
NEWS technique: easy and reliable thoracoscopic wedge resections of lung tumors
Introduction
The current standard surgical treatment for primary lung cancer is a lobectomy; however, a wedge resection is used for patients showing a reduced pulmonary reserve, the elderly, or those with other serious comorbidities (1). Meanwhile, for lung metastasis, a wedge resection is the most common procedure for treatment (2). Given that a thoracoscopic lobectomy for early-stage lung cancer will soon be a prerequisite for a new generation of thoracic surgeons, trainee physicians need to have adequate experience in minor procedures such as wedge resections, bullectomies, and biopsies (3). Thoracoscopic wedge resections for lung malignancies are normally straightforward for expert surgeons but may be more difficult for trainees. Notably, wedge resections may increase the likelihood of the recurrence of local cancer from the surgical margin compared with anatomical segmentectomy and lobectomy (4).
We herein propose a simple and reliable method for the surgical treatment for lung malignancy based on traditional three-port thoracoscopic surgery, which aims for the successful tactile localization and curative wedge resection of pulmonary tumors. This technique will enable residents and trainee physicians alike to master basic surgical skills before becoming competent in thoracoscopic lobectomies.
The NEWS method
The acronym, “NEWS”, is based on surgical treatment for lung malignancy and is created from initial capital letters of the following words: nodule or North; endoscopy or East; wedge resection or West; and stapler or South. Most such patients showed small nodules diagnosed as probable malignancies based on preoperative computed tomography (CT) images and clinical observation.
NEWS requires three skin incisions to place two trocars (11.5 mm, Thoracoport; Covidien, Dublin, Ireland), and a wound protector/retractor (3 cm, XXS, Alexis; Applied Medical, Rancho Santa Margarita, CA, USA). A 10-mm, 45-degree endoscope (thoracoscope) is introduced through one trocar. A pair of long forceps (Forester lung grasping clamp; Scanlan, ST Paul, MN, USA) and stapler (Endo GIA reloaded with Tri-staple; Medtronic, Minneapolis, MN, USA or Echelon Flex Endopath; Ethicon, Bridgewater, NJ, USA) are inserted through another trocar. Ring-shaped forceps (3.5 cm, M; P.N.CATCH, Takasago, Japan) are inserted through a wound protector/retractor (Figure 1). The two trocars and wound protector/retractor are so-called thoracic ports. We herein define each as a nodule- or North-port (N-port), stapler- or South-port (S-port), and endoscope- or East-port (E-port; Figure 2).
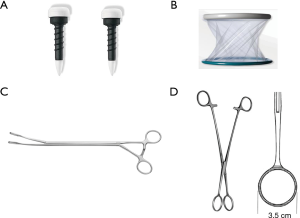
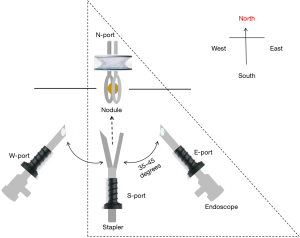
Placement of the three ports is as follows: under general anesthesia, the patient is placed in a lateral decubitus position after the insertion of a double-lumen endotracheal tube. Three positions for skin incisions are pre-planned using preoperative CT images and the following body landmarks: body surface projections, such as a scapula, nipple, and ribs; and longitudinal information, such as imaginary anterior, middle, and posterior axillary lines. The first trocar is inserted as an S-port through a skin incision, through which an endoscope is then introduced to scan the surgical field for the presence or absence of pleural adhesions, or deflated lung. A second trocar as an E-port is inserted while looking into the thoracic cavity using the endoscope in the S-port; the endoscope is subsequently extracted from the S-port and introduced through the E-port. The E-port is then used as an endoscope (thoracoscope) and is usually placed on the right at less than 45 degrees from a virtual N–S port line. Finally, a third wound protector/retractor as an N-port is placed leading to the completion of a port triangle.
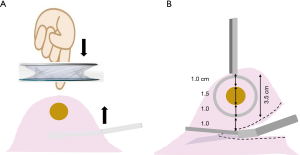
The N-port is placed on the chest wall nearest the nodule after examining preoperative CT images. A pair of long forceps inserted through the S-port is used to move lung tissue closer to the N-port to allow finger palpation (Figure 3A). When the nodule is grasped with a pair of ring-shaped forceps through the N-port, the flat plane of the ring should be in the same direction as an ideal resection line in the lung parenchyma (Figure 3B). For this reason, the S-port is positioned in the chest wall at a distance along this ideal resection line, which is defined as an N–S port line (Figure 2).
Typical examples of the NEWS method are shown in an illustration (Figure 4). Tactile localization of firm nodules located in a shallow position from the pleural surface is relatively easily made. In this case, a wedge resection had been applied because the patient was elderly and had a reduced pulmonary reserve; however, neither local recurrence nor distant metastasis occurred by 36 months after surgery.
