The role of SOX-2 on the survival of patients with non-small cell lung cancer
Introduction
Lung cancer remains the most lethal cancer worldwide, despite improvements in diagnostic and therapeutic techniques. Its incidence has not peaked in many parts of world, particularly in China, which has become a major public health challenge all the world (1). The prognosis for lung cancer patients is generally poor, with an overall 5-year survival rate of approximately 15%, and it has shown little improvement in recent decades (2,3). Several independent prognostic factors for survival have been identified: performance status (PS), disease stage, age, sex and amount of weight lost (4). Some of these factors are useful when choosing treatment options for an individual, principally disease stage and PS. However, the discriminant value of most potential prognostic biological markers is insufficient to predict the optimal therapeutic course for an individual (5,6).
SRY (sex determining region Y)-box 2, also known as SOX-2, is one of the key transcriptional factors that control the unique properties of stem cells self-renewal and pluripotency (7,8) and play a critical role inmaintaining the stem cell-like phenotype in cancer cells (9-12). Over-expression of SOX2 in NSCLC cells stimulates cellular migration and anchorage-independent growth while SOX-2 knockdown impairs cell growth (13,14).
Recently, a number of studies have reported the contribution of SOX-2 to tumorigenesis and its correlation with clinical progression of various types of tumors, including lung cancer. However, no consensus has been reached; conflicting results have been reported from different laboratories. We therefore carried out a meta-analysis of data from published studies to quantitatively review the effect of SOX-2 overexpression in tumor tissue on survival in patients with non-small-cell lung cancer (NSCLC).
Materials and methods
Search strategy and study selection
The electronic databases PubMed, Embase, and CNKI (China National Knowledge Infrastructure) were searched for studies to include in our meta-analysis. An upper date limit of Nov 01, 2014 was applied; we used no lower date limit. Searches included the terms “lung cancer”, “SOX-2”, “SRY-box 2,”, and “prognosis”. We also reviewed the Cochrane Library for relevant articles. The references reported in the identified studies were also used to complete the search.
Studies eligible for inclusion in this meta-analysis met the following criteria: (I) measure SOX-2 expression in the primary lung cancer tissue with IHC (immunohistochemistry) or other methods; (II) provide information on survival (studies investigating response rates only were excluded); (III) have a follow-up time exceeding 5 years; and (IV) When the same author reported results obtained from the same patient population in more than one publication, only the most recent report, or the most complete one, was included in the analysis. Two reviewers independently determined study eligibility. Disagreements were resolved by consensus.
Data extraction and quality assessment
The final articles included were assessed independently by two reviewers. Data retrieved from the reports included first author, publication year, patient source, histology, disease stage, test method, SOX-2 positive and survival data (Table 1). If data from any of the above categories were not reported in the primary study, items were treated as “not applicable”. We did not contact the author of the primary study to request the information.

Full table
Statistical methods
For the quantitative aggregation of the survival results, hazard ratios (HR) and their 95% confidence intervals (CIs) were combined to give the effective value. When these statistical variables were not given explicitly in an article, they were calculated from available numerical data using methods reported by Parmar et al. (15).
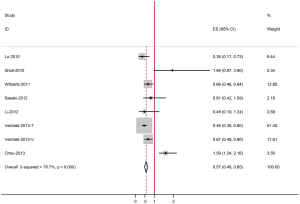
Heterogeneity of the individual HRs was calculated with Chi-squared tests according to Peto’s method (16). Meanwhile, Heterogeneity test with I2 statistic and Q statistic was performed. All the studies included were categorized by patient race, histology, disease stage. Individual meta-analysis was conducted in each subgroup. If HRs were found to have fine homogeneity, a fixed effect model was used for secondary analysis; if not, a random-effect model was used. In this meta-analysis, DerSimonian-Laird random effects analysis (17) was used to estimate the effect of SOX-2 overexpression on survival. By convention, an observed HR >1 implies worse survival for the group with SOX-2 overexpression. The impact of TTF-1 on survival was considered to be statistically significant if the 95% CI did not overlap with 1. Horizontal lines represent 95% CIs. Each box represents the HR point estimate, and its area is proportional to the weight of the study. The diamond (and broken line) represents the overall summary estimate, with CI represented by its width. The unbroken vertical line is set at the null value (HR =1.0).
