Left lower sleeve lobectomy and systematic lymph node dissection by complete video-assisted thoracic surgery
Introduction
In 1947, Price Thomas pioneered a circumferential resection of the right bronchus in a patient with bronchial adenoma (1). A decade later, in 1956, Allison reported the first case of non-small-cell lung carcinoma (NSCLC) treated by sleeve lobectomy (2). Sleeve lobectomy adequately preserves the lung function and achieves complete resection of the neoplasm, recently developed in selected cases of lung cancer patients. Various studies have shown that the long-term survival after sleeve resection is similar or sometimes better than pneumonectomy, but results in the lower perioperative risk and better quality of life (3-6).
Video-assisted thoracic surgery (VATS) lobectomy, first reported in 1992 (7), had developed an established approach for lung cancer management after 20 years of technique improvement. Due to the surgical difficultly, VATS sleeve lobectomy used to be a contraindication in lung cancer surgical treatment (8). Nowadays with improved technology and increased experiences in VATS lobectomy, thoracic surgeons have gradually mastered the techniques of sleeve lobectomy by VATS. Left lower lobectomy with sleeve bronchial resection is one of the most difficult thoracic surgical procedures (9), and approach to left bronchial anastomosis is also limited and unclear. Here we present a technical procedure for left lower sleeve lobectomy by complete VATS.
Case presentation
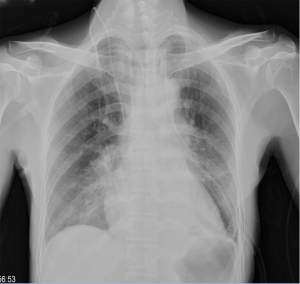
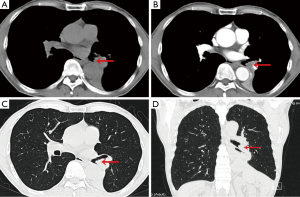
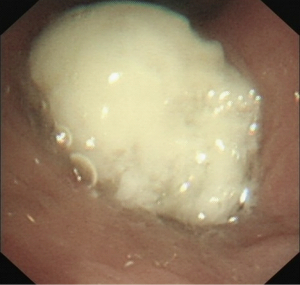
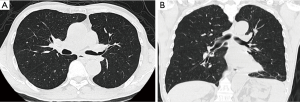
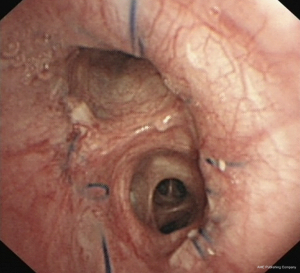
A 64-year-old male was presented with cough for 2 months. He had a history of subtotal gastrectomy, cholecystectomy and hypertension. He had smoked about 20 cigarettes per day for 40 years. A preoperative chest computed tomography (CT) revealed a mass in the left hilum of the lung, with complete left low pulmonary atelectasis (Figure 1), and fiberoptic bronchoscopy showed a neoplasm at the orifice of the left lower lobar bronchus (Figure 2). Preoperative pathological examination of the lung lesion indicated a poorly differentiated squamous cell carcinoma. Left lower Sleeve lobectomy was performed. The operation takes 200 minutes. During the operation, the estimated blood loss was 100 mL. On postoperative day (POD) 1, the amount of fluid drainage was 350 mL, and chest X-ray showed left lung inflation satisfactory (Figure 3). The chest tube was removed on POD 4. The patient was discharged on POD 6 with no complications. The tumor size was 3 cm in diameter, and the pathology confirmed lymph metastasis in station 9 while the other lymph stations were negative pathological results. Three months later, a chest CT showed no obvious locoregional recurrence (Figure 4). Six months later, fiberoptic bronchoscopy showed the intact anastomosis site and no sign of stenosis (Figure 5).
