Results of the introduction of a minimally invasive esophagectomy program in a tertiary referral center
Abstract
Background: Esophagectomy is accompanied by a high postoperative complication rate. Minimally invasive esophageal surgery appears to be a promising technique that might be associated with a lower pulmonary morbidity rate. The objective of this study was to describe the implementation of minimally invasive esophageal surgery in a tertiary referral center and to compare the results of our first series of minimally invasive esophagectomies (MIE) to conventional open esophagectomies.
Methods: MIE was implemented after several procedures had been proctored by a surgeon with extensive experience with MIE. Preoperative characteristics and the postoperative course of patients who underwent a transthoracic esophagectomy were prospectively registered. Morbidity and overall hospital stay were compared between minimally invasive and open resections performed in the same period.
Results: A total of 90 consecutive esophageal cancer patients underwent a transthoracic resection, 41 patients by means of a minimally invasive approach. Preoperative characteristics were comparable for both groups. The duration of surgery was longer in the MIE group (6.0 vs. 5.2 hours, P<0.001) and median blood loss was lower [100 vs. 500 mL (P<0.001)]. There was only a trend towards a shorter hospital stay in the MIE group (11 vs. 13 days, P=0.072), pulmonary complications occurred in 20% of patients in the MIE group vs. 31% in the open group (P=0.229). The overall complication rate was 51% in the MIE group vs. 63% in the open group, P=0.249.
Conclusions: Implementation of MIE in our center was successful and it appears to be a safe technique for patients with potentially curable esophageal carcinoma.
Key words: Esophageal cancer; surgical technical; complications; thoracoscopy
Introduction
For patients with esophageal carcinoma, surgical resection is the most important element of a potentially curative treatment (1,2). A substantial proportion of patients who undergo esophageal surgery will develop postoperative complications. Early postoperative complication rates vary between 40% and 80%, depending on the applied criteria and the extent of surgery (3,4). Transthoracic resections in particular are associated with an increased risk of developing complications; i.e. more than 50% of patients who undergo a transthoracic esophagectomy will develop pulmonary complications (1). In general, quality of life after esophageal surgery will not return to the preoperative level until at least six months postoperatively. In this type of patients, with an overall five-year survival rate of 40%, it is essential to ameliorate the postoperative course (1).
Minimally invasive esophageal surgery was introduced in an attempt to diminish postoperative complication rates and thereby reduce overall in-hospital stay. It was first described by DePaula et al. in 1995 (5) and since then, an increasing number of centers have been using minimally invasive techniques for esophagectomy. Previous studies on minimally invasive esophagectomies (MIE) have shown pulmonary complication rates reduced to as low as 12% (6,7).
Our tertiary referral center for esophageal cancer already had extensive experience in advanced laparoscopic gastrointestinal surgical procedures, mainly in the field of colorectal surgery and benign upper gastrointestinal surgery, but esophagectomies were performed by means of open procedures only. Therefore, a program was initiated in which MIE was introduced in our center. The aim of this study was to describe the implementation of MIE, and to compare morbidity and hospital stay between our first series of MIE and open esophagectomies.Materials and methods
Implementation
Table 1 described the implementation of MIE in the Academic Medical Center (AMC). The first step in the implementation of MIE was a hands on MIE course. Subsequently, a other centers that already routinely performed this technique were visited [Catharina Hospital Eindhoven and Free University Medical Center (VUMC)] to evaluate the procedures performed in these centers and decide on setup, materials and type of procedure in the AMC. Six of the first complete minimally invasive procedures were proctored by a surgeon with extensive experience in the field of MIE (MAC). The choice of technique was based on the experience in open esophageal surgery which was the McKeown technique; therefore MIE was performed by means of a thoracolaparoscopic McKeown in prone position. The surgeon performing all minimally invasive procedures (MIvBH) had extensive experience in esophageal surgery (i.e. >300 procedures) and furthermore was trained in advanced upper GI procedures (Heller’s myotomy, fundoplications) and minimally invasive colorectal procedures. The first 10 procedures were proctored. Furthermore, among the first non proctored minimally invasive procedures, several procedures were a combination of thoracoscopy and laparotomy instead of a complete minimally invasive resection. The reason for this was the assumption that the implementation of this new technique would lead to an increased duration of surgery; to limit this, a small number of procedures in the initial phase were only partially performed minimally invasive.
