Severe adenovirus community-acquired pneumonia in immunocompetent adults: chest radiographic and CT findings
Introduction
Adenovirus pneumonia typically is limited to newborns, immunodeficient hosts, and school or military camp populations (1). Severe adenovirus pneumonia has been frequently described in immunocompromised patients. However, with the advances and widespread use of viral screening, adenovirus has been increasingly found to be involved in sporadic cases and outbreaks of severe community-acquired pneumonia (CAP) in healthy adults (2,3).
Bilateral interstitial infiltrates were previously reported as the principal imaging findings in adults with adenovirus pneumonia, regardless of immune status (2,4,5). However, cytolysis, hemorrhagic necrosis, and grossly congested, consolidated lungs are the typical pathological changes seen in adenovirus pneumonia (1). More and more research supports consolidation rather than interstitial infiltrates as the main imaging characteristic of adenovirus pneumonia in immunocompetent adults (3,6-9), though only a few reports have included supporting CT findings (10,11). The purpose of this study was to describe the radiographic and CT findings of severe adenovirus CAP in eight immunocompetent adults.
Methods
Patients and clinical materials
From April 2012 to April 2014, viral screening was carried out for the etiological study of adult severe unexplained CAP at the Emergency Intensive Care Unit of Peking Union Medical College Hospital. Lower respiratory tract specimens of 128 patients were sent to the Institute of Medical Biology of Chinese Academy of Medical Sciences, to test for respiratory viruses. Eight sporadic cases were confirmed as adenovirus infection through sputum (n=2), tracheal aspirates (n=4) and bronchoalveolar lavage fluids (n=2) by means of real-time polymerase chain reaction (PCR) testing. Adenovirus serotyping were also performed (Table 1). HIV serology and microbiological tests including bacterial, fungal or viral cultures for rhinovirus, cytomegalovirus, influenza virus, metapneumovirus, parainfluenza virus, coronavirus, or respiratory syncytial virus were negative at admission in all eight patients. Three cases infected Acinetobacter baumannii during mechanical ventilation in the later phase of hospitalization. Bacterial tests included: (I) sputum specimens for staining and microbiological cultures; (II) urine specimens for Legionella pneumophila and S. pneumonia antigen detection; (III) blood culture; (IV) acute and convalescent serum samples for Chlamydia Pneumoniae, Mycoplasma pneumoniae and Legionella pneumophila antibody titer determination. We reviewed retrospectively the chest imaging manifestations of these patients. The institutional ethics review board approved this retrospective study.

Full table
There were eight patients (male-female ratio, 3:1) with an age range from 22 to 52 years (mean age, 35.9 years) (Table 1). Six patients had no significant underlying disease, while one patient had a history of cerebral infarction and hypertension, and another patient had hypertension, type 2 diabetes and asthma. None of the patients were severely immunocompromised. The major symptoms at presentation were high fever(>39 °C) (n=8), dyspnea (n=8), cough (n=8), ARDS (n=8), septic shock (n=4), hemoptysis (n=4), chest pain (n=3), diarrhea (n=2), myalgias (n=1), and sore throat (n=1). The time interval from illness onset to admission was 1–15 days with a median of 6 days. Acute Physiology and Chronic Health Evaluation 2 (APACHE II) score at admission ranged from 4 to 17 [mean, 12.5±4.8 (SD)]. Empirical broad-spectrum intravenous antibiotics were given to all the patients, while one was prescribed oseltamivir prior to the confirmation of viral pneumonia in our study. Four patients received adjuvant intravenous immunoglobulin for 3–5 days, while five received corticosteroids for 3–7 days, including hydrocortisone or methylprednisolone. Six patients required invasive mechanical ventilation with a duration range from 8 to 23 days and a median of 11.5 days. The sole death was a 52-year-old man with three comorbidities, who died after 23 days of mechanical ventilation.
Imaging techniques
Chest radiographs were performed using computed radiography or portable computed radiography at bedside, both of which were performed in the anteroposterior projection. Some of the initial CT scans were performed in different institutions, where different types of scanners and protocols were used. The technical parameters included 0.625, 0.75 or 1 mm collimation at 2–7 mm section thicknesses. All CT scans were performed at the end of a deep inhalation with the patient in the supine position, without contrast administration with the exception of one patient. Images were photographed on both lung (window width 1,200–1,600 HU; level −500 to −700 HU) and mediastinal (window width 350–450 HU; level 20–40 HU) settings.