Operative techniques
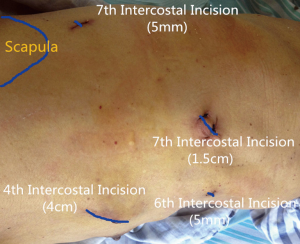
- Operative insicion: four insicions were performed (Figure 6, the initial camera port (1.5-cm-long incision) was made in the 7th intercostal space in the midaxillary line, the utility incision (4-cm-long incision) was placed at the level of 4th intercostal space in anterior axillary line. Another two 0.5-cm-long additional insicions were performed in the 7th intercostal space in the posterior axillary and in the 6th intercostal space in the anterior axillary line respectively. A 0.5 cm incisional length may reduce the impairment and compression of intercostal nerves and vessels, and increase the conveniency of operation. No rib spreader was used;
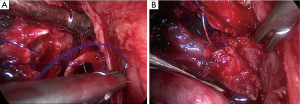
- Operative key points and procedure (Figure 7).
- After division of the inferior pulmonary ligament the lymph nodes from stations 8 and 9 were excised. The dissection continued by division of the mediastinal pleura around the pulmonary hilum;
- Mobilization of pulmonary vessels. The pulmonary artery was identified and dissected at the fissure. The oblique fissure was dissected by ultrasonic scalpel. The pulmonary artery sheath was dissected by electrocautery hook, and the lower lobe branches of pulmonary artery were mobilized. The basal and dorsal segmental artery was stapled with a vascular (white cartridge, 45-mm-long) endostapler (Endo GIA, Covidien, USA). The fissure was completed by a stapler firing. The lymph nodes of station 11 was then dissected;
- Dissection of mediastinal lymph nodes. The ring forceps was applied to pull the left lower lung forwards to expose the posterior mediastinum. We dissected along the posterior aspect of subcarinal lymph nodes (station 7), and the small vessels between esophagus and lymph nodes were cauterized with electrocautery hook to reduce bleeding. The lymph nodes of station 7 were completely removed. The station 4 lymph nodes were dissected using a combination of blunt and sharp dissection technique, with special care to expose and to preserve the left recurrent laryngeal nerve. The stations 5 and 6 lymph nodes were dissected with avoidance of the phrenic nerve and the left innominate vein damage;
- Bronchial anastomosis. The secondary carina and left main bronchus were completely dissected. The left main bronchus and upper bronchus was transected through the utility incision. The lobe was placed in a specimen bag and was then removed from the thoracic cavity. The specimen was sent for pathological frozen section, and bronchial margins were confirmed negative. A 4-0 prolene was used in continuous suture for anastomosis from posterior to anterior. The knots were made outside the bronchial lumen (Figure 8). When anastomosis proceeding, needle holder was placed through a 5 mm incision at the 6th intercostal space in the anterior axillary line or the 7th intercostal space in the posterior axillary;
- Check the anastomosis. The bronchus anastomosis was tested with lavage with distilled water, and the lung was inflated to rule out the air leak.

Comments
Sleeve lobectomy was an approach option only for thoracotomy before because of its difficulty. Patients undergoing traditional thoracotomy suffered problems like slow recovery, serious trauma, and postoperative pain. However, comparing to thoracotomy, VATS lobectomy has various potential advantages like less postoperative pain, faster recover, fewer complications, and shorter hospital stay (11). Thanks to the rapid improvement and development of thoracoscopic techniques in recent decades, VATS sleeve lobectomy gradually became a feasible, attractive and challenging procedure for selected patients.
Bronchial anastomosis in VATS sleeve lobectomy used to be difficult. The key points of this procedure are removal of subcarinal lymph nodes (station 7) and station 11 lymph nodes, and completely dissection of the secondary carina and left main bronchus. Continuous or interrupted suture can be applied to anastomosis after bronchus transection. According to our experience, the method of continuous suture takes less time. The “difficult-to-easy” principle should be followed while placing sutures from posterior to anterior along the membranous portion with 4-0 prolene. Meanwhile, suturing order should be properly arranged to prevent twisting. Single continues suture could reduce the probability of twining (9).
