
Fixed in the neck or pushed back into the thorax?—Impact of cervical anastomosis position on anastomosis healing
Introduction
Esophageal cancer is the eighth most common malignant tumor and the sixth leading cause of cancer-relating death worldwide (1). At present, esophagectomy with lymphadenectomy remains the cornerstone in patients with resectable disease.
Nonetheless, the advances of surgical technique and postoperative management, the incidence of anastomotic leakage remains high. As reported, the leakage rate varied from 4.0% to 20.5% in patients who received intrathoracic anastomosis, and it was 12.3% to 31.0% in the cervical anastomosis (2-5)
The anastomotic leakage is a troublesome complication, and it could become even more challenging when the leakage happens within the thoracic cavity. We could buy precious time for the healing of cervical leakage through the opening and draining of the cervical wound (6,7). But it becomes quite difficult to guarantee adequate drainage when it comes to the intrathoracic leakage. So the patients often die of severe infection before the healing of the intrathoracic leakage. Thus, more and more surgeons prefer choosing the minimally invasive McKeown approach with cervical anastomosis. However, the incidence of cervical anastomotic leak is significantly higher than that of intrathoracic anastomosis (2-5). Among the factors affecting the healing of cervical anastomosis, the surgical technique is one of the critical factors under the control of surgeons. We could make the gastroesophageal anastomosis by hand-sewn or by a stapler. When we finish the anastomosis, we could push it back into the thorax or fix it in the neck. These controllable factors are quite important in handling this unpredictable complication. The right choice could help both surgeons and our patients a lot. Therefore, this study aimed to identify the independent risk factors affecting cervical anastomotic healing, especially those about surgical techniques.
Methods
Patients
A retrospective review of patients who underwent esophagectomy with cervical anastomosis between January 2010 and April 2018 in West China Hospital was performed. The including criteria were: (I) patients with pathologic confirmed esophageal squamous cell carcinoma or adenocarcinoma. (II) Patients underwent McKeown approach with cervical anastomosis. (III) The anastomosis was made between the end of the proximal esophagus and the lateral wall of the gastric conduit. The exclusion criteria were: (I) patients received neoadjuvant therapy. (II) R1 resection. (III) The tumor originated from the esophagogastric junction. This study was approved by the Ethics Committee of West China Hospital, Sichuan University (No. 20180419). Since this was a retrospective cohort analysis and analyzed anonymously, the ethics committee waived the need for informed consents from those patients
Baseline data collection
The baseline characteristics of the included patients were obtained from the electronic medical records in West China Hospital. It included age, gender, preoperative comorbidities, and postoperative pathology report. The disease was staged according to the eighth edition (2017) of the American Joint Committee on Cancer staging system (8). The surgical technique, including surgical approach, anastomosis mode, and if cervical anastomosis fixation was performed, were collected from the surgery records.
Details of the cervical anastomosis
The gastric conduit was created by tailoring the entire stomach into a 3–5 cm-wide tubularized conduit. A cervical incision was made along the anterior border of the left sternocleidomastoid muscle. After the mobilization of the cervical esophagus, the conduit was pulled up to the neck through the posterior mediastinal route. Then, the anastomosis was performed either by layered hand-sewn anastomosis or circular-stapler anastomosis. The details of the two anastomosis technique have been reported previously (9). No specific indication was designed when chose the anastomosis technique. After that, the anastomosis was either fixed in the neck (by sewing to the neighboring cervical muscle) or pushed back into the thoracic cavity, depending on the surgeons’ preference.
The judgment of anastomotic leakage
For patients without any sign of anastomotic leakage, the barium esophagogram is routinely performed before the start of oral intake. For patients who have abnormal clinical manifestations, which highly indicated the appearance of anastomotic leakage, the computed tomography or barium esophagogram would be performed firstly. If these two examinations highly indicated an anastomotic leakage, the endoscopy would be performed to see the condition of the anastomosis directly. Based on these examinations, the judgment of if an anastomotic leakage happened was made.