Full Table
Patients
All patients who underwent a transthoracic resection between October 2009 and February 2011 were included in this study. Patients were selected for a MIE in case of a BMI <30 kg/m2, no history of thoracic or upper gastrointestinal surgery and MIE was performed dependent to the availability of the MIE-trained surgeon. Clinicopathological characteristics, surgical characteristics, duration of hospital stay and postoperative complications were registered in a prospectively monitored database. Patients were divided into two groups; patients who underwent a minimally invasive transthoracic esophagectomy and those who underwent a conventional open transthoracic resection. All patients with a TNM-stage larger than T1N0 according to the 6th edition of the American Joint Committee on Cancer (AJCC) (8) were treated with neoadjuvant chemoradiotherapy (carboplatin and paclitaxel combined with 41.4 Gy). Surgery was performed 6 weeks after completion of neoadjuvant therapy. Postoperative care was standardized in both groups and included early mobilization starting on postoperative day 1 and early start of clear liquid and solid diet. Postoperative complications including anastomotic leakage, chylous leakage, and pulmonary complications (pneumonia indicated by a positive sputum culture combined with a new or progressive infiltrate on a chest X-ray and atelectasis indicated by lobar collapse on a chest X-ray) were closely monitored. In addition, the severity of postoperative complications was graded according to the classification system composed by Dindo et al. This system is based on the therapeutic consequences of complications and consists of five grades. Grade I complications do not require any intervention, grade II complications require pharmacological treatment, grade III complications need radiological (IIIa) or surgical (IIIb) intervention and grade IV complications are life-threatening and indicate single-organ (grade IVa) or multiorgan (gradeIVb) dysfunction. Grade V complications lead to death. In previous studies performed at our institute, hree categories of complications were defined; no complications (category 0), minor to moderate complications (category 1, grade I to IIIb), and severe complications (category 2, grade IVa, IVb and V). Discharge criteria were defined as adequate pain control with oral analgesics, no nausea, adequate intake, passage of stool, mobilization and self support.
A proportion of patients was included in a randomized controlled trial evaluating the morbidity rate after esophageal surgery for cancer in patients undergoing MIE compared to open surgery (9).
All patients who underwent minimally invasive surgery gave their informed consent.
Surgery
Thoracolaparoscopy
Patients were placed in prone position and single lung ventilation was achieved by a bronchus blocker (EZ blocker, AnestheticIQ, IQ Medical Ventures, Delft, NL) if necessary. During most procedures, a pressure of 6 cmH2O was sufficient for optimal vision without the use of a bronchus blocker. Four ports were introduced into the chest. The pleura overlying the esophagus was divided as well as the azygos vein. The thoracic duct was clipped and the esophagus was mobilized up to the level of the thoracic inlet. The subcarinal and the right paratracheal lymph nodes were removed en block. After placement of a chest tube, the thoracic phase was completed. Subsequently, patients were rotated to a supine position and five abdominal ports were inserted. The greater curvature of the stomach was mobilized with preservation of the right gastroepiploic artery. Lymph nodes along the celiac axis were dissected. After division of the esophagus in the neck (left side) the specimen was externalized through a 4-5 cm mini laparotomy through which the gastric conduit was constructed. Subsequently, a single layer hand sewn an end-to-end or end-to-side anastomosis was performed in the neck. Finally, a feeding jejunostomy was placed.