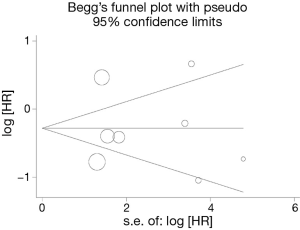
Evidence of publication bias was sought using the methods of Egger et al. (18) and of Begg et al. (19). Moreover, contour-enhanced funnel plot (20) was performed to aid in interpreting the funnel plot. If studies appear to be missing in areas of low statistical significance, then it is possible that the asymmetry is due to publication bias. If studies appear to be missing in areas of high statistical significance, then publication bias is a less likely cause of the funnel asymmetry. Intercept significance was determined by the t-test suggested by Egger (P<0.05 was considered representative of statistically significant publication bias). All calculations were performed using STATA version 11.0 (Stata Corporation, College Station, TX).
Results
Study selection and characteristics
Seven studies (21-27) published between 2010 and 2013 were eligible for this systematic review with meta-analysis. All reported the prognostic value of SOX-2 status for survival in NSCLC patients. The total number of patients included was 1,944, ranging from 44 to 758 patients per study (median 277). The major characteristics of the seven eligible publications are reported in Table 1.
These publications followed several different patient cohorts. Among the seven studies evaluating SOX-2 expression in NSCLC, three studies were performed in Asian populations, and the remaining four studies followed European or American patients. Only one of the 17 studies identified SOX-2 overexpression as an indicator of poor prognosis, two studies showed no statistically significant impact of SOX-2 overexpression on survival, and the other five studies showed for favorable significance.
Meta-analysis
The results of the meta-analysis are reported in Figure 1. Overall, the combined HR for all eligible studies evaluated SOX-2 expression in NSCLC was 0.57 (95% CI: 0.48 to 0.65), indicating that SOX-2 overexpression was an indicator of favorable prognosis for NSCLC patients. However, highly significant heterogeneity was detected among these studies (I2=76.7%, P=0.000).
Publication bias
Begg’s funnel plot and Egger’s test were performed to assess the publication bias in the literature. All seven eligible studies investigating NSCLC patients yielded a Begg’s test score of P=0.654 and an Egger’s test score of P=0.976, meanwhile according to the contour-enhanced funnel plot (Figure 2), the absence of publication bias was found in all seven studies. These results suggest that there is no publication bias at work.
Discussion
NSCLC is the leading cause of cancer death, with an overall fiveyear survival rate of less than 15% (1-3). New biological markers of NSCLC carcinogenesis may provide important progress in clinical decision making (3). Emerging evidences have suggested functional molecules involved in cell-cycle control, DNA repair, proliferation, apoptosis that may modulate response to platinum-based chemotherapy and serve as promising biomarkers for individualized chemotherapy and prognosis of NSCLC patients (28).
SOX2 expression plays a critical role incell cycle control, DNA damage response and long-term self-renewal in neural stem cells (29,30). Moreover, several studies have identified that SOX2 expression correlated with tumorigenesis, chemoresistance, and maintaining the stem cell-like phenotype in cancer cells (31). In the present meta-analysis, we have combined seven published studies including 1,944 patients with NSCLC to yield summary statistics that indicate that SOX-2 overexpression has a significant correlation with favorable survival in NSCLC. We observed a statistically significant effect of SOX-2 on favorable survival, suggesting that this good prognostic factor could be of importance not only in early-stage NSCLC but also in advanced staged NSCLC.
Recently, several systematic reviews (32-40) with meta-analyses on other biological prognostic factors for NSCLC had been reported. P53, microvessel density, HER-2, Ki-67 and RAS might be poor prognostic factors for survival in NSCLC, however, Bcl-2 might be better prognostic factor for survival in NSCLC. In order to clarify the prognostic impact of other biological factors in lung cancer, our group has performed several systematic reviews of the literature with meta-analyses. We found that VEGF (41), E-cadherin (42) and matrix metalloproteinase 2 (43) might be poor prognostic factor in NSCLC, COX-2 (44) might be poor prognostic factor for stage I NSCLC, the ground glass opacity (GGO) area (45) had a favorable prognostic value of overall survival and relapse-free survival in small lung adenocarcinoma.
Another potential source of bias is related to the method of HR and 95% CI extrapolation. If these statistics were not reported by the authors, we calculated them from the data available in the article. If this was not possible, we extrapolated them from the survival curves, necessarily making assumptions about the censoring process. Data for multivariate survival analysis reported in the article were included in the present systematic review with meta-analysis; if these data were not available, data calculated from survival curves by univariate analysis were included. These results should be confirmed by an adequately designed prospective study. Furthermore, the exact value of SOX-2 overexpression status needs to be determined by appropriate multivariate analysis. Unfortunately, few prospectively designed prognostic studies concerning biomarkers have been reported; thus, our collection of many retrospective studies revealed more significance.