Statistical analysis
The χ2 test or Fisher’s exact test was used for the qualitative data analysis. Student’s t-test or Mann-Whitney U test was applied to compare the continuously distributed data. Variables with a P value of less than 0.05 in the univariate logistic regression analysis were enrolled in the multivariable logistic regression analysis to identify the risk factors of cervical anastomotic leakage. P values of less than 0.05 were considered statistically significant. All statistical analyses were performed with the SPSS 23.0 (IBM, Armonk, NY, USA).
Results
Finally, a total of 518 patients were included in this study. Among them, fifty-one patients (9.85%) with endoscopy confirmed anastomosis leakage was assigned to the leakage group while the left 467 patients were enrolled in the non-leakage group. Among the fifty-one patients who developed anastomotic leakage, six patients died (11.76%).
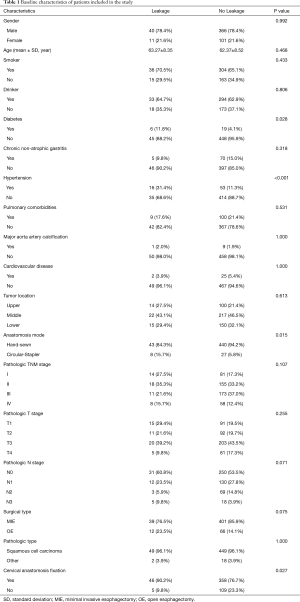
The baseline characteristics of patients enrolled in the study were shown in Table 1. No statistically significant difference was detected between the leakage group (LG) and non-leakage group (NLG) in gender, age, tumor location, histologic type, surgery approach, T stage, N stage, and pathologic TNM stage. Among the preoperative comorbidities, only the prevalence of diabetes and hypertension showed a statistically significant difference between the two groups. As for surgical techniques, the patients who underwent the procedure of cervical anastomosis fixation had a higher rate of anastomotic leakage than the patients with the anastomosis pushed back into the thoracic cavity (11.4% versus 4.4%, P=0.027). The use of circular stapler also had a higher leakage rate than that of hand-sewn anastomoses (22.9% versus 8.9%, P=0.015).
Full table
Multivariate logistic regression analysis of potential risk factors for anastomotic leakage
A multivariate analysis was performed to identify the independent risk factors of cervical anastomotic leakage for patients who underwent McKeown esophagectomy. Factors with a P value of less than 0.05 in the baseline comparison (Table 1) were included in the analysis. The results of the multivariate analysis were shown in Table 2. The statistical difference of hypertension, anastomosis mode, and cervical fixation remained statistically significant in the multivariate logistic regression analysis. Patients receiving cervical anastomosis fixation suffered from a higher risk of postoperative leakage (OR =2.922, 95% CI: 1.114, 7.660, P=0.029). And the patients who underwent stapled anastomosis were also more likely to develop postoperative leakage (OR =2.867, 95% CI: 1.182, 6.952, P=0.020). Patients with a history of hypertension also had a higher risk of postoperative leakage (OR =3.392, 95% CI: 1.734–6.634, P<0.001).

Full table
Discussion
The anastomotic leakage after the esophagectomy dramatically increases the mortality. A systematic review showed the pooled mortality after cervical anastomosis was 14% (10). The mortality in the patients suffered from leakage was 11.76% in our study. What’s more, the anastomotic leakage also had a negative impact on long-term outcomes (11-13). The study conducted by Tanaka et al. showed that anastomotic leakage was a risk factor for the development of anastomotic stricture (11). Fransen et al. reported the patients who suffered from anastomotic leakage would have a poorer long-term survival (12). The healing of esophagogastric anastomoses is affected by many factors. Among all these factors, the adequate blood supply to the tubularized conduit counts most. The blood supply of the gastric conduit mostly comes from the right gastroepiploic vessels with negligible contributions from the right gastric artery and vein (14). The risk of anastomotic leakage increases when insufficient arterial blood flow or venous congestion in the tubularized conduit occurs. Several techniques have been designed to improve the blood perfusion of the conduit (15-18). Nagawa et al. reported the additional anastomosis between gastric vessels and cervical vessels was effective in reducing the risk of anastomotic leakage (16). Kechagias et al. reported the ischemic conditioning before esophagectomy could reduce the incidence of anastomotic leakage as well (17). However, these techniques are complicated and not suitable for every patient. So they are not widely used now.