Conventional open transthoracic esophagectomy
Conventional open transthoracic resections including a two-field lymphadenectomy were performed according to a previous publication by our center (1). In short, posterolateral thoracotomy was the first step in transthoracic resection with extended en bloc lymphadenectomy. The thoracic duct, azygos vein, ipsilateral pleura, and all peri-esophageal tissue in the posterior mediastinum were dissected en bloc. The specimen included the lower and middle mediastinal, subcarinal, and right-sided paratracheal lymph nodes. Aortopulmonary window nodes were dissected separately. During the abdominal phase, the paracardial, lesser curvature, left gastric artery, celiac trunk, common hepatic artery and splenic artery nodes were dissected. The gastric conduit was constructed after which a single layer hand sewn an end-to-end or end-to-side anastomosis was performed in the left side of the neck.
Statistical analysis
Patients were divided into two groups based on the type of surgery; minimally invasive or conventional open. Data were compared between both groups. To compare proportions the chi-square or Fisher’s exact test was performed. Differences between groups were tested using the Mann-Whitney U test for continuous data. A two-sided p-value of <0.05 was considered statistically significant. Statistical analysis was performed with PASW® for Windows version 18 (SPSS, Chicago, IL, USA).
Results
Between October 2009 and February 2011 a total of 90 patients underwent a transthoracic esophagectomy for carcinoma, 41 patients by means of a minimally invasive procedure. Six instead of all of the first 10 procedures that were performed entirely minimally invasive, were proctored by a surgeon with extensive experience in the field of MIE. After every procedure, evaluation took place and after six procedures both the proctor and the principal surgeon agreed that further proctoring was not necessary.
The number of patients included in the TIME trial was 18 in the open group (44%) versus 17 in the MIE groups (35%). The majority of patients in both groups was male with a median age around 64 years. There were no significant differences in ASA-classification, BMI or medical history between patients who underwent a MIE or conventional open transthoracic esophagectomy (TTE). Most patients in both groups were diagnosed with adenocarcinoma of the esophagus (65% open TTE vs. 63% MIE) and more than 90% of all patients was treated with neoadjuvant chemoradiation (Table 2). Reasons for surgery without preoperative chemoradiotherapy were stage I disease (n=2), and previous parasternal radiotherapy (n=3).
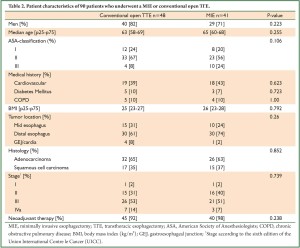
Full Table
Of all minimally invasive esophagectomies, 7 procedures (17%) were a combination of thoracoscopy and laparotomy. All other minimally invasive procedures were entirely thoracolaparoscopic. The conversion rate was 5% (2 patients); pulmonary adhesions were the reason for conversion in both patients. The median duration of surgery was significantly longer in the MIE group with 6 vs. 5.2 hours (P<0.001), but median peroperative blood loss was lower in the MIE group (100 vs. 500 mL, P<0.001). The lymph node harvest in the open group was 25 vs. 19 in the MIE group (P<0.001). More than 90% of patients in both groups underwent a microscopically radical resection (92% vs. 98% in the MIE group, P=NS) (Table 3).

Full Table
The MIE group was subdivided into four groups of 10 patients for the purpose of an impression of the learning curve (Table 4). Patients who underwent surgery in the earliest period were included in group 1. The duration of surgery increased after the first 10 procedures; most likely this is due to the fact that the first group was proctored by an experienced surgeon and that the abdominal part in 7 of these 10 patients was performed by means of a laparotomy instead of a laparoscopy. After the second group of 10 patients, the duration decreased up to the level of the open procedures.
Full Table
The median lymph node harvest in the first 10 patients who underwent MIE was 14; after the first 10 patients the median lymph node harvest increased up to 23, which was comparable to the median harvest of 25 nodes in the open group.
The postoperative course is described in Table 5. Primary in-hospital stay after a MIE was 11 days versus 13 days in patients who underwent an open TTE (P=0.072). Although not significant the complication rate in the MIE group was more than 10% less than in the open TTE group (51% vs. 63%, P=NS), as well as the pulmonary complication rate (20% vs. 31%, P=NS). When postoperative complications were categorized according to the Dindo classification, there were no significant differences between patients who underwent an open or minimally invasive esophagectomy (P=0.486). The percentage of patients with severe complications was 24% versus 17% in the open and MIE groups respectively.
