
Initial pH and shockable rhythm are associated with favorable neurological outcome in cardiac arrest patients resuscitated with extracorporeal cardiopulmonary resuscitation
Introduction
Refractory cardiac arrest, usually defined as persistent cardiac arrest despite 30 minutes of medical cardio-pulmonary resuscitation (CPR) (1), is associated with an extremely low probability of survival with conventional CPR. In this setting, extracorporeal CPR with veno-arterial extracorporeal membrane oxygenation (ECMO) has been found to be associated with better neurological outcome compared to conventional CPR (2-4). A recent analysis of the Extracorporeal Life Support Organization (ELSO) database showed that the use of extracorporeal CPR (eCPR) has increased during the last decade (5). Because no prospective randomized trial is available, there is currently no strong recommendation that precisely details the exact indications for eCPR. Indeed, the guidelines of the European Research Council of 2015 only stated that eCPR should be considered as a rescue therapy for patients in whom initial advanced life support measures are unsuccessful (6). Because eCPR is associated with a very high mortality rate, identification of early prognostic factors is needed for avoiding futile care. In the setting of out of hospital cardiac arrest (OHCA), a recent metanalysis has identified initial shockable rhythm, low flow duration, pH and lactate values as prognostic factors (7). In the setting of in-hospital cardiac arrest (IHCA), initial shockable rhythm, low flow duration, lactate, creatinine and Sequential Organ Failure Assessment (SOFA) have been found to be associated with outcome (8). While such factors obviously reflect severity, data regarding to critical thresholds are lacking.
Objectives of this retrospective chart review were first, to identify factors associated with a favorable neurological outcome, and second, to build a score calculable at the time of ECMO insertion predicting the prognosis of patients benefiting from eCPR.
Methods
Population
We performed a retrospective chart review of all patients admitted to our tertiary regional intensive care unit (ICU) who underwent eCPR implantation for refractory cardiac arrest between February 2010 and March 2017. Cases were identified through a prospectively maintained electronic database. Refractory cardiac arrest was defined as persistent cardiac arrest after 15 minutes of medical CPR (9). Because of a potentially different prognosis (10,11), patients suffering from post-cardiac arrest cardiogenic shock were excluded. After initial resuscitation, for persistent comatose patients, decision of withdrawing life-support was based upon multimodal evaluation indicating a poor neurological outcome. This decision was made in the ICU during a collegial meeting associating experienced medical doctors and nurses (12,13).
This was a retrospective observational study reporting data of a large cohort and not about an individual patient. Therefore, according to French legislation, neither informed consent nor approval of the ethics committee was required. However, general information of the families was made in the ICU by a poster explaining that data concerning their next of kin might be used later for scientific analyses and could be published with anonymized data unless they express their opposition. No opposition was expressed.
Local procedure for eCPR patient’s selection
Between 2010 and 2015, decision to perform eCPR was based upon the French recommendations of 2009 (1). Then, a protocol has been implemented at our institution in 2015. In the setting of refractory OHCA, the patient was transferred to our hospital under automatized mechanical chest compressions (Lucas®, Chest Compression System, Physio-Control Inc., Redmond, WA, USA) by helicopter when available. At the same time, the emergency medical dispatcher reached the on call cardiac surgeon for eCPR if five pre-specified criterions were both present: (I) suspected cardiac origin, (II) age younger than 65-year-old, (III) witnessed collapse and immediate efficient CPR, (IV) expected time between collapse and eCPR start below 150 minutes, (V) EtCO2 at any time <5 mmHg no longer than 5 minutes.
eCPR management
After cut-down cannulation performed at the femoral site, targeted ECMO blood flow was set at 2.4 L/min/m2. A canula was inserted in the superficial femoral artery to prevent limb ischemia. Targeted temperature management (TTM) was let at the discretion of the clinician in charge but usually followed the TTM recommendation during the first 24 hours. When intra-abdominal pressure was rising leading to insufficient ECMO blood flow, the patient was sedated and paralyzed. Patients were weaned from ECMO if they had a left ventricular ejection fraction >25%, and maintained an aortic time-velocity integral >10 cm and a lateral mitral annulus peak systolic velocity >6 cm/s at ECMO minimal flow, with low dose of vasopressors (14).
