
Feasibility of thoracoscopic pulmonary bullectomy using a transareolar approach for treatment of primary spontaneous pneumothorax
Introduction
Primary spontaneous pneumothorax (PSP) is defined as the presence of air in the thorax between the chest wall and lung (1). This disease is very common among young patients, particularly men. Surgery is necessary to stop the prolonged air leakage or reduce the rate of pneumothorax recurrence; Brock initially reported the necessity of surgical treatment for PSP in 1948 (2). Although Brock used a thoracotomy approach, the thoracoscopic approach became common in the 1990s when several clinicians demonstrated that it was less invasive and resulted in more rapid recovery (3,4).
The most common approach during the early years was triportal. However, several clinicians have recently reported the efficacy of a uniportal thoracoscopic approach for PSP (5-9). Our group also reported the usefulness of uniportal thoracoscopic surgery for PSP using multidegree forceps (10). However, a uniportal approach results in poor maneuverability due to interference among surgical instruments through a single port; thus, surgeons hesitate to use this approach, although it carries advantages of rapid recovery and reduced invasiveness. In September 2017, we introduced a transareolar approach to overcome poor maneuverability. We expected that interference among surgical instruments would be avoided, in a manner similar to that of the conventional triportal thoracoscopic approach. In addition, greater cosmetic satisfaction would be achieved for the operative wound, as the resulting wounds were less visible. In the present study, we prospectively identified the feasibility of thoracoscopic pulmonary bullectomy using a transareolar approach for treatment of PSP. We present the following article in accordance with the Strenghening the Reporting of Observational Studies in Epidemiology (STROBE) reporting checklist (11) (available at http://dx.doi.org/10.21037/jtd-20-1548).
Methods
Patient selection
This prospective, single-center clinical trial enrolled 10 patients with PSP who were undergoing thoracoscopic pulmonary bullectomy using a transareolar approach. Inclusion criteria were as follows: (I) male sex; (II) age ≤40 years; (III) presence of localized blebs or bullae (i.e., not diffuse type) (12); (IV) absence of underlying pulmonary diseases, such as chronic obstructive pulmonary disease, asthma, or pulmonary fibrosis; (V) absence of hemothorax; and (VI) no history of surgery on the affected side. While the patient aged older than 40 occasionally reveal pneumothorax caused by the underlying diseases, which is considered as secondary spontaneous pneumothorax, the patients aged 40 or younger rarely have the underlying disease. This approach is for PSP. Therefore, we excluded the patients aged more than 40 from this prospective study. Moreover, female patient was defined as a contra-indication because subcutaneous mammary gland hindered putting surgical ports on areolar appropriately. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study protocol was approved by the Institutional Review Board and Ethics Committee of the Japanese Red Cross Maebashi Hospital (approval no. 29-9). Written informed consent to participate was obtained from each patient.
Sample size
We have performed triportal thoracoscopic surgery for so many patients with primary or spontaneous pneumothorax. The transareolar approach was performed by means of three ports and was expected to be completed relatively easily with a steep learning curve for several cases. Therefore, we estimated that 10 patients would constitute an appropriate sample size to evaluate the feasibility of this technique. We performed thoracoscopic pulmonary bullectomy via transareolar approach on 10 patients consecutively. Data were collected between September 2017 and March 2019 at Japanese Red Cross Maebashi Hospital.
Preoperative treatments
We diagnosed patients with PSP in accordance with the 2010 British Thoracic Society pleural disease guidelines (13). All patients presented to our department with sudden dyspnea and chest pain. A chest tube drain was inserted on admission when a chest X-ray revealed moderate or severe collapse. All patients received a 16-Fr thoracostomy tube (1 cm skin incision) through the fifth intercostal space at the anterior axillary line. Subsequently, elective surgical treatment was performed. Regardless of whether air leakage stopped and the pneumothorax episode was the first for a particular patient, surgical treatment was performed if desired by the patient, as this was expected to reduce the recurrence rate. When the chest X-ray revealed mild collapse, the patient was followed up in the outpatient clinic.