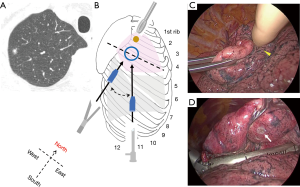
In the case of nodules in the posterior basal segment (S10) of the right or left lower lobe, dissection of the pulmonary ligament enables deflated lung tissue close to the N-port to be lifted using a pair of long forceps, allowing for subsequent tactile localization (Figure 5). The patient shown had undergone a standard wedge resection for lung metastasis, and showed no local recurrence 24 months after surgery.
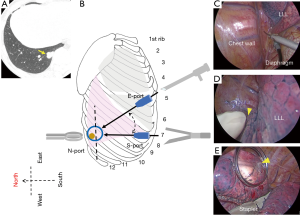
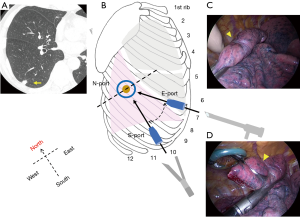
When nodules are located in the apex of the lung, that is, in the apical segment (S1) of the right upper lobe (Figure 6) or the apical posterior segment (S1+2) of the left upper lobe, an N-port would only be placed in the 3rd or 4th intercostal space to avoid potential interference from chest wall structures such as the scapula or thick muscular layers (Figure 6B). However, apical portions can be pulled closer to the actual N-port with a pair of long forceps due to good mobility of the apex (Figure 6C). The patient shown underwent a wedge resection with 3.0-cm resection margin because of a diagnosis and treatment for a 0.5-cm subsolid nodule, found after chemotherapy for a malignant lymphoma. Given this was a primary lung adenocarcinoma, but probably of a minimally invasive type in intraoperative pathology and with a comorbidity of malignant lymphoma, a lobectomy was not performed. This patient showed no local recurrence or distant metastasis 33 months after surgery.
Results
Between 2016 and 2019, 37 patients (25 males; 69 years, median age, 49–85) with 40 lesions underwent wedge resections using the NEWS method performed either by trainee physicians or residents with advising surgeons. CT revealed target lesions to be 11 ground-glass (size, 0.7–2.0 cm; median 1.2) and 29 solid (size, 0.4–3.0 cm; median 1.2) nodules. Resection margins of a 2.0-cm median length (range, 1.5–4.0 cm) were maintained. No complications, such as intraoperative injury to blood vessels, or postoperative atelectasis or pneumonia of surgical sites, occurred.
Pathologically, a total of 40 lesions revealed 18 lung metastases, 14 primary adenocarcinomas, four primary squamous cell carcinomas, two lymphomas, and two benign intrapulmonary lymph nodes. Subsequently, patients have not experienced local recurrences between 3 and 40 months (median, 20) after surgery.
Comments
“NEWS” is a simple and reliable method, based on traditional three-port thoracoscopic surgery, that allows the successful tactile localization and curative wedge resection of lung tumors. The principles of this method consist of four steps: (I) appropriate port-positioning for a target, (II) tactile localization, (III) grasping with ring-shaped forceps, and (IV) safe stapling and resection.
Regarding steps 1 and 2, the positioning of three ports in line with a target allows physicians to easily confirm the presence of a nodule with a finger. Notably, lung tissue containing a nodule, especially in the middle, lower lobes, and lingular segments, often moves cranially due to deflation of the relevant lung during one-lung ventilation. Therefore, in order to allow touching in such cases, an actual N-port should be moved toward the head for one intercostal space from a virtual N-port place as estimated from preoperative CT images.
However, tactile localization for smaller and/or deeply located nodules is challenging during thoracoscopic surgery. In this regard, a modified technique for the tactile localization of deep pulmonary nodules as well as the development of a newly designed tactile mechanoreceptor during thoracoscopic surgery have been described (5,6). Recently, many other techniques identifying such nodules have been developed. They include placement of a hook-wire or microcoil, and injection of lipiodol or dye as preoperative CT-guided percutaneous marking; and the instillation of a barium mixture, dye, or microcoil as a preoperative bronchoscopic marking. In addition, the intraoperative use of an electromagnetic navigation bronchoscope and cone-beam CT has been reported (7). However, these methods have their own pros and cons. The most common method, the CT-guided placement of a hook-wire, is associated with several complications such as pneumothorax, hemorrhage, dislocation, and even rare but critical air embolisms. Although our classical finger-touch method does not take the place of the above methods, if feasible, it can be a complementary technique to save time and effort (5).