Variables assessment
Demographic data collected included age, gender, cause of cardiac arrest, and any known comorbidities. Cardiac arrest related data included the out-of-hospital or in-hospital setting, witnessed, duration of no flow and low flow, and initial rhythm. Low flow duration was defined as time between collapse and effective eCPR start. In hospital treatments included percutaneous coronary intervention, use of vasopressors and inotropes, left ventricle unloading, renal replacement therapy, mechanical ventilation and transfusion. Severity related data included blood lactates and pH at the time of admission to the operating room and just before eCPR start, or just after initiation of ECMO, and 24 hours later, SOFA and SAPS2 scores. ICU length of stay were also recorded. ECMO related data included duration of ECMO and successful weaning. ECMO related complications included bleeding, and lower limb ischemia. Bleedings were defined as type 3 of the Bleeding Academic Research Consortium (BARC) (15). Occurrence of abdominal compartment syndrome (ACS) was also recorded. Because of the particular setting of ECMO, ACS was defined as the association of intra-abdominal pressure above 15 mmHg and insufficient ECMO blood flow. Intra-abdominal pressure was assessed by the validated trans-bladder technic (16).
Endpoints
Primary objective was to identify factors associated with survival with favorable neurological outcome at ICU discharge, defined by a Cerebral Performance Category scale (CPC) score of 1 or 2 (12).
Statistical analysis
Qualitative variables were expressed as number and percentage and compared using the Fisher’s exact test. Quantitative variables were expressed as median and interquartile range and compared using the Wilcoxon test. After description of the study population, a univariate analysis comparing patients with favorable neurological outcome (CPC 1 or 2) and patients with poor neurological outcome (CPC 3 to 5) was performed. Factors associated with CPC 1 or 2 by univariate analysis were dichotomized using the median value of the overall population. Finally, two logistic regression models with multivariable backward analysis were performed in order to identify variables independently associated with favorable neurological outcome. First, a complete model, using all factors associated with CPC 1 or 2 identified by univariate analysis. Second, and an “admission” model including only data available at the time of operating room admission and before eCPR start. The variables of the second model were secondly chosen in order to build a predictive score. All analyses were performed using SAS 9.4 (SAS Institute Inc, Cary, NC, USA).
Results
Population characteristics
Between February 2010 and March 2017, 113 adult patients benefited from eCPR in the University Hospital of Besancon, including 53 OHCA (47%). Two patients died in the operating room because of cannulation failure. Sixteen out of 60 IHCA patients were already in the ICU at the time of cardiac arrest. Baseline characteristics of the population and differences between IHCA and OHCA patients are reported in Table 1.
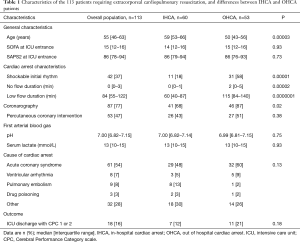
Full table
eCPR
The cannulas were surgically inserted in all cases. All except two were inserted through femoral vessels. In the whole cohort, median fluid intake during the first 24 hours was 7.0 [4.0–8.5] L. Median extracorporeal life support duration was 39 [11–96] hours in the whole cohort, and 82 [64–120] hours in long term survivors. Forty-four patients (39%) died before 24 hours of eCPR. Among the 69 patients alive at 24 hours, data on ECMO blood flow was available for 67, and its median value was 3.7 [3.1–4.2] L/min. Twenty-one patients (19%) have benefited from left ventricle unloading technic. Among them, 15 patients (13%) were assisted by intra-aortic balloon pump. Nine patients (8%) needed another left ventricle venting technic, including surgical apical venting (n=5), atrioseptotomy (n=3), and PulseCath™ LV21 catheter (Intra-Vasc NL, Groningen, The Netherlands) (n=1).
Prognosis
At ICU discharge, 19 patients were alive (17%), of whom 18 had a CPC 1 or 2 (16%). At day 90, all of the 18 alive patients were CPC 1 or 2 (16%). Three out of 11 OHCA patients (27%) with a low flow duration longer than 150 minutes were discharged alive from ICU with CPC 1 or 2. Median ICU length of stay was 2 [0–7] days in the whole cohort, and 12 [10–18] days in patients with CPC 1 or 2.
Prognostic factors
By univariate analysis, initial rhythm, first arterial pH, serum bicarbonates, aspartate aminotransferase, creatinine and lactates concentrations, ACS during ICU stay, SOFA and SAPS2 at ICU entrance were associated with neurological outcome at ICU discharge (Table 2). Because low-flow duration was not associated with neurological outcome at ICU discharge by univariate analysis, it was not considered in the multivariate analysis.