Full Table
Discussion
The results of this study suggest that the morbidity after a minimally invasive esophagectomy or a conventional open transthoracic esophagectomy is at least comparable in the start-up phase of this project on minimally invasive esophageal surgery. Furthermore, we found a trend towards a shorter duration of hospital stay after minimally invasive surgery.
A successful implementation of a minimally invasive technique implies that the postoperative morbidity and mortality are similar, if not improved compared to open procedures. In absolute figures, there was a favorable outcome in the MIE patients in the present series regarding the morbidity rate, in particular the overall and pulmonary complication rate, although this did not reach statistical significance. This is probably due to the small numbers of patients in both groups. A lower pulmonary complication rate is in accordance with results from other studies that have compared MIE and open esophagectomies (10,11). The possibility that respiratory complications, especially pneumonia, are less common after MIE is likely the consequences of the avoidance of thoracotomy. Moreover, MIE in prone position allows for ventilation and oxygenation of the lung which reduces or prevents arteriovenous shunting with a better preserved oxygenation (7).
In-hospital stay after MIE as reported in the literature varies between 6 and 20 days (6,7,10-14); and in case of a comparison between MIE and open esophageal resections, in-hospital stay has been shown to be shorter after MIE which is in accordance with our results (10-12,14). Furthermore, in the present study in-hospital mortality was only 2% in the MIE group which is similar to other high-volume esophagectomy centers (6,11,13).
The oncologic outcome after MIE should be comparable to the outcome after open esophagectomies. Although it was impossible to evaluate long term survival in the present study, the proportion of patients with a microscopically radical resection in the MIE group was similar to that in the conventional open TTE group. The overall median lymph node harvest was significantly lower in the MIE group, however, the number of harvested nodes increased after the first 10 resections and was similar to that of the median number of resected lymph nodes in the 30 patients that underwent surgery most recently during the learning curve. The significant difference in lymph node harvest between the MIE and open group is most likely based on the median harvest of 14 lymph nodes in the first 10 patients. Unfortunately, we could not find an explanation for this difference, however the quality and technique especially in this group were guaranteed since most of these procedures were proctored. The current AJCC manual recommends a lymph node harvest of 12-22 nodes (15), which was achieved in all four MIE groups of the learning curve in the present series. The percentage of microscopically radical resections in the MIE group was comparable with the R0 rate in the open group. Therefore, the oncologic outcome after MIE in the present study is acceptable and comparable to the results of conventional open procedures. Long-term survival could not yet be determined due to the current insufficient duration of follow-up.
With the implementation of a new technique it is inevitable that surgeons go through a learning curve. In minimally invasive esophageal surgery, little is known about the duration of the learning curve. Song et al. evaluated the operative time and postoperative course in 28 patients who underwent a MIE (16). Despite the small cohort, patients were subdivided into two groups; the latter group had a shorter duration of surgery and in-hospital stay, and less postoperative complications. In a series of more than 100 minimally invasive esophagectomies the operative time improved after the first 50 cases (13). The authors attributed the decreased duration of hospital stay to standardization of the steps of the procedure and the surgeon’s improvement in laparoscopic and thoracoscopic skills.
With over 90 esophagectomies performed yearly, our clinic has extensive experience with esophageal surgery. Furthermore, minimally invasive procedures for benign upper gastrointestinal disorders were common practice for the team before MIE was implemented in our center. We believe that the duration of the learning curve is surgeon dependent and that extensive experience with the open esophagectomies and other advanced laparoscopic procedures is the basis for implementing minimally invasive procedure. The choice of MIE technique is dependent on the local situation; in our center the McKeown technique was used in open esophagectomies and therefore MIE was performed according to the same technique. Furthermore, the implementation of a new technique is a team effort; not only the surgeon is involved, but also the anesthesiology staff and the nursing staff.