As advantages to steps 3 and 4, this technique can adjust the resection margin to an intended distance using a ring width measuring 3.5 cm of the ring-shaped forceps during surgery. In wedge resections of non-small cell lung cancers, an insufficient surgical margin distance affected local recurrence and patient survival (4,8). This can be caused by remnant cancer cells beyond the surgical margin. Hence, prior studies have indicated successful wedge resections due to surgical margin distances greater than the nodule diameter or 1.5–2.0 cm (9-11). Similar to wedge resections for colorectal lung metastases, local recurrence can contribute to satellite tumor cells and the aerogenous spread of floating cancer cell clusters surrounding the metastatic nodule (12,13). Accordingly, a safety margin distance of more than 0.5–2.0 cm has been indicated (2).
Careful manipulations in steps 1–4 can result in reduced deformity of the remaining lung and avoid damage to central blood vessels. When a nodule is located near the interlobar area, the direction of the resection line (a flat plane of the ring) should be oriented toward the angle of the involved interlobar fissure.
Regarding the outcomes of this method, no local recurrence associated with resection lines has occurred in 37 patients operated on by trainee physicians and residents. However, the further accumulation of cases and long-term follow-up are needed to determine the usefulness of this method in an educational setting and clinical practice.
In conclusion, the NEWS method is a simple and reliable technique, useful when tactile localization is feasible, which, as a complementary method, can save time and effort. In addition, this easy and safe procedure, suitable for residents and trainee physicians alike, may act as a gateway to mastering thoracoscopic lobectomies and reduced-portal surgery.
Acknowledgments
Funding: None.
Footnote
Peer Review File: Available at http://dx.doi.org/10.21037/jtd-19-4020
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd-19-4020). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the Declaration of Helsinki (as revised in 2013). Written informed consent was obtained from patients for publication of this manuscript and any accompanying images, and their anonymity was ensured.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Donington J, Ferguson M, Mazzone P, et al. American College of Chest Physicians and Society of Thoracic Surgeons consensus statement for evaluation and management for high-risk patients with stage I non-small cell lung cancer. Chest 2012;142:1620-35. [Crossref] [PubMed]
- Chung JH, Lee SH, Yi E, et al. Impact of resection margin length and tumor depth on the local recurrence after thoracoscopic pulmonary wedge resection of a single colorectal metastasis. J Thorac Dis 2019;11:1879-87. [Crossref] [PubMed]
- Sandri A, Filosso PL, Lausi PO, et al. VATS lobectomy program: the trainee perspective. J Thorac Dis 2016;8:S427-30. [Crossref] [PubMed]
- Sawabata N. Locoregional recurrence after pulmonary sublobar resection of non-small cell lung cancer: can it be reduced by considering cancer cells at the surgical margin? Gen Thorac Cardiovasc Surg 2013;61:9-16. [Crossref] [PubMed]
- Zheng H, Jiang S, Chen C. Stepwise tactile localization and wedge resections for deep pulmonary nodules during video-assisted thoracoscopic surgery. Thorac Cardiovasc Surg 2016;64:182-6. [Crossref] [PubMed]
- Barmin V, Sadovnichy V, Sokolov M, et al. An original device for intraoperative detection of small indeterminate nodules†. Eur J Cardiothorac Surg 2014;46:1027-31. [Crossref] [PubMed]
- Sato M. Precise sublobar lung resection for small pulmonary nodules: localization and beyond. Gen Thorac Cardiovasc Surg 2020;68:684-91. [Crossref] [PubMed]
- Sato M, Kobayashi M, Kojima F, et al. Effect of virtual-assisted lung mapping in acquisition of surgical margins in sublobar lung resection. J Thorac Cardiovasc Surg 2018;156:1691-701.e5. [Crossref] [PubMed]
- Sawabata N, Ohta M, Matsumura A, et al. Optimal distance of malignant negative margin in excision of nonsmall cell lung cancer: a multicenter prospective study. Ann Thorac Surg 2004;77:415-20. [Crossref] [PubMed]
- Mohiuddin K, Haneuse S, Sofer T, et al. Relationship between margin distance and local recurrence among patients undergoing wedge resection for small (≤2 cm) non-small cell lung cancer. J Thorac Cardiovasc Surg 2014;147:1169-77. [Crossref] [PubMed]
- Wolf AS, Swanson SJ, Yip R, et al. The Impact of Margins on Outcomes After Wedge Resection for Stage I Non-Small Cell Lung Cancer. Ann Thorac Surg 2017;104:1171-8. [Crossref] [PubMed]
- Welter S, Theegarten D, Trarbach T, et al. Safety distance in the resection of colorectal lung metastases: a prospective evaluation of satellite tumor cells with immunohistochemistry. J Thorac Cardiovasc Surg 2011;141:1218-22. [Crossref] [PubMed]
- Welter S, Arfanis E, Christoph D, et al. Growth patterns of pulmonary metastases: should we adjust resection techniques to primary histology and size? Eur J Cardiothorac Surg 2017;52:39-46. [Crossref] [PubMed]