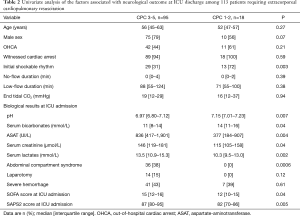
Full table
By multivariate analysis, in a complete model, initial shockable rhythm [(OR 4.7; 95% CI, 1.2–18.2), pH ≥7.0 (OR 7.4; 95% CI, 1.3–41.1)], and serum creatinine <141 µmol/L (OR 4.3; 95% CI, 1.1–17.5) were independently associated with a favorable neurological outcome at ICU discharge. In the “admission” model using only parameters available at the operating room before eCPR start, initial shockable rhythm (OR 5.9; 95% CI, 1.8–19.6) and pH ≥7.0 (OR 3.9; 95% CI, 1.1–14.4) were independently associated with a favorable neurological outcome at ICU discharge (Table 3).
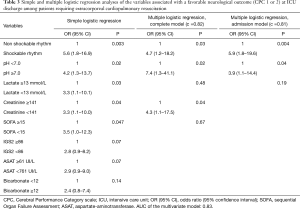
Full table
Considering these results, we established a prognostic score integrating initial shockable rhythm and pH ≥7.0 on the first arterial blood gas (Table 4). A score of 1 corresponded to a patient who met both two criterions. A score of 2 corresponded to a patient who met one of the two criterions. A score of 3 corresponded to a patient who met none of the criterions. All the patients presenting with a score of 3 died in ICU. The negative predictive value for CPC 1 or 2 of a score ≤2 was 100% (Table 5).

Full table
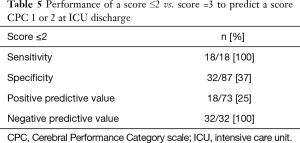
Full table
Complications
Complications are reported in Table S1. Thirty-six patients (32%) had ACS. Their median low flow duration was 110 [83–133] minutes. Their median fluid intake during the 24 first hours was 8 [7–10] L. Out them 14 (12%) underwent laparostomy for ACS. In this subgroup, median red blood cell transfusion was 7 [4–14]. All of these 36 patients died after a median duration of 19 [6–48] hours.
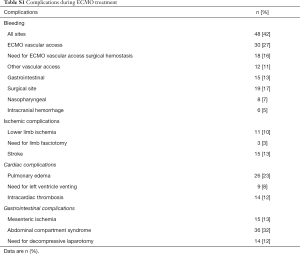
Full table
Discussion
In this retrospective review of our eCPR experience, we found a 16% survival at ICU discharge with CPC 1 or 2, with no difference between OHCA and IHCA. In our cohort, all patients presenting with initial non-shockable rhythm and first pH <7.0 died in the ICU.
Outcome
Our results regarding the neurological outcome in OHCA patients are consistent with those previously published. Indeed, in a recent meta-analysis of 15 observational studies on 841 OHCA patients, survival with favorable neurological outcome was 15% (7).
However, in contrast with previously published studies, we found only a 12% survival with favorable neurological outcome in IHCA. In the meta-analysis by D’Arrigo et al. 35% of the 635 patients treated by eCPR for IHCA were discharged alive with CPC 1 or 2 (8). Controversial results are reported and are explained by an older age, different etiology, severity of the patients. Our poorer results in this setting might be explained by several factors. First, this subgroup was characterized by a high rate of non-shockable initial rhythm (82%). Second, 26% of them were already in ICU at the time of cardiac arrest, reflecting a high baseline severity. Third, in our institution cardiac surgeons are on-call. Compare to on-duty surgical team, it is likely to favor longer low-flow time.
Prognostic risk factors
We found that only initial shockable rhythm, first serum creatinine <141 µmol/L and first arterial pH ≥7.0 were independently associated with survival with favorable neurological outcome. These results are partially consistent with those previously published. Higher arterial pH was previously reported to be associated with favorable neurological outcome only in OHCA patients (7). In both OHCA and IHCA, previously published prognostic factors were initial shockable rhythm, but also shorter low-flow duration and lower serum lactate concentration (7,8). It is likely that in our cohort, low-flow duration was overall too long to remain a prognostic factor. We cannot exclude that a lack of power could explain the absence of correlation observed between low flow and prognosis.
Although some experts recommend avoiding eCPR when low flow duration is longer than 60 minutes (9), we measured in the setting of OHCA large low-flow duration within 115 [84–140] minutes. This result might reflect the combination of both the rural area of our region and the absence of an ECMO mobile team. However, interestingly, 3 out of 11 OHCA patients presenting with a low flow duration longer than 150 minutes were discharged alive from ICU with CPC 1 or 2. Based on our results, we propose a prognostic score based upon initial shockable rhythm and first arterial pH. When the patients had both non-shockable rhythm and pH <7.0, the negative predictive value for survival with CPC 1 or 2 was 100%. Interestingly, this score does not consider low-flow duration. This can be of interest considering that exact assessment of low-flow duration might be difficult, with sometimes alternance of return of spontaneous circulation and cardiac arrest. Whereas longer low flow duration is a risk factor for ischemia phenomenon, we think that arterial pH is a more direct reflect of this ischemia phenomenon. Indeed, the first pH might take into account both the low-flow duration and the efficiency of cardiopulmonary resuscitation. Sampled just before arterial catheterization, it might help to identify patients in whom eCPR is futile. The interest of a score for eCPR eligibility, based upon analysis of the first rhythm and first pH, which is simple, and could be calculable before ECMO insertion, should be prospectively evaluated.
