Thoracoscopic left upper lobectomy with systematic lymph nodes dissection under left pulmonary artery clamping
Introduction
A stage I or II lung cancer without or with mild lymph node metastasis can be easily operated using an anatomical resection or the single-direction approach. However, multiple hilar and mediastinal lymph nodes metastasis increases the difficulty of a VATS procedure. This is particularly the case, where the pulmonary artery or its major branches are suspected to be invaded by metastasized lymph nodes, which significantly increases the risk of arterial bleeding. Pulmonary artery clamping following an anatomical lobectomy can reduce the risk of an uncontrollable bleeding from a pulmonary artery, and allow the thoracoscope procedure to proceed. There is no doubt that, before the pulmonary artery clamping, it is critical to conduct a surgical exploration, removing the metastatic lymph nodes and fibrofatty tissues surrounding the artery to be clamped, and determine the feasibility of the clamping and the necessity of an open conversion. With a successful upstream and downstream control, we would be able to confidently manage the pulmonary artery or its major branches with suspected tumor invasion.
Summary of clinical case
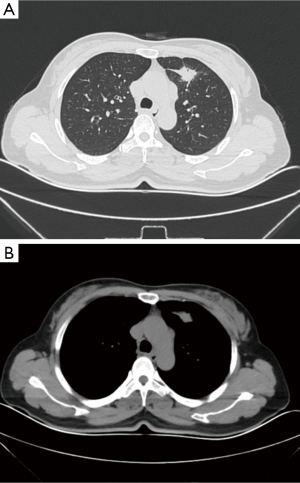
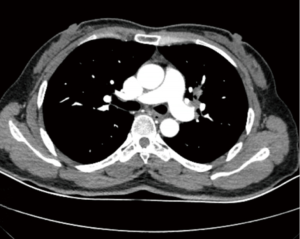
The patient was a 49-year-old woman, who was found to have a left upper lobe mass during a health check (Figure 1A,B). A chest CT with enhancement revealed multiple enlarged hilar and mediastinal lymph nodes. The nature of the mass was confirmed to be adenocarcinoma by transpercutaneous lung biopsy. There was no evidence of any distant metastasis or surgical contraindication judging from the preoperative workups. We decided to operate the patient using VATS left upper lobectomy with systematic lymph node dissection. The challenge of our preoperative patient evaluation came from the fact that, around the apical branch of the left upper pulmonary artery, there were enlarged lymph nodes with potential tumor metastasis (Figure 2), which would bring difficulty to the dissection of the apical segmental artery. Thoracoscopic pulmonary artery clamping might be needed during the procedure.
Surgical techniques
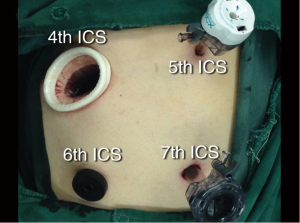
The patient was placed on her right lateral decubitus position. The operation was conducted under general anesthesia with double-lumen endotracheal intubation. The camera port (1 cm in diameter) was placed in the seventh intercostal space (ICS) on the left posterior axillary line. The utility port (3 cm) was placed in the fourth ICS on the left anterior axillary line. The first auxiliary port (1 cm in length) was placed in the sixth ICS on the left anterior axillary line. The second auxiliary port (0.5 cm) was placed in the fifth ICS below the subscapular angle (Figure 3).
Step 1: dissecting the station 5 lymph nodes
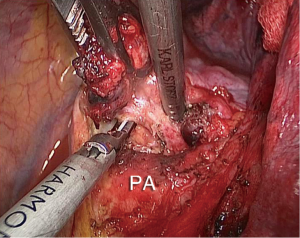
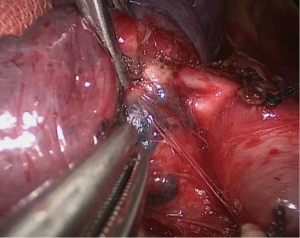
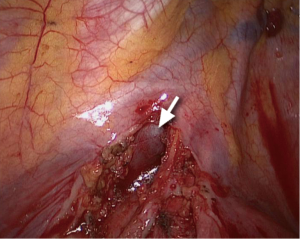
The mediastinal pleura on the surface of the left superior pulmonary vein was opened using a hook-shaped electrocautery. The station 5 lymph nodes were dissected, along the space superior to the left pulmonary vein, using dissecting forceps and a Harmonic scalpel (Figure 4). During the dissection process, attention and precaution were given to protect the phrenic nerve and recurrent laryngeal nerve. Attention was given to determine the relationship between the enlarged lymph nodes and the anterior wall of the left main pulmonary artery. In the event the enlarged lymph nodes tightly adhere to the left main pulmonary artery, subsequent thoracoscopic mobilization of the artery would carry a fairly high risk of an uncontrollable major bleeding, if this artery were injured. This procedure started with the dissection of the station 5 lymph nodes, with the objective to explore and assess whether the proximal left pulmonary artery could dissected and exposed for subsequent arterial clamping. The second objective was to assess whether the aortic arch had been invaded. It would be very difficult to clamp the left pulmonary artery, if its anterior wall could not be exposed by dissection.