In the McKeown esophagectomy, the cervical anastomosis is compressed by the rigid spine, sternum, and trachea at the level of the thoracic inlet (7,15,19), leading to poor perfusion to the tubularized conduit. Kunisaki and his colleagues reported by the resection of the head of the left clavicle and manubrium could release the compression, and therefore improve the blood perfusion to the conduit (15). However, it unavoidably increases the surgical trauma.
The gastric vessels do not need to pass through the narrow thoracic inlet any more when we push the anastomosis back into the thorax. Therefore, pushing the anastomosis back into the thorax can improve both the venous blood flow return and the arterial blood perfusion theoretically. Our statistical analyses demonstrated that patients with anastomosis pushed back into the right thorax had a considerably lower incidence of leakage than those with the cervical anastomosis fixed in the neck (4.4% vs. 11.4%, P=0.027). What is more, the multivariate analysis also confirmed that the fixation procedure is an independent risk factor in the development of anastomotic leakage.
In accordance with the previous publication, hypertension also was identified as an independent risk factor of anastomotic leakage (20). Hypertension may decrease the stability of blood supply and leads to insufficient blood flow in the anastomotic region. So the healing ability declines in patients with hypertension.
The leakage group had a higher prevalence of diabetes in the baseline characteristics comparison, but the statistical difference did not remain significant in the multivariate logistic regression analysis. Diabetes has a passive impact on microvascular circulation (21). Furthermore, the high-glucose state can impair the healing of the wound (22,23). However, whether diabetes increases the risk of anastomotic leakage remains controversial. A meta-analysis shows diabetes is significantly associated with a higher risk of anastomotic leakage (23). In contrast, a prospectively randomized study shows that diabetes does not increase the leakage rate of cervical anastomosis (24). Our result is consistent with the latter. It did not support diabetes to be a risk factor of cervical anastomotic leakage.
There are two major technical methods to accomplish the anastomosis, the hand-sewn anastomosis and the mechanical anastomosis using a circular stapler. The two anastomotic models have their specialties. Which anastomotic model is better in the control of postoperative anastomotic leakage remains controversial (25-27). Our multivariate logistic regression analysis indicated the circular stapler anastomosis as an independent risk factor of anastomotic leakage. Here are the three reasons that may explain our findings. Firstly, the sizes of staplers include 21, 25, 29, and 33 mm in diameter. The stapler of 21 mm in diameter is too small, which often leads to anastomotic stenosis after esophageal anastomosis. Moreover, the stapler of 29 and 33 mm in diameter are too large to put into the esophagus for Chinese patients. Therefore, the size of the stapler used in our study is fixed at 25 mm in diameter. So the circular stapler is not always consistent with the diameter of the esophagus, thereby leading to poor tissue apposition. Secondly, most surgeons use their fingers to check the tissue apposition of the anastomosis after creating anastomosis with a stapler. This method is not as accurate as direct-eye assessment in the mode of the hand-sewn anastomosis. Thirdly, the limited experience of applying the circular stapler could be the technological aspect of why it had a higher risk of leakage. After all, only 35 circular-stapler anastomoses were performed. Lastly, hand-sewn anastomosis is conducted in a layer to layer manner. Thus, the hand-sewn anastomosis may have a much better reconstruction of the digestive tract.