Image analysis
Two chest radiologists (with 15 and 8 years of experience in thoracic imaging, respectively) interpreted the images obtained before and after admission independently, with the decisions made by consensus in case of disagreement. They were blinded to the clinical information of the patients. Each lung was divided into upper and lower zones on radiographs for purposes of analysis. Abnormalities were considered to be distributed to the upper lung zone when they were located above the hilum, to the lower lung zone when they were located under the hilum, or random when both zones were involved. Criteria for chest radiographic and CT findings findings were preferentially defined according to the “Fleischner Society’s Glossary of Terms” (12).
Results
Chest radiographic findings
Seven of the eight initial chest radiographs, which were obtained at 1–9 days [mean, 4.8±2.5 (SD) days] after the onset of illness, showed abnormal findings. The abnormal findings were unilateral (n=4) or bilateral (n=3), including consolidation (n=4), dense patchy infiltration (n=3) (Figure 1A), ground-glass opacity (GGO) (n=1) and pleural effusion (n=1) (Table 1). No reticular opacities were found on chest radiography. The areas of parenchymal abnormalities involved mainly the lower lung zones in four patients, the upper lung zones in one patient, and random in two patients. One patient had a normal initial chest radiograph, though corresponding CT images showed an obvious right lower lung focal consolidation.
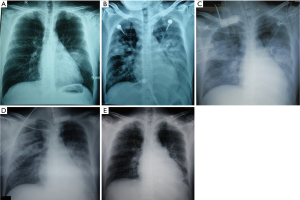
Seven patients had follow-up chest radiographs, the time interval between initial chest radiographs and the first follow-up chest radiographs ranged from 1–5 days with a median of 3 days [mean, 3.2±1.6 (SD) days]. The first follow-up radiographs in five of seven patients showed a remarkable increase in both opacity and size of the abnormalities with more extensive bilateral consolidation compared with the initial chest radiographs (Figure 1B). Most follow-up chest radiographs showed a rapid progression of lesions within ten days of illness onset (Figure 1C), and then consolidation began to dissipate at about two weeks after illness onset (Figure 1D,E).
CT findings
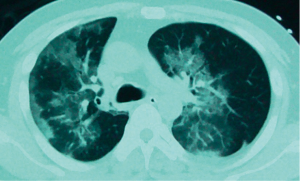
The initial CT scans of all eight patients showed abnormalities. The initial CT scans were performed at 2–13 days [mean, 6.0±3.4 (SD) days] after illness onset. The abnormal findings of initial CT scans were unilateral (n=5) or bilateral (n=3), including consolidation (n=8), GGO (n=2) (Figure 2) and small nodules (n=1) (Table 1). Pleural effusion was found in three patients, being bilateral in one out of three. On the initial CT scans, focal consolidation (Figures 3A) were the main findings in six patients whose initial CT scans were examined within one week after illness onset. Predominant bilateral consolidation with adjacent GGO were seen in one patient’s initial CT which was performed seven days after illness onset. Predominant GGO with adjacent consolidation were observed in one patient whose initial CT was examined nine days after illness onset (Figure 2).

All eight patients had follow-up chest CT images. The time interval between initial CT and the first follow-up CT ranged from 2 to 7 days with a median of 6 days [mean, 5.4±1.6 (SD) days]. The first follow-up CT scans which were performed within ten days of illness onset showed rapid progression of the abnormalities (Figure 3B); including, more extensive unilateral (1 of 6) or bilateral (5 of 6) areas of consolidation, and unilateral (2 of 6) or bilateral (4 of 6) pleural effusion. Follow-up CT scans of most patients had mixed patterns showing predominant consolidation with adjacent GGO. Numbers of lung lobe involved by consolidation in all eight patients were significantly more at the time of maximum severity (2 to 5, mean 4 lobes) than at the presentation (1 to 4, mean 1.7 lobes). In accordance with chest radiographs, CT parenchymal abnormalities began to absorb at about two weeks of illness onset (Figure 3C,D). The serial CT findings showed no appearances of fibrosis such as reticular opacities, cystic lesions, or traction bronchiectasis.
