Bilateral multiple intrapulmonary nodules: a puzzled and rare disease
Introduction
Primary intrapulmonary thymomas (PIT) are defined as a very rare tumor exists in the lung without any associated mediastinal components. However primary intrapulmonary spindle cell thymomas (PISCT) are even rare. Only five cases of solitary PISCT had been reported in literature (1-5). However no case of bilateral multiple PISCT was reported. We reported the first case and hope to provide helpful experience.
Case report
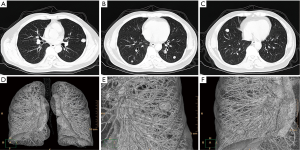
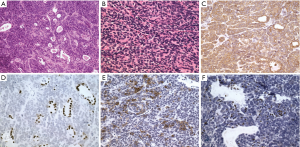
A 62-year-old man presented with bilateral multiple pulmonary nodules which discovered incidentally in 2006. The patient was admitted to our medical center in 2008 for advanced management. He denied any uncomfortable symptom. His past medical history was unremarkable, except as quarrier for 10 years in his 20th age. Chest X-ray film and computed tomography confirmed multiple intrapulmonary nodules without any mediastinal lesions (Figure 1). Bilateral video assisted thoracic surgery was performed and all nodules were excised. Frozen section revealed possible low-grade malignancy. Final pathological diagnoses was PISCT and type A in WHO classification. Histologically, the nodules were consisted of benign-appearing cell population of oval and spindle-shaped epithelial cells lacking nuclear atypia and accompanied by very few identified lymphocytes (Figure 2A,B). Immunohistochemical stains demonstrated tumor cells were strong positive for cytokeratin (Figure 2C). The tumor cells were negative for TTF1 and the pulmonary alveolar epithelial cells were mild positive for TTF1 (Figure 2D). Partial tumor cells were positive for CD20 (Figure 2E) and very few lymphocytes in the interstitial area were TdT positive (Figure 2F). The recovery was uneventful and the patient was discharged on the seventh postoperative day. Adjuvant radiotherapy was recommended, however the patient refused to receive any treatment. With 6 years follow-up, the computed tomography confirmed no abnormal nodules of recurrence in the lung. Final status exam also demonstrated no lesions detected in other organs. The patient feels comfortable and fortunately remains alive with being free of recurrence till now.
Discussion
Since McBurnery reported PIT in 1951, few articles about PIT cases had been published (1). Only five solitary PISCT cases were reported. PIT was predominantly solitary and less multiple. Only two cases were multifocal but in the ipsilateral lobe (6). For distribution, a vast majority of PIT is peripheral and very few near hilum. Most solitary lesion is distributed in the right lung with preferences of upper lobe. Similar with PIT, a majority of PISCT for distribution is peripheral and very few near hilum. Most solitary lesion is distributed with preferences of right lung (1-5).
The pathogenesis and origin of PISCT continues to be a subject of speculation. The most frequently invoked theory is embryological displacement of thymus into ectopic lung. Any errors occurring during descending phase of primordial thymus can cause dissemination of aberrant intrapulmonary nodules. Another school of speculation suggests that ectopic thymus arise from uncommitted germinative stem cells capable of differentiating along a variety of lines. This theory may explain the occurrence corresponding to well-defined cells in the lung (3).
The majority of PIT remains asymptomatic more commonly. Most lesions were discovered incidentally on chest roentgenogram. When symptomatic, chief complaints included hemoptysis, recurrent pneumonia and chest pain. Compared with mediastinal thymoma, myasthenia gravis, red cell aplasia and other paraneoplastic syndromes involving PIT are rare. Among five reported cases of PISCT, the lesion in four cases was also discovered incidentally on chest roentgenogram, except for one case with symptoms of common cough and expectoration. In addition, the patient in our case also did not present any symptom when the lesions were identified. Most PISCT remains asymptomatic more commonly just as PIT.
