
Blood lead levels in relation to smoking and chronic obstructive pulmonary disease (COPD): a study from Korean National Health and Nutrition Examination Survey (KNHANES)
Introduction
Although lead concentrations in the general population have declined over the past two decades, lead exposure is a resurgent environmental issue globally (1-3).
Chronic lead exposure has a wide spectrum of multi-systemic adverse effects through various biochemical mechanisms (4). Adult lead toxicity is defined as blood lead level ≥10 µg/dL; however low-level environmental lead exposure (blood lead level lower than 10 µg/dL) may accelerate progressive renal insufficiency and can be an important risk factor for cardiovascular mortality in US adults (5,6), It is recommended that pregnant women should avoid occupational lead exposure that would result in blood lead concentrations >5 µg/dL due to the possible consequences of spontaneous abortion and postnatal developmental delay (4).
The association between blood lead levels and increased all-cause and organ-specific mortality was reported on a worldwide level (3,7). One report showed that blood lead levels were linked to increased all-cause and cardiovascular mortality at substantially lower levels (below 10 µg/dL) than previously reported (7).
Lead as a pollutant originating from traffic-related and industrial emissions contributes to air pollution (8). Although a majority of elevated blood lead levels in adults originate from workplace exposures, smoking can be a source of lead exposure (9,10). Smoking is a major causative risk factor for chronic obstructive pulmonary disease (COPD); nevertheless, outdoor air pollution is considered a significant factor for the development of COPD (11,12). However, no study has been conducted so far concerning the blood lead levels based on risk factors of COPD and the smoking status in patients with COPD.
Therefore, this study investigated the association between elevated blood levels and clinical variables and attempted to examine the blood lead levels based on COPD and clinical variables associated with COPD, such as smoking status and socioeconomic factors.
Methods
Study design
Our data (total number =53,829) were collected from participants in the Korean National Health and Nutrition Examination Survey (KNHANES). Six cycles of KNHANES data, IV (n=9,744 in 2008, n=10,533 in 2009), V (n=8,958 in 2010, n=8,518 in 2011, n=8,058 in 2012), and VI (n=8,018 in 2013) were selected to represent South Korea population. KNHANES is a nationally representative cross-sectional complex survey including approximately 10,000 individuals each year, collecting information on socioeconomic status, health-related behaviors, quality of life, healthcare utilization, anthropometric measures, biochemical and clinical profiles for non-communicable diseases, and dietary intakes with three component surveys: health interview, health examination and nutrition survey (13). The health interview and health examination were conducted by trained staff members comprising physicians, medical technicians, and health interviewers (13). This data includes additional information to account for complex survey design such as sample weight, description of computing and adjusting weights, primary sampling units, sampling size, and stratification levels. The data were adjusted and weighted for non-response and unequal selection probability to provide national estimates.
Definition of each comorbidity
COPD was determined using the spirometric airflow limitation (forced expiratory volume in one second/forced vital capacity <0.70) (14). Former smokers were defined as those who answered that they used to smoke daily but now does no longer smoke every day and have smoked more than 100 cigarettes in their lifetime. Never smokers were defined as those who answered either they have never smoked at all or have smoked less than 100 cigarettes in their lifetime. Current smokers were defined as those who answered that they smoke daily or sometimes and have smoked more than 100 cigarettes in their lifetime. Comorbidities, including cerebral stroke, osteoporosis, asthma, diabetes, and depression, were defined based on the physician’s diagnosis or patient’s affirmative answer in the survey questionnaire. Hypertension was defined as systole ≥140 mmHg and diastole ≥90 mmHg or being on antihypertensive medication after a physician’s diagnosis.
Measurements
According to the guidelines of the American Thoracic Society and European Respiratory Society, spirometry (Elite-DX or CPFS; Medgraphics, St Paul, MN, USA) was performed, and the acceptability and repeatability criteria of the spirometry were established (15). Normal predictive values of spirometry in the Korean population were obtained based on the second KNHANES (16). Economic status was divided based on quartile value of income level, as follows: 1Q, lowest 0–25 percentile; 2Q, lower-middle 25–50 percentile; 3Q, upper-middle 50–75 percentile; and 4Q, highest 75–100 percentile.