Discussion
Adenovirus, which is the cause of up to 3% of CAP, may be an under recognized cause of fatal pneumonia (13). More than 60% of adenovirus pneumonia patients need intubation and mechanical ventilation (2). Chest imaging has an important role in determining a presumptive etiology and severity assessment of CAP for clinical decision making. The most common initial radiological findings of our study population were focal consolidation, which are also commonly seen with bacterial pneumonia.
In accordance with the reputation of respiratory viruses for causing diffuse interstitial infiltrates, Clark reported widespread bilateral interstitial shadowing as the most common abnormality in 22 immunocompetent adults of adenovirus pneumonia (2). Patchy GGO with or without consolidation on CT were found most frequently both in immunocompromised and immunocompetent patients (4). Histopathologically, GGO reflects acute diffuse alveolar damage including hyaline membrane formation, interstitial lymphocyte infiltration, type II cell hyperplasia, and intraalveolar hemorrhage, edema and fibrin deposits (14). In our study, GGO as a subsidiary manifestation were also frequently observed in the progressive stages of severe adenovirus pneumonia, with only one in eight patients showed predominant GGO with adjacent consolidation.
To date, adenovirus is the only virus known to cause focal or lobar consolidation (the typical manifestation of bacterial pneumonia) as its main imaging abnormality (15-17). Besides grossly congested and consolidated lungs, pathologic data revealed nuclear inclusions and smudge cells, typical histologic evidence of adenovirus pneumonia associated with necrotizing bronchiolitis and bronchopneumonia (6). However, similar to our study, bilateral infiltrates or consolidation were also reported frequently in adenovirus pneumonia (3,7,10,18). As Gu concluded, single lobar or segmental consolidation was more common in patients without ARDS, while patients with ARDS had bilateral and multilobar lung consolidation (11). In our study, focal consolidation were the primary findings within one week after illness onset, which indicates that multilobar involvement rapidly derived from single lobe infiltrates in severe adenovirus pneumonia with ARDS. Consistent with Sun’s findings (10), delay from onset to a single lobar consolidation, then to bilateral multilobar lung infiltrates were about one week in our study, when significant deterioration was observed.
Parenchymal abnormalities of adenovirus pneumonia began to absorb at about two weeks after illness onset, which is about one week sooner than other viral pneumonia such as avian influenza virus infection (19). Focal consolidation on the initial CT scans in our study did not show any preference for specific segments of lung, though Hwang et al. (9) summarized that the right lower lobe was the specific zone of lung infiltration in adenovirus pneumonia. Pleural effusion was thought to be less commonly noted in adenovirus pneumonia (2,3). However 75% of patients in our study and 66.7% of ARDS patients in Gu’s research (11) had pleural effusion, which might be related to more serious illness (8). Appearances of fibrosis which are common in other viral pneumonia were not observed in the present study, which may be another imaging characteristic that resembles bacterial pneumonia.
Our study has several limitations. First, it is retrospective and the scanning intervals for patients were dissimilar. Second, there were a relatively small number of cases owing to the absence of viral screening in non-serious cases combined with the relatively low incidence of adenovirus pneumonia. All the patients in our study had ARDS and no patients with mild clinical disease were included. Third, the relationship between CT and histopathology could not be evaluated since no lung tissue biopsies were available. Finally, technical parameters in image acquisition were not identical, since some initial chest radiographs and CT scans were performed at other institutions prior to a patient being transferred.
In conclusion, severe adenovirus CAP in immunocompetent adults manifests usually as focal consolidation followed by a rapid progression to bilateral consolidation, usually accompanied by adjacent GGO and pleural effusion. The initial image findings and clinical presentation of adenovirus pneumonia may resemble bacterial pneumonia. Screening of adenovirus should be considered in cases of severe pneumonia.