Step 2: opening the posterior mediastinal pleura and exploring the tissues posterior to the hilum
The posterior mediastinal pleura were opened towards superiorly, to determine whether there were any significantly enlarged hilar or mediastinal lymph nodes, and whether major tissues behind the hilum were immobilized due to the invasion and adhesion by metastasized lymph nodes. In this procedure, the tissues anterior to the hilum were found to be fairly mobile, although the lymph nodes were enlarged.
Step 3: dissecting the left superior pulmonary vein
We returned to the area anterior to the hilum, where we mobilized the left superior pulmonary vein sufficiently using dissecting forceps. As we were mobilizing the posterior wall, the dissecting forceps were placed as much as closer to the left upper lobe bronchus, to avoid potential injury to the left superior pulmonary vein. Afterwards, a double 7-0 suture was placed across the vein for traction purposes.
Step 4: probing the proximal left main pulmonary artery
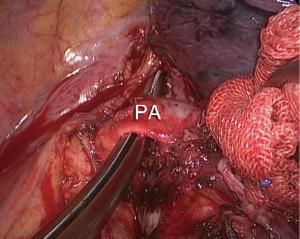
The superior pulmonary vein was pulled downwards. The sheath of the left main pulmonary artery was dissected bluntly using dissecting forceps, and it was opened using a Harmonic scalpel. The proximal trunk of the left main pulmonary artery was found to be fairly mobile, although posteriorly there were station 4 lymph nodes, which, however, did not invade the left pulmonary artery (Figure 5). Therefore, we were confident that the proximal trunk of the left main pulmonary artery could be released and mobilized.
Step 5: managing the fissure and mobilizing left upper lobe arterial branches
After confirming that the proximal pulmonary artery trunk could be mobilized, we proceed to manage the oblique fissure. The objective was to know whether there were enlarged lymph nodes surrounding the trunk of the left lower lobe pulmonary artery, which might preventing us from freeing the artery. In addition, the left upper lobe lingual segmental artery and the posterior segmental artery could be managed, after the oblique fissure was separated. The apical artery with suspected tumor invasion would be divided, after the left pulmonary artery clamping. In this patient, the fissure was well developed, and we open the fissure using an electrocautery and a Harmonic scalpel. The lymph nodes in front of the lingual artery were also dissected, and the lingual artery and posterior segmental artery were subsequently divided in an anterior-to-posterior direction. While dissecting the apical artery, we found stiff lymph nodes adhering to the apical artery (Figure 6), and we therefore chose to mobilize this artery after the clamping the left main pulmonary artery and left lower lobe pulmonary artery.
Step 6: dividing the left upper pulmonary vein
Following the surgical exploration, we confirmed that the left upper lobe and its tumor tissues could be completely resected under left pulmonary artery clamping. We divided the left superior pulmonary vein using an Ethicon 45 linear stapler with a white cartridge. The anterior wall of the left main pulmonary artery could now be accessed without any barrier.
Step 7: complete mobilization of the proximal left main pulmonary and the dissection of station 4 lymph nodes
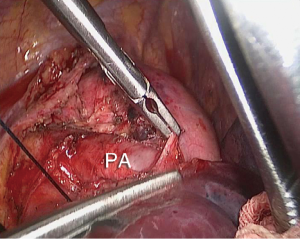
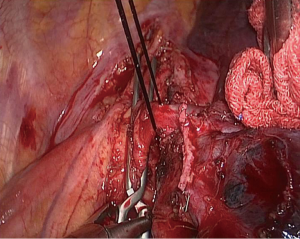
The sheath of the proximal trunk of the left main pulmonary artery was further opened using dissecting forceps and a Harmonic scalpel. The space between this artery and the left upper lobe bronchus was further dissected and expanded. The left upper lobe was pulled anteriorly, exposing the posterior wall of the left main pulmonary artery. The station 4 lymph nodes were removed, and the posterior wall of the left main pulmonary artery was able to be fully mobilized. A pair of ring forceps was then placed across this artery to lay down the traction suture (Figure 7), finishing the preparation for the proximal clamping of the left main pulmonary artery.