Due to the retrospective nature of our study, it had several limitations. The overall rate of neoadjuvant therapy is low in China. So did our patients. Our study enrolled patients who underwent esophagectomy between January 2010 and April 2018. The efficacy of neoadjuvant therapy was uncertain at that time. Most of our patients were from rural areas, and they believed the surgery was the best treatment. They were afraid of the development of the disease during neoadjuvant therapy and lost the chance of operation. So they were unwilling to receive neoadjuvant therapy. These may be the reasons why the patient with locally advanced disease didn’t receive neoadjuvant therapy in this study. Although the study enrolled 518 patients, the sample size of the leakage group was only fifty-one. Prospective studies with larger sample sizes are needed to test our results further.
In conclusion, this study identified the cervical fixation as a novelly identified risk factor affecting cervical anastomosis healing for the first time. The procedure fixing the anastomosis in the neck increases the incidence of postoperative leakage. Therefore, the fixation of the anastomosis in the neck after the cervical anastomosis is unnecessary. Conversely, pushing the anastomosis back into the right thoracic cavity improves the venous blood flow return and the arterial blood perfusion of the gastric conduit. It has a statistically significant lower rate of anastomotic leakage than fixation of the anastomosis in the neck.
Acknowledgments
We would like to thank Mr. Shi-De Wu from the High School Attached to Northeast Normal University for his linguistic assistance to this manuscript.
Funding: This work was supported by the National Natural Science Foundation of China (grant number: 81672291, 31071210 to YD Lin).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd.2020.04.36). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was approved by the institutional review board of West China Hospital, Sichuan University (No. 20180419).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Pennathur A, Gibson MK, Jobe BA, et al. Oesophageal carcinoma. Lancet 2013;381:400-12. [Crossref] [PubMed]
- Kassis ES, Kosinski AS, Ross P Jr, et al. Predictors of anastomotic leak after esophagectomy: an analysis of the society of thoracic surgeons general thoracic database. Ann Thorac Surg 2013;96:1919-26. [Crossref] [PubMed]
- Klink CD, Binnebosel M, Otto J, et al. Intrathoracic versus cervical anastomosis after resection of esophageal cancer: a matched pair analysis of 72 patients in a single center study. World J Surg Oncol 2012;10:159. [Crossref] [PubMed]
- Chasseray VM, Kiroff GK, Buard JL, et al. Cervical or thoracic anastomosis for esophagectomy for carcinoma. Surg Gynecol Obstet 1989;169:55-62. [PubMed]
- van Workum F, van der Maas J, van den Wildenberg FJ, et al. Improved Functional Results After Minimally Invasive Esophagectomy: Intrathoracic Versus Cervical Anastomosis. Ann Thorac Surg 2017;103:267-73. [Crossref] [PubMed]
- Urschel JD. Esophagogastrostomy anastomotic leaks complicating esophagectomy: a review. Am J Surg 1995;169:634-40. [Crossref] [PubMed]
- Jones CE, Watson TJ. Anastomotic Leakage Following Esophagectomy. Thorac Surg Clin 2015;25:449-59. [Crossref] [PubMed]
- Rice TW, Ishwaran H, Ferguson MK et al. Cancer of the Esophagus and Esophagogastric Junction: An Eighth Edition Staging Primer. J Thorac Oncol 2017;12:36-42.
