing levels and types of air pollution. I. Prevalence of respiratory morbidity. Am J Respir Crit Care Med 1999;159:760-7. [PubMed]
- McConnell R, Berhane K, Gilliland F, et al. Air pollution and bronchitic symptoms in Southern California children with asthma. Environ Health Perspect 1999;107:757-60. [PubMed]
- Dockery DW, Cunningham J, Damokosh AI, et al. Health effects of acid aerosols on North American children: respiratory symptoms. Environ Health Perspect 1996;104:500-5. [PubMed]
- Schwartz J, Marcus A. Mortality and air pollution in London: a time series analysis. Am J Epidemiol 1990;131:185-94. [PubMed]
- Dockery DW, Pope CA 3rd, Xu X, et al. An association between air pollution and mortality in six U.S. cities. N Engl J Med 1993;329:1753-9. [PubMed]
- Pope CA 3rd, Thun MJ, Namboodiri MM, et al. Particulate air pollution as a predictor of mortality in a prospective study of U.S. adults. Am J Respir Crit Care Med 1995;151:669-74. [PubMed]
- Laden F, Schwartz J, Speizer FE, et al. Reduction in fine particulate air pollution and mortality: Extended follow-up of the Harvard Six Cities study. Am J Respir Crit Care Med 2006;173:667-72. [PubMed]
- Pope CA 3rd, Burnett RT, Thun MJ, et al. Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. JAMA 2002;287:1132-41. [PubMed]
- Zanobetti A, Schwartz J. The effect of fine and coarse particulate air pollution on mortality: a national analysis. Environ Health Perspect 2009;117:898-903. [PubMed]
- Raizenne M, Neas LM, Damokosh AI, et al. Health effects of acid aerosols on North American children: pulmonary function. Environ Health Perspect 1996;104:506-514. [PubMed]
- Gauderman WJ, Avol E, Gilliland F, et al. The effect of air pollution on lung development from 10 to 18 years of age. N Engl J Med 2004;351:1057-67. [PubMed]
- Pope CA 3rd, Ezzati M, Dockery DW. Fine-particulate air pollution and life expectancy in the United States. N Engl J Med 2009;360:376-86. [PubMed]
- Aunan K. Exposure-response functions for health effects of air pollutants based on epidemiological findings. Risk Anal 1996;16:693-709. [PubMed]
- Pope CA 3rd, Burnett RT, Krewski D, et al. Cardiovascular mortality and exposure to airborne fine particulate matter and cigarette smoke: shape of the exposure-response relationship. Circulation 2009;120:941-8. [PubMed]
- Rothenberg SJ, Rothenberg JC. Testing the dose-response specification in epidemiology: public health and policy consequences for lead. Environ Health Perspect 2005;113:1190-5. [PubMed]
- Smith KR, Peel JL. Mind the gap. Environ Health Perspect 2010;118:1643-5. [PubMed]
- Tillett T. A break in the continuum: analyzing the gap in particle exposure research. Environ Health Perspect 2010;118:a543. [PubMed]
- U.S. EPA. Integrated Science Assessment for Particulate Matter (Final Report). U.S. Environmental Protection Agency, Washington, DC, EPA/600/R-08/139F, 2009.
- U.S. EPA. Quantitative health risk assessment for particulate matter. North Carolina: U.S. EPA; 2010. EPA-452/R-10-005.
- Daniels MJ, Dominici F, Zeger SL, et al. The National Morbidity, Mortality, and Air Pollution Study. Part III: PM10 concentration-response curves and thresholds for the 20 largest US cities. Res Rep Health Eff Inst 2004;(94 Pt 3):1-21; discussion 23-30. [PubMed]
- Schwartz J. The effects of particulate air pollution on daily deaths: a multi-city case crossover analysis. Occup Environ Med 2004;61:956-61. [PubMed]
- Pope CA 3rd. Particulate matter-mortality exposure-response relations and threshold. Am J Epidemiol 2000;152:407-12. [PubMed]
- Schwartz J, Marcus A. Mortality and air pollution in London: a time series analysis. Am J Epidemiol 1990;131:185-94. [PubMed]
- Samoli E, Analitis A, Touloumi G, et al. Estimating the exposure-response relationships between particulate matter and mortality within the APHEA multicity project. Environ Health Perspect 2005;113:88-95. [PubMed]
- Balakrishnan K, Cohen A, Smith KR. Addressing the burden of disease attributable to air pollution in India: the need to integrate across household and ambient air pollution exposures. Environ Health Perspect 2014;122:A6-7. [PubMed]
- Pope CA 3rd, Burnett RT, Turner MC, et al. Lung cancer and cardiovascular disease mortality associated with ambient air pollution and cigarette smoke: shape of the exposure-response relationships. Environ Health Perspect 2011;119:1616-21. [PubMed]
- Lim SS, Vos T, Flaxman AD, et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012;380:2224-60. [PubMed]
- Ostro B. Outdoor air pollution. WHO Environmental Burden of Disease Series, 2004;5.
- Speizer FE. Studies of acid aerosols in six cities and in a new multi-city investigation: design issues. Environ Health Perspect 1989;79:61-7. [PubMed]
- Braun-Fahrländer C, Vuille JC, Sennhauser FH, et al. Respiratory health and long-term exposure to air pollutants in Swiss schoolchildren. SCARPOL Team. Swiss Study on Childhood Allergy and Respiratory Symptoms with Respect to Air Pollution, Climate and Pollen. Am J Respir Crit Care Med 1997;155:1042-9. [PubMed]
- Zemp E, Elsasser S, Schindler C, et al. Long-term ambient air pollution and respiratory symptoms in adults (SAPALDIA study). The SAPALDIA Team. Am J Respir Crit Care Med 1999;159:1257-66. [PubMed]
- Heinrich J, Hoelscher B, Wichmann HE. Decline of ambient air pollution and respiratory symptoms in children. Am J Respir Crit Care Med 2000;161:1930-6. [PubMed]
- Bayer-Oglesby L, Grize L, Gassner M, et al. Decline of ambient air pollution levels and improved respiratory health in Swiss children. Environ Health Perspect 2005;113:1632-7. [PubMed]
- Pierse N, Rushton L, Harris RS, et al. Locally generated particulate pollution and respiratory symptoms in young children. Thorax 2006;61:216-20. [PubMed]
- Schindler C, Keidel D, Gerbase MW, et al. Improvements in PM10 exposure and reduced rates of respiratory symptoms in a cohort of Swiss adults (SAPALDIA). Am J Respir Crit Care Med 2009;179:579-87. [PubMed]
- Pan G, Kagawa J, Zhang S, et al. Comparison of total suspended particulate concentration-response relationships for respiratory symptoms between Chinese children with a different susceptibility status. Sci Total Environ 2012;421-422:111-7. [PubMed]
- Speizer FE. Asthma and persistent wheeze in the Harvard Six Cities Study. Chest 1990;98:191S-5S. [PubMed]
- Sarnat SE, Coull BA, Ruiz PA, et al. The influences of ambient particle composition and size on particle infiltration in Los Angeles, CA, residences. J Air Waste Manag Assoc 2006;56:186-96. [PubMed]
- Sacks JD, Stanek LW, Luben TJ, et al. Particulate matter-induced health effects: who is susceptible? Environ Health Perspect 2011;119:446-54. [PubMed]