When considering this study, there are some limitations. The non-randomized design is a shortcoming of the study; however a proportion of patients was included in a randomized clinical trial, which minimizes bias. Due to the small patient numbers it was not possible for the difference in complication rate and pulmonary complications between MIE and conventional open TTE groups to reach statistical significance; nevertheless there was a trend towards a shorter duration of hospital stay after MIE.
The morbidity after a minimally invasive resection or a conventional open transthoracic esophageal resection is comparable. Furthermore, there is a trend towards a shorter hospital stay after minimally invasive esophagectomy. Therefore, the implementation of MIE in our center was successful and MIE appears to be a safe technique for patients with potentially curable esophageal carcinoma. Benefits of MIE have to be proven in a randomized trial.
Acknowledgements
The authors would like to thank Dr. G.A.P. Nieuwenhuijzen for being able to visit his setup of minimally invasive esophageal surgery at the Catharina Hospital, Eindhoven, the Netherlands.
Disclosure: None of the authors have received a research grant. There was no funding or support for this study.
References
- Hulscher JB, van Sandick JW, de Boer AG, et al. Extended transthoracic resection compared with limited transhiatal resection for adenocarcinoma of the esophagus. N Engl J Med 2002;347:1662-9.
- Whooley BP, Law S, Murthy SC, et al. Analysis of reduced death and complication rates after esophageal resection. Ann Surg 2001;233:338-44.
- Hulscher JB, Tijssen JG, Obertop H, et al. Transthoracic versus transhiatal resection for carcinoma of the esophagus: a meta-analysis. Ann Thorac Surg 2001;72:306-13.
- Swisher SG, Deford L, Merriman KW, et al. Effect of operative volume on morbidity, mortality, and hospital use after esophagectomy for cancer J Thorac Cardiovasc Surg 2000;119:1126-32.
- DePaula AL, Hashiba K, Ferreira EA, et al. Laparoscopic transhiatal esophagectomy with esophagogastroplasty. Surg Laparosc Endosc 1995;5:1-5.
- Luketich JD, Alvelo-Rivera M, Buenaventura PO, et al. Minimally invasive esophagectomy: outcomes in 222 patients. Ann Surg 2003;238:486-94; discussion 494-5.
- Palanivelu C, Prakash A, Senthilkumar R, et al. Minimally invasive esophagectomy: thoracoscopic mobilization of the esophagus and mediastinal lymphadenectomy in prone position--experience of 130 patients. J Am Coll Surg 2006;203:7-16.
- Green FL, Page DL, Fleming ID, et al. AJCC Cancer Staging Handbook. Philadelphia: Lippincott Raven Publishers, 2002.
- Biere SS, Maas KW, Bonavina L, et al. Traditional invasive vs. minimally invasive esophagectomy: a multi-center, randomized trial (TIME-trial). BMC Surg 2011;11:2.
- Schoppmann SF, Prager G, Langer FB, et al. Open versus minimally invasive esophagectomy: a single-center case controlled study. Surg Endosc 2010;24:3044-53.
- Zingg U, McQuinn A, DiValentino D, et al. Minimally invasive versus open esophagectomy for patients with esophageal cancer. Ann Thorac Surg 2009;87:911-9.
- Lazzarino AI, Nagpal K, Bottle A, et al. Open versus minimally invasive esophagectomy: trends of utilization and associated outcomes in England. Ann Surg 2010;252:292-8.
- Nguyen NT, Hinojosa MW, Smith BR, et al. Minimally invasive esophagectomy: lessons learned from 104 operations. Ann Surg 2008;248:1081-91.
- Smithers BM, Gotley DC, Martin I, et al. Comparison of the outcomes between open and minimally invasive esophagectomy. Ann Surg 2007;245:232-40.
- Edge SB, Byrd DR, Compton CC, et al. AJCC Cancer Staging Manual. New York: Springer, 2010.
- Song SY, Na KJ, Oh SG, et al. Learning curves of minimally invasive esophageal cancer surgery. Eur J Cardiothorac Surg 2009;35:689-93.