- Okuyama M, Motoyama S, Suzuki H, et al. Hand-sewn cervical anastomosis versus stapled intrathoracic anastomosis after esophagectomy for middle or lower thoracic esophageal cancer: a prospective randomized controlled study. Surg Today 2007;37:947-52. [Crossref] [PubMed]
- Verstegen MHP, Bouwense SAW, van Workum F, et al. Management of intrathoracic and cervical anastomotic leakage after esophagectomy for esophageal cancer: a systematic review. World J Emerg Surg 2019;14:17. [Crossref] [PubMed]
- Tanaka K, Makino T, Yamasaki M, et al. An analysis of the risk factors of anastomotic stricture after esophagectomy. Surg Today 2018;48:449-54. [Crossref] [PubMed]
- Fransen LFC, Berkelmans GHK, Asti E, et al. The Effect of Postoperative Complications After Minimally Invasive Esophagectomy on Long-term Survival: An International Multicenter Cohort Study. Ann Surg 2020. [Epub ahead of print]. [Crossref] [PubMed]
- Goense L, Meziani J, Ruurda JP, et al. Impact of postoperative complications on outcomes after oesophagectomy for cancer. Br J Surg 2019;106:111-19. [Crossref] [PubMed]
- Liebermann-Meffert DM, Meier R, Siewert JR. Vascular anatomy of the gastric tube used for esophageal reconstruction. Ann Thorac Surg 1992;54:1110-5. [Crossref] [PubMed]
- Kunisaki C, Makino H, Akiyama H, et al. Predictive factors for anastomotic leakage in the neck after retrosternal reconstruction for esophageal cancer. Hepatogastroenterology 2008;55:98-102. [PubMed]
- Nagawa H, Seto Y, Nakatsuka T, et al. Microvascular anastomosis for additional blood flow in reconstruction after intrathoracic esophageal carcinoma surgery. Am J Surg 1997;173:131-3. [Crossref] [PubMed]
- Kechagias A, van Rossum PSN, Ruurda JP, et al. Ischemic Conditioning of the Stomach in the Prevention of Esophagogastric Anastomotic Leakage After Esophagectomy. Ann Thorac Surg 2016;101:1614-23. [Crossref] [PubMed]
- Fujioka M, Hayashida K, Fukui K, et al. Venous superdrained gastric tube pull-up procedure for hypopharyngeal and cervical esophageal reconstruction reduces postoperative anastomotic leakage and stricture. Dis Esophagus 2017;30:1-6. [Crossref] [PubMed]
- Mine S, Watanabe M, Okamura A, et al. Superior Thoracic Aperture Size is Significantly Associated with Cervical Anastomotic Leakage After Esophagectomy. World J Surg 2017;41:2598-604. [Crossref] [PubMed]
- Aminian A, Panahi N, Mirsharifi R, et al. Predictors and outcome of cervical anastomotic leakage after esophageal cancer surgery. J Cancer Res Ther 2011;7:448-53. [Crossref] [PubMed]
- Barrett EJ, Liu Z, Khamaisi M, et al. Diabetic Microvascular Disease: An Endocrine Society Scientific Statement. J Clin Endocrinol Metab 2017;102:4343-410. [Crossref] [PubMed]
- Irons RF, Cahill KW, Rattigan DA, et al. Acceleration of diabetic wound healing with adipose-derived stem cells, endothelial-differentiated stem cells, and topical conditioned medium therapy in a swine model. J Vasc Surg 2018;68:115S-25S. [Crossref] [PubMed]
- Li SJ, Wang ZQ, Li YJ, et al. Diabetes mellitus and risk of anastomotic leakage after esophagectomy: a systematic review and meta-analysis. Dis Esophagus 2017;30:1-12. [Crossref] [PubMed]
- Gupta NM, Gupta R, Rao MS, et al. Minimizing cervical esophageal anastomotic complications by a modified technique. Am J Surg 2001;181:534-9. [Crossref] [PubMed]
- Laterza E. Manual compared with mechanical cervical oesophagogastric anastomosis: a randomised trial. Eur J Surg 1999;165:1051-4. [Crossref] [PubMed]
- Hsu HH, Chen JS, Huang PM, et al. Comparison of manual and mechanical cervical esophagogastric anastomosis after esophageal resection for squamous cell carcinoma: a prospective randomized controlled trial. Eur J Cardiothorac Surg 2004;25:1097-101. [Crossref] [PubMed]
- Aquino JL, Camargo JG, Said MM, et al. Cervical esophagogastric anastomosis evaluation with a mechanical device versus manual suture in patients with advanced megaesophagus. Rev Col Bras Cir 2009;36:19-23. [Crossref] [PubMed]