Publication bias (46) is a major concern for all forms of meta-analysis; positive results tend to be accepted by journals, while negative results are often rejected or not even submitted. The present analysis does not support publication bias; the obtained summary statistics likely approximate the actual average. However, it should be noted that our meta-analysis could not completely exclude biases. For example, the study was restricted to papers published in English and Chinese, which probably introduced bias.
In conclusion, SOX-2 overexpression is associated with a favorable prognosis in patients with NSCLC in present meta-analysis, but there is a high heterogeneity between the studies. These results should be confirmed by an adequately designed prospective study.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Chen W, Zheng R, Zhang S, et al. Report of cancer incidence and mortality in China, 2010. Ann Transl Med 2014;2:61. [PubMed]
- Zhou C. Lung cancer molecular epidemiology in China: recent trends. Transl Lung Cancer Res 2014;3:270-9. [PubMed]
- Alberg AJ, Ford JG, Samet JM, et al. Epidemiology of lung cancer: ACCP evidence-based clinical practice guidelines (2nd edition). Chest 2007;132:29S-55S.
- Palmer JD, Zaorsky NG, Witek M, et al. Molecular markers to predict clinical outcome and radiation induced toxicity in lung cancer. J Thorac Dis 2014;6:387-98. [PubMed]
- Okada M, Mimae T, Tsutani Y, et al. Segmentectomy versus lobectomy for clinical stage IA lung adenocarcinoma. Ann Cardiothorac Surg 2014;3:153-9. [PubMed]
- Lazarus MA, Schachter L, Xavier M. Social factors, treatment, and survival in patients with advanced-stage non-small cell lung cancer. Transl Cancer Res 2014;3:146-51.
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006;126:663-76. [PubMed]
- Masui S, Nakatake Y, Toyooka Y, et al. Pluripotency governed by Sox2 via regulation of Oct3/4 expression in mouse embryonic stem cells. Nat Cell Biol 2007;9:625-35. [PubMed]
- Yuan P, Kadara H, Behrens C, et al. Sex determining region Y-Box 2 (SOX2) is a potential cell-lineage gene highly expressed in the pathogenesis of squamous cell carcinomas of the lung. PLoS One 2010;5:e9112. [PubMed]
- Gontan C, de Munck A, Vermeij M, et al. Sox2 is important for two crucial processes in lung development: branching morphogenesis and epithelial cell differentiation. Dev Biol 2008;317:296-309. [PubMed]
- Singh S, Trevino J, Bora-Singhal N, et al. EGFR/Src/Akt signaling modulates Sox2 expression and self-renewal of stem-like side-population cells in non-small cell lung cancer. Mol Cancer 2012;11:73. [PubMed]
- Hussenet T, du Manoir S. SOX2 in squamous cell carcinoma: amplifying a pleiotropic oncogene along carcinogenesis. Cell Cycle 2010;9:1480-6. [PubMed]
- Hussenet T, Dali S, Exinger J, et al. SOX2 is an oncogene activated by recurrent 3q26.3 amplifications in human lung squamous cell carcinomas. PLoS One 2010;5:e8960. [PubMed]
- Bass AJ, Watanabe H, Mermel CH, et al. SOX2 is an amplified lineage-survival oncogene in lung and esophageal squamous cell carcinomas. Nat Genet 2009;41:1238-42. [PubMed]
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med 1998;17:2815-34. [PubMed]
- Yusuf S, Peto R, Lewis J, et al. Beta blockade during and after myocardial infarction: an overview of the randomized trials. Prog Cardiovasc Dis 1985;27:335-71. [PubMed]
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7:177-88. [PubMed]
- Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315:629-34. [PubMed]
- Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994;50:1088-101. [PubMed]
- Palmer TM, Peters JL, Sutton AJ, et al. Contour enhanced funnel plots for meta-analysis. Stata J 2008;8:242-54.