All patients received a physical examination, laboratory testing, a chest computed tomography scan, and (when necessary) a cardiac consultation before surgery.
Surgical procedures
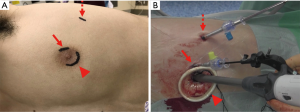
Surgical procedures were performed under general anesthesia using a single-lung ventilation technique and a double-lumen endotracheal tube. Before surgery began, the patient was placed in a lateral decubitus position; the patient’s arm was technically adjusted to situate the areola at the fourth intercostal space. A 5-mm arc incision was made for the forceps port at the upper edge of the areola; a 2-cm arc incision for stapling was made at the lower part of the areola through the fourth intercostal space (Figure 1A). Figure 1B shows photographs of the operative field when using the transareolar approach. A 3-mm rigid port was inserted through the 5-mm incision, while the 2-cm incision was covered with an XXS size wound retractor (Alexis Wound Retractor; Applied Medical, Rancho Santa Margarita, CA, USA). A 1-cm incision at the fifth intercostal space of the mid-axial line for chest tube drainage was used as a thoracoscopic port. If the patient had not received a preoperative chest tube drain, a thoracoscopic port was added in the same position. A 5-mm flexible type of thoracoscope was used to explore the thoracic cavity, specifically focusing on the region where blebs or bullae were detected by preoperative computed tomography.
Visceral blebs or bullae were excised using an Endostapler (Powered Echelon Flex GST System; Johnson & Johnson, Parsippany, NJ, USA). Subsequently, polyglycolic acid felt (Neoveil; sheet type; Igaki Medical Planning Co., Ltd., Kyoto, Japan) was applied to the staple lines using fibrin glue sealant (Beriplast P; CSL Behring, King of Prussia, PA, USA) to prevent any postoperative air leakage and reduce the recurrence rate (14). Therefore, we have not used pleurodesis techniques including administration of talc, pleurectomy or pleural abrasion. We performed no sealing test during the surgery in any case because the targeted bullae or blebs for resection were detected in the preoperative CT examination and the resected region by staplers was expected to rarely reveal air leak. At the end of surgery, a 19-Fr chest drain (19-Fr Blake Drain; Ethicon, Paramus, NJ, USA) was placed with the drain positioned anterior-to-posterior through the apex in an inverted-U shape. This chest drain was put into the thorax via subcutaneous tunnel between areolar and 5th intercostal incision in order to skip stapling when we removed it on patient’s bed side. The day after air leakage had stopped, the chest tube drain was removed when a well-expanded lung was confirmed by chest X-ray. The patient was finally discharged if a chest X-ray on the following day revealed no collapse of the affected lung. Video 1 demonstrates these surgical procedures using a transareolar approach.
Data collection and follow-up
We recorded operating time, blood loss volume, duration of postoperative drainage, postoperative hospital stays, postoperative complications, and recurrence. Recurrence was defined as detection of lung collapse by chest X-ray or computed tomography within 1 year postoperatively. Moreover, we assessed postoperative wound-related pain using numerical rating scale (NRS), and cosmetic satisfaction of surgical wound using following scale (1, poor; 2, not bad; 3, good; 4, excellent) at 1 week postoperatively during a hospital visit and at 12 months postoperatively by telephone. Observations for each patient were completed by telephone.
Statistical analysis
Statistical analysis was not applicable in this study because this study did not perform comparative analysis.
Results
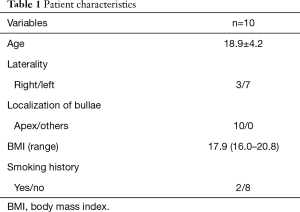
Table 1 summarized the patients’ characteristics in this cohort.