Proper incision placement is critical in bronchial anastomosis of VATS sleeve lobectomy. We chose three routine incisions for the most of VATS lobectomies. We also added a port in the left lower sleeve lobectomy. During bronchial anastomosis procedure, the needle holder was put through the 5-mm-long incision at the 6th intercostal space in the anterior axillary line and the 7th intercostal space in the posterior axillary. A 0.5 cm of incisional length may reduce the impairment of chest wall and the adjacent tissues, especially the intercostal nerves, and could increase the conveniency of operation (12).
VATS sleeve lobectomy is feasible and safe. However, VATS sleeve lobectomy, like all other procedures, has its learning curve for surgeons and assistants. According to our own experience, we suggest that a surgeon needs to perform at least 100 cases of VATS lobectomy and 20 cases of open sleeve resection procedures before beginning to perform VATS sleeve lobectomy. In addition, the cases for VATS sleeve lobectomy should be highly selected, limited to a few conditions especially like small central tumors.
In conclusion, VATS sleeve lobectomy should be an ideal option in patients with specific surgical indications. Development of VATS techniques may gradually make VATS sleeve lobectomy a more common procedure in thoracic surgery.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Thomas CP. Conservative resection of the bronchial tree. J R Coll Surg Edinb 1956;1:169-86. [PubMed]
- Allison PR. Course of thoracic surgery in Groningen. Ann R Coll Surg 1954;25:20-22.
- Okada M, Yamagishi H, Satake S, et al. Survival related to lymph node involvement in lung cancer after sleeve lobectomy compared with pneumonectomy. J Thorac Cardiovasc Surg 2000;119:814-9. [PubMed]
- Terzi A, Lonardoni A, Falezza G, et al. Sleeve lobectomy for non-small cell lung cancer and carcinoids: results in 160 cases. Eur J Cardiothorac Surg 2002;21:888-93. [PubMed]
- Merritt RE, Mathisen DJ, Wain JC, et al. Long-term results of sleeve lobectomy in the management of non-small cell lung carcinoma and low-grade neoplasms. Ann Thorac Surg 2009;88:1574-81; discussion 1581-2. [PubMed]
- Ma Z, Dong A, Fan J, et al. Does sleeve lobectomy concomitant with or without pulmonary artery reconstruction (double sleeve) have favorable results for non-small cell lung cancer compared with pneumonectomy? A meta-analysis. Eur J Cardiothorac Surg 2007;32:20-8. [PubMed]
- Roviaro G, Rebuffat C, Varoli F, et al. Videoendoscopic pulmonary lobectomy for cancer. Surg Laparosc Endosc 1992;2:244-7. [PubMed]
- McKenna RJ Jr, Houck W, Fuller CB. Video-assisted thoracic surgery lobectomy: experience with 1,100 cases. Ann Thorac Surg 2006;81:421-5; discussion 425-6. [PubMed]
- Gonzalez-Rivas D, Delgadoa M, Fieira E. Left lower sleeve lobectomy by uniportal video-assisted thoracoscopic approach. Interact. CardioVasc Thorac Surg 2014;18:237-9. [PubMed]
- Fan JQ, Yao J, Chang ZB, et al. Video-assisted thoracic surgery, left lower sleeve lobectomy. Asvide 2014;1:364. Available online: http://www.asvide.com/articles/398.
- Shaw JP, Dembitzer FR, Wisnivesky JP, et al. Video-assisted thoracoscopic lobectomy: state of the art and future directions. Ann Thorac Surg 2008;85:S705-9. [PubMed]
- Fan J, Chang Z, Ye C, et al. Video-assisted thoracoscopic superior segmentectomy of the right lower lobe. J Thorac Dis 2013;5:S287-8. [PubMed]