- Lu Y, Futtner C, Rock JR, et al. Evidence that SOX2 overexpression is oncogenic in the lung. PLoS One 2010;5:e11022. [PubMed]
- Sholl LM, Barletta JA, Yeap BY, et al. Sox2 protein expression is an independent poor prognostic indicator in stage I lung adenocarcinoma. Am J Surg Pathol 2010;34:1193-8. [PubMed]
- Wilbertz T, Wagner P, Petersen K, et al. SOX2 gene amplification and protein overexpression are associated with better outcome in squamous cell lung cancer. Mod Pathol 2011;24:944-53. [PubMed]
- Sasaki H, Yokota K, Hikosaka Y, et al. Increased Sox2 copy number in lung squamous cell carcinomas. Exp Ther Med 2012;3:44-48. [PubMed]
- Li X, Wang J, Xu Z, et al. Expression of Sox2 and Oct4 and their clinical significance in human non-small-cell lung cancer. Int J Mol Sci 2012;13:7663-75. [PubMed]
- Velcheti V, Schalper K, Yao X, et al. High SOX2 levels predict better outcome in non-small cell lung carcinomas. PLoS One 2013;8:e61427. [PubMed]
- Chou YT, Lee CC, Hsiao SH, et al. The emerging role of SOX2 in cell proliferation and survival and its crosstalk with oncogenic signaling in lung cancer. Stem Cells 2013;31:2607-19. [PubMed]
- Wang LE, Yin M, Dong Q, et al. DNA repair capacity in peripheral lymphocytes predicts survival of patients with non-small-cell lung cancer treated with first-line platinum-based chemotherapy. J Clin Oncol 2011;29:4121-8. [PubMed]
- Marqués-Torrejón MÁ, Porlan E, Banito A, et al. Cyclin-dependent kinase inhibitor p21 controls adult neural stem cell expansion by regulating Sox2 gene expression. Cell Stem Cell 2013;12:88-100. [PubMed]
- Peng C, Li N, Ng YK, et al. A unilateral negative feedback loop between miR-200 microRNAs and Sox2/E2F3 controls neural progenitor cell-cycle exit and differentiation. J Neurosci 2012;32:13292-308. [PubMed]
- Tian T, Zhang Y, Wang S, et al. Sox2 enhances the tumorigenicity and chemoresistance of cancer stem-like cells derived from gastric cancer. J Biomed Res 2012;26:336-45. [PubMed]
- Steels E, Paesmans M, Berghmans T, et al. Role of p53 as a prognostic factor for survival in lung cancer: a systematic review of the literature with a meta-analysis. Eur Respir J 2001;18:705-19. [PubMed]
- Meert AP, Paesmans M, Martin B, et al. The role of microvessel density on the survival of patients with lung cancer: a systematic review of the literature with meta-analysis. Br J Cancer 2002;87:694-701. [PubMed]
- Meert AP, Martin B, Paesmans M, et al. The role of HER-2/neu expression on the survival of patients with lung cancer: a systematic review of the literature. Br J Cancer 2003;89:959-965. [PubMed]
- Martin B, Paesmans M, Berghmans T, et al. Role of Bcl-2 as a prognostic factor for survival in lung cancer: a systematic review of the literature with meta-analysis. Br J Cancer 2003;89:55-64. [PubMed]
- Martin B, Paesmans M, Mascaux C, et al. Ki-67 expression and patients survival in lung cancer: systematic review of the literature with meta-analysis. Br J Cancer 2004;91:2018-25. [PubMed]
- Mascaux C, Iannino N, Martin B, et al. The role of RAS oncogene in survival of patients with lung cancer: a systematic review of the literature with meta-analysis. Br J Cancer 2005;92:131-9. [PubMed]
- Meert AP, Martin B, Delmotte P, et al. The role of EGF-R expression on patient survival in lung cancer: a systematic review with meta-analysis. Eur Respir J 2002;20:975-81. [PubMed]
- Nakamura H, Kawasaki N, Taguchi M, et al. Survival impact of epidermal growth factor receptor overexpression in patients with non-small cell lung cancer: a meta-analysis. Thorax 2006;61:140-5. [PubMed]
- Fan J, Wang L, Jiang GN, et al. The role of survivin on overall survival of non-small cell lung cancer, a meta-analysis of published literatures. Lung Cancer 2008;61:91-6. [PubMed]
- Zhan P, Wang J, Lv XJ, et al. Prognostic value of vascular endothelial growth factor expression in patients with lung cancer: a systematic review with meta-analysis. J Thorac Oncol 2009;4:1094-103. [PubMed]
- Wu Y, Liu HB, Ding M, et al. The impact of E-cadherin expression on non-small cell lung cancer survival: a meta-analysis. Mol Biol Rep 2012;39:9621-8. [PubMed]
- Qian Q, Wang Q, Zhan P, et al. The role of matrix metalloproteinase 2 on the survival of patients with non-small cell lung cancer: a systematic review with meta-analysis. Cancer Invest 2010;28:661-9. [PubMed]
- Zhan P, Qian Q, Yu LK. Prognostic value of COX-2 expression in patients with non-small cell lung cancer: a systematic review and meta-analysis. J Thorac Dis 2013;5:40-7. [PubMed]
- Aggarwal C. Targeted therapy for lung cancer: present and future. Ann Palliat Med 2014;3:229-35. [PubMed]
- Begg CB, Berlin JA. Publication Bias: A Problem in Interpreting Medical Data. J R Stat Soc A 1988;151:419-63.