Blood samples for lead measurements were drawn into trace-metal-free ethylenediaminetetraacetic acid (EDTA) tubes, and specimens were immediately transferred at 2–8 °C to a central laboratory for analysis (NeoDin Medical Institute, Seoul, Korea). Lead concentrations in venous whole blood were measured using graphite furnace atomic absorption spectrometry (model AAnalyst 600; PerkinElmer, Finland) (17). The limits of detection (LODs) were 0.12 µg/dL for blood lead, and none of the adult samples exhibited values below the LOD. For internal quality assurance and control, commercial standard reference materials were obtained from Bio-Rad (Lyphochek® Whole Blood Metals Control; Bio-Rad, Hercules, CA, USA). The NeoDin Medical Institute has a certified license from the Korean Ministry of Labor for external quality control of heavy metals laboratory analyses and has passed the German External Quality Assessment Scheme, the US CDC program, and the Korea Occupational Safety and Health Administration (Korea OSHA) program.
Statistical analysis
Given that KHANES data were obtained from a complex sampling survey, for all analyses, appropriate sample weights to account for the unequal probability of sampling and non-response were used for computing reliable estimates, and primary sampling unit was also specified in the analysis to account for design effects of clustering and stratification. Standard errors were estimated using Taylor series linearization. The characteristics of all survey participants are presented with a weighted percentage and standard error, which were compared based on the Rao-Scott modified chi-square test. Univariate regression analyses were performed to select meaningful risk factors associated with blood lead levels. Then, multivariable regression analysis using significant factors obtained from univariate analyses was conducted to find meaningful variables independently associated with blood lead levels among covariates, including demographics and comorbidities. Another multivariable analysis adjusting for significant risk factors proven by univariate analysis was done to find the association among smoking, COPD, and blood lead levels. As a subgroup analysis, it was stratified with sex, age group (≥50 vs. <50 years), education level (higher education vs. lower education), occupation level (farmer or fisherman, technician, labor worker vs. manager or professional, office worker, service, unemployed) to assess the association solely between COPD and smoking on blood lead level. The results of regression analysis were provided and the estimation of blood lead levels was presented with a 95% confidence interval. Statistical analyses were performed with SAS software version 9.4 (SAS Institute, Cary, NC, USA). A P value of less than 0.05 was deemed statistically significant.
Ethics statement
The current study was approved by the institutional review board of Ajou Medical Center (approval number: AJIRB-MED-EXP-9-161). The requirement for informed consent is waived by the ethics review board due to the retrospective nature of the study.
Results
Baseline characteristics
Table 1 presents the demographic profile: an almost equal sex ratio (men 49.2%), a high proportion of older subjects (≥50 years 40.6%, 40–49 years 21.3%, 30–39 years 20.5%, 20–29 years 17.6%), smoking status (current 31.5%, former 11.6%, never 56.9%), and education levels (below middle school 20.3%, middle school 10.3%, high school 38.1%, above high school 31.4%).
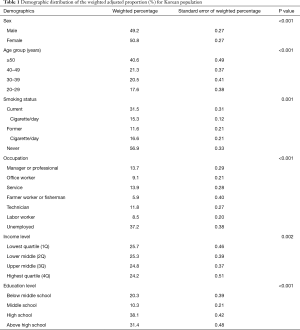
Full table
Univariate analyses of clinical variables including comorbidities
Demographic data showed that blood lead levels were higher among men, older subjects, smokers, lower education group, and lower income group (P value <0.05) (Table 2). Comparison based on occupation revealed that blood lead levels were higher among farmers, fishermen, and technicians than among managers or professionals, office workers, employees in the service sector, and the unemployed (P value <0.05) (Table 2). COPD was associated with higher blood lead (P value <0.001), but other comorbidities such as hypertension, cerebral stroke, osteoporosis, asthma, diabetes, and depression were not related (Table 3).
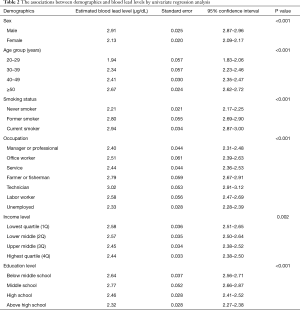
Full table
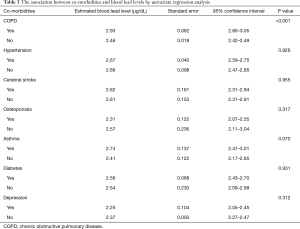
Full table
Multivariable regression analysis
Multivariable linear regression analysis using significant factors obtained from univariate analyses demonstrated that male sex (P value <0.001), older age (P value <0.001), smoking (P value <0.001), occupation (P value =0.002), and education levels (P value <0.001) were significantly associated with higher blood lead levels, whereas income level (P value =0.384) and COPD (P value =0.386) were not (Table 4). Another multivariable linear regression analysis according to the smoking status and the presence of COPD with adjusting for other covariates demonstrated that current smoking was independently associated with high blood lead levels, and COPD was not related (Figure 1).