Full table
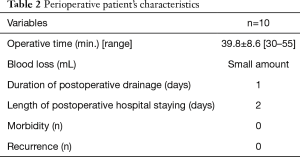
Perioperative outcomes are shown in Table 2. All procedures were completed using a transareolar approach without any additional ports or conversion to thoracotomy in any patient. The mean operative time was 39.8±8.6 min (range 30 to 55). The mean blood loss volume during surgery was extremely small in all patients. The duration of postoperative drainage was 1 day, while the length of postoperative hospital stay was 2 days in all patients. Figure 2 demonstrates the postoperative course of the surgical wound in one patient. The surgical wound was fixed and covered with surgical tape immediately postoperatively (Figure 2A). On postoperative day (POD) 7, the wound became discreet after removal of surgical tape (Figure 2B). No obvious surgical scar was detected on the areola during postoperative month (POM) 12 (Figure 2C). No patient developed a wound infection. In addition, local complications (e.g., mastitis or a hypertrophic scar) were not observed in any patient. No postoperative respiratory complications occurred, including pneumonia, postoperative re-collapse, or prolonged air leakage. Moreover, no patient developed recurrence of PSP during the study period.
Full table
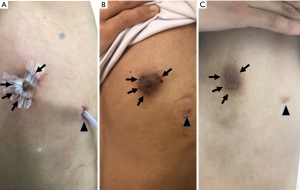
Table 3 shows the postoperative cosmetic satisfaction and pain scores. On POD 7, the mean cosmetic satisfaction score of the surgical wound was 3.3, while the median score was 3.5. Five patients reported a cosmetic satisfaction score of 4; three patients reported a cosmetic satisfaction score of 3; the remaining two patients reported a cosmetic satisfaction score of 2. At POM12, the mean cosmetic score was 3.2, while the median score was 3.0. Four patients reported a cosmetic satisfaction score of 4; 4 patients reported a cosmetic satisfaction score of 3; the remaining two patients reported a cosmetic satisfaction score of 2. Notably, no patient reported a cosmetic satisfaction score of 1 on POD 7 or at POM 12. On POD 7, the pain score ranged from 0 to 3 in all patients (mean score, 1.5; median score, 2.0). Only one patient reported a pain score of 3. None of the patients had any pain at POM 12.

Full table
Discussion
Xu and colleagues firstly reported the efficacy of transareolar bullectomy although this was a retrospective nature and included the efficacy of other approach such as subxiphoid one for female patients (15). Subsequently, Lin et al. retrospectively reported the feasibility and safety of transareolar bullectomy for treatment of patients with PSP, although their report described a retrospective cohort study (16). To the best of our knowledge, they were the only reports describing transareolar bullectomy for PSP. In their report, Xu used uniportal skin incision on the areola while Lin did a 5-mm skin incision for a thoracoscope and a 20-mm skin incision for surgical instruments including stapler or forceps on the areola. The difference between our procedure and theirs was the port position for the thoracoscope. We made a skin incision for the thoracoscope at the fifth intercostal space on the anterior axillary line, because most patients with PSP in our department, who were undergoing surgical treatment to stop a prolonged air leak or reduce the recurrence rate, had received a chest tube drain on admission. The skin incision for the drain was made at the fifth intercostal space on the anterior axillary line. Therefore, we used this incision as the thoracoscope port. Although we may be able to insert a chest tube through the areola, we have not performed this procedure because chest tubes for a PSP are inserted by clinicians in the emergency department; these clinicians are not familiar with placement of a drain through the areola.
Notably, we could not obtain a good surgical view of the posterior portion of the lung when the thoracoscope was inserted through the areolar incision. Missed blebs or bullae are considered a main reason for postoperative recurrence of PSP. Therefore, it is important to explore the entire lung through the thoracoscope to avoid missing blebs or bullae. Accordingly, we added a thoracoscopic port in all patients. Moreover, both of the two transareolar incisions were put on the 4th intercostal space in our procedure while Lin and colleagues put the two transareolar incisions on different intercostal spaces. We speculated some readers might consider surgeons struggled to manipulate surgical instruments appropriately if the areola was small. However, when these incisions were placed on the same intercostal space, we could manipulate any surgical instruments appropriately in any case regardless of the size of areola.