Step 8: mobilizing the left main lower pulmonary artery
The left lower lobe artery was dissected bluntly, and a double 7-0 traction suture was placed across the artery, to facilitate subsequent vascular clamping.
Step 9: simultaneous clamping of the proximal left main pulmonary artery and the main left inferior pulmonary artery
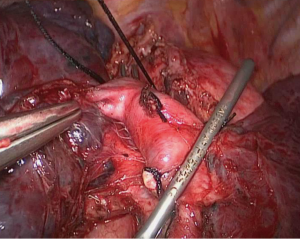
The left pulmonary artery (Figure 8) and left inferior pulmonary artery (Figure 9) were clamped using an endoscopic bulldog clamp.
Step 10: blunt mobilization of the apical artery of the left upper lobe
The left pulmonary artery and left lower lobe pulmonary artery were clamped, and the apical artery with possible tumor invasion was then dissected bluntly. While there were enlarged lymph nodes surrounding the apical artery; however, the lymph nodes did not fully cover the artery. Instead, there was a short free space, which allowed us to divide the artery using an Ethicon 45 linear stapler loaded with a white cartridge. All high risk steps of this procedure were now completed.
Step 11: transection of the left upper lobe bronchus
The left upper lobe bronchus was mobilized from its surrounding tissues, and was then divided using an Ethicon 45 linear stapler loaded with a green cartridge. The left upper lobe specimen was subsequently retrieved through the utility port.
Step 12: further mediastinal lymph nodes dissection
After the left upper lobe resection, we proceeded with the dissection of station 6 and 7 lymph nodes. We followed the principle of regional lymph nodes dissection. By definition, station 7 lymph nodes refer to those below the trachea bifurcation, above the lower pulmonary vein, posterior to the pericardium, and anterior to the esophageal wall. All lymph nodes and fat tissues within this range were resected in an en bloc manner. The inferior wall of the left innominate vein must be revealed, following a successful dissection of the station 6 lymph nodes and associated fatty tissues (Figure 10).
Comments
In thoracoscopic left upper lobectomy under left main pulmonary artery clamping, we first need to dissect the tissue anterosuperior to the left hilum, and explore the anterior wall of the left main pulmonary artery. If station 5 and 10 lymph nodes are enlarged due to metastasis, these lymph nodes need to be dissected. If these lymph nodes show significant extranodal invasion and appear to be immobilized, managing these lymph nodes thoracoscopically carries an extremely high risk of a major bleeding, and open conversion would be a better option. As demonstrated in Figure 11, the station 5 lymph nodes with metastasis were dissected with caution, and the anterior wall of the left main pulmonary artery was revealed. Afterwards, we shifted our focus to the posterior wall of the left main pulmonary artery, where station 4 lymph nodes were located. The removal of these nodes allowed us to dissect within the space between the left pulmonary artery and left main bronchus, towards the anterior wall of the left main pulmonary artery. An endoscopic bulldog clamp was then placed on the left mainly pulmonary artery, in an anterior-to-posterior direction. Because the artery that we wish to manage was a branch of the left upper lobe pulmonary artery, simply clamping proximal left main pulmonary artery (upstream) would not be sufficient. The inferior pulmonary artery (downstream) would have to clamp as well, so as to prevent blood backflow. This is what we call “inflow-outlfow bidirectional occlusion”, which enabled us to mobilize the apical branch of the left upper lobe with confidence. We were pleased to find that this arterial branch was not fully covered by metastasized lymph nodes. This gave us the space to manipulate an Ethicon stapler, and successfully complete the high risk steps of this left upper lobectomy.

Another challenge that we faced was how to manage the arterial branches with suspected tumor invasion. Our practice is that this branch shall be mobilized first, because transection after mobilization is the safest and most reliable approach. In the event this branch is circumferentially covered by tumor or metastatic lymph nodes, or if the main trunk at the bifurcation is also invaded, the only option would be left pulmonary artery wall resection or sleeve resection. However, this approach is very challenging, and is better completed by a thoracic surgeon with extensive experiences. Another safe option is open conversion.
Thoracoscopic pulmonary artery clamping is not a routine surgery. It demands a surgeon with extensive thoracoscopic surgery experiences and accurate and objective intra-operative judgment. What we advocate is a safe and effective surgery, not a minimally invasive procedure for the sake of minimally invasiveness.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Dong YN, Sun N, Ren Y, et al. Thoracoscopic left upper lobectomy with systematic lymph nodes dissection under left pulmonary artery clamping. Asvide 2014;1:373. Available online: http://www.asvide.com/articles/407