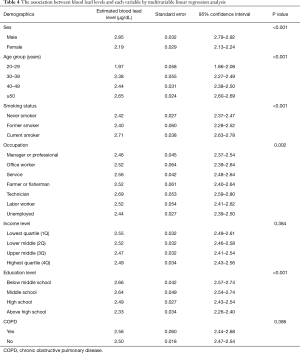
Full table
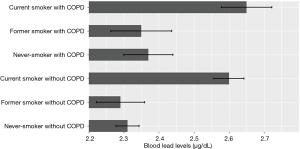
Subgroup analysis
Multivariable linear regression analyses of estimated blood lead levels were performed by stratification according to age, sex, education level, and occupation with a reference group as never-smokers without COPD. Subgroup analyses showed that never-smokers with COPD had higher blood lead levels in male, female, higher education subgroup (high school or more), and the subgroup of office workers and managerial workers, compared to never-smokers without COPD (P value <0.05) (Figures 2,3).
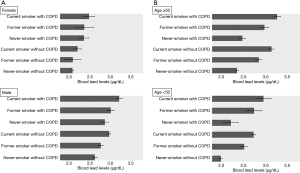
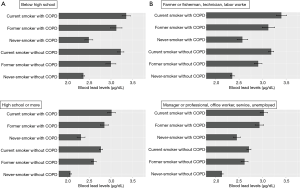
Discussion
Our multivariable regression analyses demonstrated that older age, male sex, smoking, occupation, and lower education levels were independently associated with higher blood lead levels; however, COPD was not related after adjustment of all possible confounding variables. These findings suggest that the higher blood lead levels in COPD should be attributable to the smoking status and socioeconomic factors, such as lower education level and occupation, rather than COPD per se. A careful history inquiring about a broad spectrum of potential lead sources is required and many confounding variables should be considered in analyzing the high blood level of COPD, because COPD has many phenotypes and heterogeneous origins (14). Although smoking is the dominant etiologic source, COPD is often associated with lower socioeconomic status (18). Nevertheless, further studies are necessary concerning the effect of higher blood lead levels on the clinical course of COPD, considering our subgroup analyses showing that some COPD subgroups had higher blood lead levels regardless of smoking behavior.
The average blood lead level of each subgroup in our study was lower than 5 µg/dL. However, blood lead levels lower than 10 µg/dL can have detrimental health consequences including renal injury, cardiovascular mortality, cognitive dysfunction, and adverse reproductive outcomes in women in a dose-dependent manner (4-7). Inorganic lead adversely affects multiple physiologic functions and organ systems throughout various biochemical events (4).
This study revealed that lower education levels and certain occupations were independently associated with higher blood lead levels. These findings highlight the association between socioeconomic factors and blood lead levels reported by other environmental studies (1,19). Most lead exposure is occupational because individuals in lower educational levels usually work in environments with more lead exposure (1,10,19). Our data showed higher blood lead levels in farmers, fishermen, and technicians than among those in managerial jobs and unemployed groups. Hence, occupational sources of lead exposure should be further investigated for reducing environmental lead pollution and improving public health.
The current study revealed that older age and smoking were independently associated with higher lead levels. Our study could not explain the causal linkage which requires further studies, but previous evidence indicated that clinical variables, such as lead absorption through the gastrointestinal tract and nutritional deficiencies including iron deficiency and calcium deficiency rendered older subjects more susceptible to lead poisoning (20,21). Recent evidence also suggested that despite declining smoking rates and secondhand smoke exposure, tobacco smoke continues to be a substantial source of lead exposure in vulnerable and general populations (9,10). Therefore, smoking status should be considered in identifying sources of lead exposure and developing preventative strategies for the management of lead poisoning.
Our study did not prove the relationship between blood lead levels and some comorbidities including diabetes, hypertension, cerebral stroke, etc. except COPD. COPD was associated with higher blood lead levels in the univariate analysis, not in the multivariable analysis adjusted for all confounding factors. However, when stratified and adjusted only with smoking in the subgroup analysis, COPD was statistically associated with higher blood lead levels among males, females, higher education subgroup, and office workers.