In this study, female patient was defined as a contra-indication because subcutaneous mammary gland hindered putting surgical ports on areola appropriately. Based on the similar reason, obese male patients might not be suitable for this transareolar approach due to the subcutaneous rich fat tissue under areola. We did not set obesity on exclusion from this prospective study because we rarely encountered obese male patients with PSP, which was proved by the data of BMI in the 10 patients. However, it might be reasonable that obese patients are defined as a contra-indication in the future study.
In the present study, we estimated postoperative pain using an NRS. The NRS scores ranged from 0 to 3 on POD 7, indicating minimal postoperative pain. In addition, no patients had pain at POM 12. Although this was not a comparative study between a uniportal and triportal approach, we considered this result to be satisfactory. Several previous studies have revealed that a uniportal approach can reduce postoperative pain, compared to the conventional triportal approach (8). Nachira et al. insisted that reduction of postoperative pain might be related to a more anterior port position, where the intercostal space is wider, as well as the use of a wound retractor (9). These factors could protect intercostal tissue and the neurovascular bundle from scratching and compression. They may be relevant to our transareolar approach because of the wide intercostal space on the areola. In addition, the timing of postoperative pain evaluation is controversial. Some clinicians evaluate postoperative pain of patients with PSP on POD1 after uniportal or triportal surgery; the uniportal approach has achieved better outcomes (6,9). We did not evaluate postoperative pain on POD1 because a chest tube affects pain. Moreover, all the patients discharged on POD2, which was sometimes expected to fail to ask the postoperative pain to patients. Therefore, we established the evaluation of postoperative pain on POD7 as our short-term outcome and POM12 as our long-term outcome.
Cosmetic satisfaction after the PSP operation is very important because PSP occurs frequently in young patients. The cosmetic satisfaction outcome in this study was considered generalizable to broad patient groups. The cosmetic advantage of our transareolar approach is an invisible operative scar, although there are more skin incisions for the transareolar approach than for the uniportal approach. However, if the operative scar on the areola develops hypertrophy, it can be easily noticed because it is located on the anterior portion of the body. Fortunately, no patient in our study developed a hypertrophic operative scar.
To our best knowledge, there have been no studies comparing the perioperative results, postoperative pain and cosmetic satisfaction between transareolar and conventional triportal approaches in pulmonary bullectomy. Xu’s report is the only one comparing them between transareolar and multiportal approaches although the multiportal approach had two ports. In this report, transareolar approach revealed statistically superior postoperative pain relief and cosmetic satisfaction to multiportal approach with the equivalent perioperative results.
Our study did not show specific postoperative complications correlated with using transareolar ports, which was similar to other previous studies using transareolar approach for thoracic surgeries (15-17). Although we worried about the occurrence of mastitis before beginning this prospective study, we have not encountered this complication as the other previous studies also did not show it. However, the number of patients in these studies including ours is relatively small. Therefore, we have to care about the possible complications including mastitis in the future.
In addition to the good postoperative pain and cosmetic satisfaction outcomes, our approach ensures better maneuverability of surgical instruments, compared to the uniportal approach. The uniportal approach involves difficulties in terms of maneuverability because of interference among surgical instruments through a single incision, which is markedly different from the conventional triportal approach. However, our transareolar approach involves the thoracoscopic principles of triangulation, as well as the conventional triportal approach. Therefore, maneuverability was good, which might facilitate good perioperative outcomes, including operative time or postoperative recurrence rate.
The limitations of our study were the small number of patients and the single-arm design of the trial. However, this is the first study to prospectively investigate the efficacy of thoracoscopic transareolar pulmonary bullectomy. In the future, we will conduct a prospective study to compare the transareolar and uniportal approaches.