Acknowledgements
Funding: This work was supported by China’s Ministry of Health (Special scientific research funds for health industry 201302017), Ministry of Education (Funds for Innovation team), and Peking Union Medical College Hospital (Project of preeminent youth fund).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Lynch JP 3rd, Fishbein M, Echavarria M. Adenovirus. Semin Respir Crit Care Med 2011;32:494-511. [Crossref] [PubMed]
- Clark TW, Fleet DH, Wiselka MJ. Severe community-acquired adenovirus pneumonia in an immunocompetent 44-year-old woman: a case report and review of the literature. J Med Case Rep 2011;5:259. [Crossref] [PubMed]
- Lewis PF, Schmidt MA, Lu X, et al. A community-based outbreak of severe respiratory illness caused by human adenovirus serotype 14. J Infect Dis 2009;199:1427-34. [Crossref] [PubMed]
- Chong S, Lee KS, Kim TS, et al. Adenovirus pneumonia in adults: radiographic and high-resolution CT findings in five patients. AJR Am J Roentgenol 2006;186:1288-93. [Crossref] [PubMed]
- Motallebi M, Mukunda BN, Ravakhah K. Adenoviral bronchopneumonia in an immunocompetent adult: computed tomography and pathologic correlations. Am J Med Sci 2003;325:285-7. [Crossref] [PubMed]
- Barker JH, Luby JP, Sean Dalley A, et al. Fatal type 3 adenoviral pneumonia in immunocompetent adult identical twins. Clin Infect Dis 2003;37:e142-6. [Crossref] [PubMed]
- Hakim FA, Tleyjeh IM. Severe adenovirus pneumonia in immunocompetent adults: a case report and review of the literature. Eur J Clin Microbiol Infect Dis 2008;27:153-8. [Crossref] [PubMed]
- Klinger JR, Sanchez MP, Curtin LA, et al. Multiple cases of life-threatening adenovirus pneumonia in a mental health care center. Am J Respir Crit Care Med 1998;157:645-9. [Crossref] [PubMed]
- Hwang SM, Park DE, Yang YI, et al. Outbreak of febrile respiratory illness caused by adenovirus at a South Korean military training facility: clinical and radiological characteristics of adenovirus pneumonia. Jpn J Infect Dis 2013;66:359-65. [Crossref] [PubMed]
- Sun B, He H, Wang Z, et al. Emergent severe acute respiratory distress syndrome caused by adenovirus type 55 in immunocompetent adults in 2013: a prospective observational study. Crit Care 2014;18:456. [Crossref] [PubMed]
- Gu L, Liu Z, Li X, et al. Severe community-acquired pneumonia caused by adenovirus type 11 in immunocompetent adults in Beijing. J Clin Virol 2012;54:295-301. [Crossref] [PubMed]
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology 2008;246:697-722. [Crossref] [PubMed]
- Johansson N, Kalin M, Tiveljung-Lindell A, et al. Etiology of community-acquired pneumonia: increased microbiological yield with new diagnostic methods. Clin Infect Dis 2010;50:202-9. [Crossref] [PubMed]
- Kim EA, Lee KS, Primack SL, et al. Viral pneumonias in adults: radiologic and pathologic findings. Radiographics 2002;22:S137-49. [Crossref] [PubMed]
- Cunha BA. Severe adenovirus community-acquired pneumonia mimicking Legionella. Eur J Clin Microbiol Infect Dis 2009;28:313-5. [Crossref] [PubMed]
- Komshian SV, Chandrasekar PH, Levine DP. Adenovirus pneumonia in healthy adults. Heart Lung 1987;16:146-50. [PubMed]
- Lee YJ, Palomino-Guilen P, Babady NE, et al. Disseminated adenovirus infection in cancer patients presenting with focal pulmonary consolidation. J Clin Microbiol 2014;52:350-3. [Crossref] [PubMed]
- Cao B, Huang GH, Pu ZH, et al. Emergence of community-acquired adenovirus type 55 as a cause of community-onset pneumonia. Chest 2014;145:79-86. [Crossref] [PubMed]
- Dai J, Zhou X, Dong D, et al. Human infection with a novel avian-origin influenza A (H7N9) virus: serial chest radiographic and CT findings. Chin Med J (Engl) 2014;127:2206-11. [PubMed]