A previous study performed in Copenhagen County, Denmark, suggested a significant univariate association of blood lead levels with total mortality, coronary heart disease, and cardiovascular disease (22). Current evidence suggests that increased bone lead levels are associated with hypertension (23,24). Some studies have attempted to further examine the linkage between blood lead levels and environmental factors, such as stress and diet, though further validations are required (25,26). One study reported that the effect of lead on hypertension is most pronounced among highly stressed individuals (25). Western dietary patterns are also reported to be associated with increased lead concentrations (26,27).
Some genes including delta-aminolevulinic acid dehydratase gene, vitamin D receptor gene, and hemochromatosis gene can make certain individuals more vulnerable to environmental lead exposure, influencing the lead absorption and thereby affecting the blood and bone lead levels in human populations (28,29). Genetic polymorphisms are also reported to have a relationship with the impact of lead exposure on neurodegenerative diseases such as Alzheimer’s disease and neurodevelopmental disorder (30,31).
This study had some limitations. First, the etiologic causes of lead exposure were not analyzed in our study, as it was not a longitudinal cohort study. Second, environmental factors, including water supply source, residence, alcohol consumption, and diet, were not accounted for in this study. Third, significant outcomes such as the mortality and cancer risk based on blood lead levels in each comorbidity were not investigated. Fourth, comorbidities, including osteoporosis, asthma, and diabetes were diagnosed using survey questionnaire and physician diagnosis rather than by actual measurements. Fifth, air pollution was not investigated as a variable in our analysis. Sixth, post-bronchodilator values were not obtained due to the lack of information in KNHANES data, and the definition of COPD by less than 70% of FEV1/FVC can harbor the potential risk of overdiagnosis in older patients.
In conclusion, our findings demonstrated that smoking, occupation, and lower education levels along with old age and male sex were independently associated with higher blood lead levels, whereas COPD was not related after adjustment of all possible confounding variables.
Acknowledgments
The authors appreciate the KNHANES team for their effort to collect the KNHANES data. KNHANES is an open and public data to which any researcher can get access through the website (https://knhanes.cdc.go.kr/knhanes/main.do).
Funding: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI16C0992).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd-20-739). The authors have no conflicts of interest to declare.
Ethics Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The current study was approved by the institutional review board of Ajou Medical Center (approval number: AJIRB-MED-EXP-9-161). The requirement for informed consent is waived by the ethics review board due to the retrospective nature of the study.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Neuwirth LS. Resurgent lead poisoning and renewed public attention towards environmental social justice issues: A review of current efforts and call to revitalize primary and secondary lead poisoning prevention for pregnant women, lactating mothers, and children within the U.S. Int J Occup Environ Health 2018;24:86-100. [Crossref] [PubMed]
- Liu J, Ai Y, McCauley L, et al. Blood lead levels and associated sociodemographic factors among preschool children in the South Eastern region of China. Paediatr Perinat Epidemiol 2012;26:61-9. [Crossref] [PubMed]
- Kim MG, Ryoo JH, Chang SJ, et al. Blood Lead Levels and Cause-Specific Mortality of Inorganic Lead-Exposed Workers in South Korea. PLoS One 2015;10:e0140360. [Crossref] [PubMed]
- Kosnett MJ, Wedeen RP, Rothenberg SJ, et al. Recommendations for medical management of adult lead exposure. Environ Health Perspect 2007;115:463-71. [Crossref] [PubMed]
- Lanphear BP, Rauch S, Auinger P, et al. Low-level lead exposure and mortality in US adults: a population-based cohort study. Lancet Public Health 2018;3:e177-84. [Crossref] [PubMed]
- Lin JL, Lin-Tan DT, Li YJ, et al. Low-level environmental exposure to lead and progressive chronic kidney diseases. Am J Med 2006;119:707.e1-9. [Crossref] [PubMed]