In conclusion, this study demonstrated that transareolar thoracoscopic pulmonary bullectomy is feasible and safe for treatment of PSP, and that it yielded a high degree of cosmetic satisfaction and minimal postoperative pain. This new approach could be a novel option for surgical treatment of PSP.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/jtd-20-1548
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jtd-20-1548
Peer Review File: Available at http://dx.doi.org/10.21037/jtd-20-1548
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd-20-1548). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study protocol was approved by the Institutional Review Board and Ethics Committee of the Japanese Red Cross Maebashi Hospital (approval no. 29-9). Written informed consent to participate was obtained from each patient.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Weissberg D, Refaely Y. Pneumothorax: experience with 1,199 patients. Chest 2000;117:1279-85. [Crossref] [PubMed]
- Brock RC. Recurrent and chronic spontaneous pneumothorax. Thorax 1948;3:88-111. [Crossref] [PubMed]
- Pagès PB, Delpy JP, Falcoz PE, et al. Videothoracoscopy versus thoracotomy for the treatment of spontaneous pneumothorax: a propensity score analysis. Ann Thorac Surg 2015;99:258-63. [Crossref] [PubMed]
- Sedrakyan A, van der Meulen J, Lewsey J, et al. Video assisted thoracic surgery for treatment of pneumothorax and lung resections: systematic review of randomised clinical trials. BMJ 2004;329:1008. [Crossref] [PubMed]
- Tsuboshima K, Wakahara T, Matoba Y, et al. Single-incision thoracoscopic surgery using a chest wall pulley for lung excision in patients with primary spontaneous pneumothorax. Surg Today 2015;45:595-9. [Crossref] [PubMed]
- Song IH, Lee SY, Lee SJ. Can single-incision thoracoscopic surgery using a wound protector be used as a first-line approach for the surgical treatment of primary spontaneous pneumothorax? A comparison with three-port video-assisted thoracoscopic surgery. Gen Thorac Cardiovasc Surg 2015;63:284-9. [Crossref] [PubMed]
- Ocakcioglu I, Alpay L, Demir M, et al. Is single port enough in minimally surgery for pneumothorax? Surg Endosc 2016;30:59-64. [Crossref] [PubMed]
- Qin SL, Huang JB, Yang YL, et al. Uniportal versus three-port video-assisted thoracoscopic surgery for spontaneous pneumothorax: a meta-analysis. J Thorac Dis 2015;7:2274-87. [PubMed]
- Nachira D, Ismail M, Meacci E, et al. Uniportal vs. triportal video-assisted thoracic surgery in the treatment of primary pneumothorax-a propensity matched bicentric study. J Thorac Dis 2018;10:S3712-9. [Crossref] [PubMed]
- Igai H, Kamiyoshihara M, Ibe T, et al. Single-incision thoracoscopic surgery for spontaneous pneumothorax using multi-degrees of freedom forceps. Ann Thorac Cardiovasc Surg 2014;20:974-9. [Crossref] [PubMed]
- von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. PLoS Med 2007;4:e296. [Crossref] [PubMed]
- Lee HY, Kim EY, Kim YS, et al. Prognostic significance of CT-determined emphysema in patients with small cell lung cancer. J Thorac Dis 2018;10:874-81. [Crossref] [PubMed]
- MacDuff A, Arnold A, Harvey J. BTS Pleural Disease Guideline Group. Management of spontaneous pneumothorax: British Thoracic Society Pleural Disease Guideline 2010. Thorax 2010;65 Suppl 2:ii18-ii31. [Crossref] [PubMed]
- Itano H. The optimal technique for combined application of fibrin sealant and bioabsorbable felt against alveolar air leakage. Eur J Cardiothorac Surg 2008;33:457-60. [Crossref] [PubMed]
- Xu K, Bian W, Xie H, et al. Single-port video-assisted thoracoscopic wedge resection: novel approaches in different genders. Interact Cardiovasc Thorac Surg 2016;23:202-7. [Crossref] [PubMed]
- Lin JB, Chen JF, Lai FC, et al. Transareolar pulmonary bullectomy for primary spontaneous pneumothorax. J Thorac Cardiovasc Surg 2016;152:999-1005. [Crossref] [PubMed]
- Chen JF, Lin JB, Tu YR, et al. Nonintubated transareolar single-port thoracic sympathicotomy with a needle scope in a series of 85 male patients. Surg Endosc 2016;30:3447-53. [Crossref] [PubMed]