PISCT may pose a serious challenge of pathological diagnosis. Immunohistochemical stains combined with microscopic pattern of the tumor and clinical documents are helpful in differentiation. The spindle cell growth pattern of PISCT probably accounts for the majority of errors in diagnosis because it resembles to a variety of soft tissue sarcomas (1). As our case, PISCT always presents bland appearance of oval and spindle-shaped epithelial cells lacking mitotic activity, very few lymphoid cell components scattered. This alerts pathologists to the possibility of PISCT. Immunohistochemical stains will aid in this context by showing cytokeratin positivity in spindle cells. Characterization of few lymphocytic components with markers for immature T-lymphocytes, such as TdT, may also be useful in diagnosis (1). Just as mediastinal thymoma, histopathologic classification of PIT was done according to the WHO classification after review of histology and immunohistochemistry micrographs and descriptions. PIT was distributed between different forms of the WHO classification of thymic epithelial tumors. Of note, there were no primary intrapulmonary thymic carcinomas (type C). PISCT is the type A of PIT. Like mediastinal thymoma, PISCT was considered as benign forms of thymoma without metastasis. In literatures, metastasis of PISCT also was not reported. In our case all preoperative examinations could not identified the evidence of primary mediastinal tumors. In addition, nodules in bilateral lungs were resected and diagnosed as PISCT for exclusion of metastasis.
Up to date, it still seems difficult to reach conclusions of standard treatment and prognosis on PISCT because of limited data available due to its rarity. So more treatment methods were referred from the management of PIT and personal experience. Complete surgical resection is considered as optimal choice of treatment in PIT. According to six cases of PISCT, one patient received lobectomy only, three patients received lobectomy with lymph nodes dissection, and one patient received wedge resection. However in our cases, the patient received bilateral wedge section due to its multiple lesions. Adjuvant chemotherapy is considered not effective, however adjuvant radiotherapy should be recommended if incomplete resection or extension of thymic tissue beyond a circumscribed capsule is noted in PIT (2-5). No patients in PISCT cases underwent neoadjuvant treatment. Two patients, one wedge resection and one lobectomy combined with lymph nodes dissection, received adjuvant radiotherapy after surgery. Other four cases did not received any adjuvant treatment, but follow-up. All six patients were followed up from 6 months to 6 years separately and no recurrence was found.
In the unique case of bilateral multiple PISCT after 6-year follow-up, good prognosis of this patient with complete resection without adjuvant therapy provided new idea and possibly useful treat experience for multiple PISCT.
Conclusions
PISCT are very rare, even no bilateral multiple lesions and associated treatment had been reported. We reported this first unique case of bilateral multiple PISCT and provided useful experience for readers. For advanced establishment of standard treatment, more cases of multiple PISCT should be still needed and analyzed.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Moran CA, Suster S, Fishback NF, et al. Primary intrapulmonary thymoma. A clinicopathologic and immunohistochemical study of eight cases. Am J Surg Pathol 1995;19:304-12. [PubMed]
- Gong L, Li YH, He XL, et al. Primary intrapulmonary thymomas: case report and review of the literature. J Int Med Res 2009;37:1252-7. [PubMed]
- Srivastava A, Padilla O, Alroy J, et al. Primary intrapulmonary spindle cell thymoma with marked granulomatous reaction: report of a case with review of literature. Int J Surg Pathol 2003;11:353-6. [PubMed]
- Kiriş I, Karahan N, Peker O, et al. A case of spindle cell primary intrapulmonary thymoma: a rare histologic subtype. Turk J Thorac Cardiovasc Surg 2005;13:412-15.
- Katsura M, Kouso H, Shikada Y, et al. Primary intrapulmonary thymoma. Gen Thorac Cardiovasc Surg 2015;63:56-9. [PubMed]
- Myers PO, Kritikos N, Bongiovanni M, et al. Primary intrapulmonary thymoma: a systematic review. Eur J Surg Oncol 2007;33:1137-41. [PubMed]