ACS
Bowel swelling due to non-occlusive mesenteric ischemia might be responsible for hypovolemia and intra-abdominal hypertension, both favoring a decrease of ECMO preload. Because fluid infusion is necessary to restore ECMO preload and blood flow, eCPR patients are at high risk of ACS. After ensuring of the correct position of the venous canula into the right atrium, laparostomy might appear as a relevant therapeutic option in order to stop this vicious circle. However, we found that laparostomy only delay death by a few hours at the price of massive transfusion. Indeed, laparostomy in the setting of anti-thrombotic treatment for acute coronary syndrome, is likely to favor bleeding. In this work, none of the patients requiring laparostomy for ACS survived.
Study limitations
Our study has several limitations. First, because of the retrospective design, some important data such as TTM are lacking. However, benefit of such intervention during eCPR remains uncertain (17). Second, because we aimed to assess the global effectiveness of eCPR in our institution, we have analyzed OHCA and IHCA together, which might be discussed as previous studies have reported a better outcome in IHCA. However, as others, we did not find any difference regarding to neurological outcome between IHCA and OHCA. This might be due to a very low number of initial shockable rhythm in the IHCA subgroup (18%). Moreover, despite in-hospital setting, median low flow duration was 60 [40–87] minutes, which is more than previously reported (18-20). Third, timing for first arterial blood gas sampling was not standardized. While it was sampled on the extra-corporeal circuit just after ECMO start when cannulation was performed in the operating room, timing for arterial blood gas sampling was let to the discretion of intensivist when eCPR was performed in ICU. This might theoretically limit the use of arterial pH to decide to not perform eCPR. However, it is very unlikely that an arterial pH sampled before eCPR show a value higher than after eCPR start. Fourth, because we think that they are not relevant in the setting of ECMO, we did not use criterions of ACS of the World Society of Abdominal Compartment Syndrome to define ACS. Indeed, because of the negative pressure generated in the inferior vena cava, intra-abdominal hypertension below 20 mmHg might lead to insufficient ECMO preload. As a consequence, we used more pragmatical criterions as intra-abdominal pressure above 15 mmHg and decrease ECMO blood-flow despite of adequate fluid expansion. This threshold of 15 mmHg corresponds to a grade 2 intra-abdominal hypertension. While questionable, use of such pragmatical criterions in the setting of ECMO has been previously reported (21,22).
Conclusions
In a mixed population of OHCA and IHCA, 18 patients (16%) were discharged alive of ICU with CPC 1 or 2. Despite a shorter low flow time in IHCA, there was not any significant difference in neurological outcome between OHCA and IHCA. At the time of eCPR start, only initial shockable rhythm and arterial pH ≥7.0 predicted neurological outcome. A selection of the patients who might benefit from eCPR, based upon initial rhythm and arterial pH rather than on low flow time, should be further evaluated.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. According to French legislation, neither informed consent nor approval of the ethics committee was required. However, general information of the families was made in the ICU by a poster explaining that data concerning their next of kin might be used later for scientific analyses and could be published with anonymized data unless they express their opposition. No opposition was expressed.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Conseil français de réanimation cardiopulmonaire, Société française d’anesthésie et de réanimation, Société française de cardiologie, et al. Guidelines for indications for the use of extracorporeal life support in refractory cardiac arrest. French Ministry of Health. Ann Fr Anesth Reanim 2009;28:182-90.