- Menke A, Muntner P, Batuman V, et al. Blood lead below 0.48 micromol/L (10 microg/dL) and mortality among US adults. Circulation 2006;114:1388-94. [Crossref] [PubMed]
- North CM, Rice MB, Ferkol T, et al. Air Pollution in the Asia-Pacific Region. A Joint Asian Pacific Society of Respirology/American Thoracic Society Perspective. Am J Respir Crit Care Med 2019;199:693-700. [Crossref] [PubMed]
- Richter PA, Bishop EE, Wang J, et al. Trends in tobacco smoke exposure and blood lead levels among youths and adults in the United States: the National Health and Nutrition Examination Survey, 1999-2008. Prev Chronic Dis 2013;10:E213. [Crossref] [PubMed]
- Adult blood lead epidemiology and surveillance--United States, 2008-2009. MMWR Morb Mortal Wkly Rep 2011;60:841-5. [PubMed]
- Bellou V, Belbasis L, Konstantinidis AK, et al. Elucidating the risk factors for chronic obstructive pulmonary disease: an umbrella review of meta-analyses. Int J Tuberc Lung Dis 2019;23:58-66. [Crossref] [PubMed]
- Salvi SS, Barnes PJ. Chronic obstructive pulmonary disease in non-smokers. Lancet 2009;374:733-43. [Crossref] [PubMed]
- Kweon S, Kim Y, Jang MJ, et al. Data resource profile: the Korea National Health and Nutrition Examination Survey (KNHANES). Int J Epidemiol 2014;43:69-77. [Crossref] [PubMed]
- Rabe KF, Hurd S, Anzueto A, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med 2007;176:532-55. [Crossref] [PubMed]
- Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J 2005;26:319-38. [Crossref] [PubMed]
- Choi JK, Paek D, Lee JO. Normal Predictive Values of Spirometry in Korean Population. Tuberc Respir Dis 2005;58:230-42. [Crossref]
- Choi YH, Park SK. Environmental Exposures to Lead, Mercury, and Cadmium and Hearing Loss in Adults and Adolescents: KNHANES 2010-2012. Environ Health Perspect 2017;125:067003. [Crossref] [PubMed]
- Kanervisto M, Vasankari T, Laitinen T, et al. Low socioeconomic status is associated with chronic obstructive airway diseases. Respir Med 2011;105:1140-6. [Crossref] [PubMed]
- Kim KR, Lee SW, Paik NW. Cross-sectional analysis of blood lead level of entire Korean lead workers. Ind Health 2006;44:318-27. [Crossref] [PubMed]
- McCabe EB. Age and sensitivity to lead toxicity: a review. Environ Health Perspect 1979;29:29-33. [Crossref] [PubMed]
- Mahaffey KR. Environmental lead toxicity: nutrition as a component of intervention. Environ Health Perspect 1990;89:75-8. [Crossref] [PubMed]
- Møller L, Kristensen TS. Blood lead as a cardiovascular risk factor. Am J Epidemiol 1992;136:1091-100. [Crossref] [PubMed]
- Zheutlin AR, Hu H, Weisskopf MG, et al. Low-Level Cumulative Lead and Resistant Hypertension: A Prospective Study of Men Participating in the Veterans Affairs Normative Aging Study. J Am Heart Assoc 2018;7:e010014. [Crossref] [PubMed]
- Navas-Acien A, Schwartz BS, Rothenberg SJ, et al. Bone lead levels and blood pressure endpoints: a meta-analysis. Epidemiology 2008;19:496-504. [Crossref] [PubMed]
- Peters JL, Kubzansky L, McNeely E, et al. Stress as a potential modifier of the impact of lead levels on blood pressure: the normative aging study. Environ Health Perspect 2007;115:1154-9. [Crossref] [PubMed]
- Wang X, Ding N, Tucker KL, et al. A Western Diet Pattern Is Associated with Higher Concentrations of Blood and Bone Lead among Middle-Aged and Elderly Men. J Nutr 2017;147:1374-83. [Crossref] [PubMed]
- Ding N, Wang X, Tucker KL, et al. Dietary patterns, bone lead and incident coronary heart disease among middle-aged to elderly men. Environ Res 2019;168:222-9. [Crossref] [PubMed]
- Onalaja AO, Claudio L. Genetic susceptibility to lead poisoning. Environ Health Perspect 2000;108 Suppl 1:23-8. [PubMed]
- Szymańska-Chabowska A, Laczmanski L, Jedrychowska I, et al. The relationship between selected VDR, HFE and ALAD gene polymorphisms and several basic toxicological parameters among persons occupationally exposed to lead. Toxicology 2015;334:12-21. [Crossref] [PubMed]
- Wang Z, Claus Henn B, Wang C, et al. Genome-wide gene by lead exposure interaction analysis identifies UNC5D as a candidate gene for neurodevelopment. Environ Health 2017;16:81. [Crossref] [PubMed]
- Lee J, Peterson SM, Freeman JL. Sex-specific characterization and evaluation of the Alzheimer's disease genetic risk factor sorl1 in zebrafish during aging and in the adult brain following a 100 ppb embryonic lead exposure. J Appl Toxicol 2017;37:400-7. [Crossref] [PubMed]