- Ouweneel DM, Schotborgh JV, Limpens J, et al. Extracorporeal life support during cardiac arrest and cardiogenic shock: a systematic review and meta-analysis. Intensive Care Med 2016;42:1922-34. [Crossref] [PubMed]
- Matsuoka Y, Ikenoue T, Hata N, et al. Hospitals’ extracorporeal cardiopulmonary resuscitation capabilities and outcomes in out-of-hospital cardiac arrest: a population-based study. Resuscitation 2019;136:85-92. [Crossref] [PubMed]
- Patricio D, Peluso L, Brasseur A, et al. Comparison of extracorporeal and conventional cardiopulmonary resuscitation: a retrospective propensity score matched study. Crit Care 2019;23:27. [Crossref] [PubMed]
- Richardson ASC, Schmidt M, Bailey M, et al. ECMO Cardio-Pulmonary Resuscitation (ECPR), trends in survival from an international multicentre cohort study over 12-years. Resuscitation 2017;112:34-40. [Crossref] [PubMed]
- Soar J, Nolan JP, Böttiger BW, et al. European Resuscitation Council Guidelines for Resuscitation 2015: Section 3. Adult advanced life support. Resuscitation 2015;95:100-47. [Crossref] [PubMed]
- Debaty G, Babaz V, Durand M, et al. Prognostic factors for extracorporeal cardiopulmonary resuscitation recipients following out-of-hospital refractory cardiac arrest. A systematic review and meta-analysis. Resuscitation 2017;112:1-10. [Crossref] [PubMed]
- D’Arrigo S, Cacciola S, Dennis M, et al. Predictors of favourable outcome after in-hospital cardiac arrest treated with extracorporeal cardiopulmonary resuscitation: A systematic review and meta-analysis. Resuscitation 2017;121:62-70. [Crossref] [PubMed]
- Guglin M, Zucker MJ, Bazan VM, et al. Venoarterial ECMO for Adults: JACC Scientific Expert Panel. J Am Coll Cardiol 2019;73:698-716. [Crossref] [PubMed]
- Pineton de Chambrun M, Bréchot N, Lebreton G, et al. Venoarterial extracorporeal membrane oxygenation for refractory cardiogenic shock post-cardiac arrest. Intensive Care Med 2016;42:1999-2007. [Crossref] [PubMed]
- Bougouin W, Aissaoui N, Combes A, et al. Post-cardiac arrest shock treated with veno-arterial extracorporeal membrane oxygenation: An observational study and propensity-score analysis. Resuscitation 2017;110:126-32. [Crossref] [PubMed]
- Sandroni C, Cariou A, Cavallaro F, et al. Prognostication in comatose survivors of cardiac arrest: an advisory statement from the European Resuscitation Council and the European Society of Intensive Care Medicine. Resuscitation 2014;85:1779-89. [Crossref] [PubMed]
- Nolan JP, Soar J, Cariou A, et al. European Resuscitation Council and European Society of Intensive Care Medicine 2015 guidelines for post-resuscitation care. Intensive Care Med 2015;41:2039-56. [Crossref] [PubMed]
- Aissaoui N, Luyt C-E, Leprince P, et al. Predictors of successful extracorporeal membrane oxygenation (ECMO) weaning after assistance for refractory cardiogenic shock. Intensive Care Med 2011;37:1738-45. [Crossref] [PubMed]
- Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium. Circulation 2011;123:2736-47. [Crossref] [PubMed]
- Kirkpatrick AW, Roberts DJ, De Waele J, et al. Intra-abdominal hypertension and the abdominal compartment syndrome: updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med 2013;39:1190-206. [Crossref] [PubMed]
- Kim YS, Cho YH, Sung K, et al. Target Temperature Management May Not Improve Clinical Outcomes of Extracorporeal Cardiopulmonary Resuscitation. J Intensive Care Med 2019;34:790-6. [Crossref] [PubMed]
- Chen YS, Lin JW, Yu HY, et al. Cardiopulmonary resuscitation with assisted extracorporeal life-support versus conventional cardiopulmonary resuscitation in adults with in-hospital cardiac arrest: an observational study and propensity analysis. Lancet 2008;372:554-61. [Crossref] [PubMed]
- Bednarczyk JM, White CW, Ducas RA, et al. Resuscitative extracorporeal membrane oxygenation for in hospital cardiac arrest: a Canadian observational experience. Resuscitation 2014;85:1713-9. [Crossref] [PubMed]
- Blumenstein J, Leick J, Liebetrau C, et al. Extracorporeal life support in cardiovascular patients with observed refractory in-hospital cardiac arrest is associated with favourable short and long-term outcomes: A propensity-matched analysis. Eur Heart J Acute Cardiovasc Care 2016;5:13-22. [Crossref] [PubMed]
- Maj G, Calabrò MG, Pieri M, et al. Abdominal compartment syndrome during extracorporeal membrane oxygenation. J Cardiothorac Vasc Anesth 2012;26:890-2. [Crossref] [PubMed]
- Glowka TR, Schewe JC, Muenster S, et al. Decompressive laparotomy for the treatment of the abdominal compartment syndrome during extracorporeal membrane oxygenation support. J Crit Care 2018;47:274-9. [Crossref] [PubMed]


