Evaluation of pulmonary nodules in Asian population
Introduction
A solitary pulmonary nodule (SPN) is defined as rounded radiological opacity in the lungs measuring up to 3 cm in diameter, fully surrounded by pulmonary parenchyma, in the absence of atelectasis, mediastinal lymphadenopathy or pleural effusion (1). Pulmonary nodules are relatively common, and lung cancer screening trials in patients with high risk for lung cancer have reported prevalence of SPN of 8–51%, with malignancy found in 1.1–12% (1,2). In clinical practice, a pulmonary nodule may be detected incidentally on chest radiograph (CXR) or computed tomographic (CT) scan ordered for other reasons (3). With the increasing use of CT imaging, it is likely that the number of pulmonary nodules detected will continue to rise, with the physician’s primary concern of excluding malignancy. However, some of these SPN detected may be benign such as healed granulomas, active granulomatous infections such as tuberculosis (TB), hamartomas, and arterio-venous malformation (4) out of which TB is the most common disease presenting as nodule in the Asian population.
American College of Chest Physicians (ACCP) provides guideline based algorithm to manage pulmonary nodules (5). Are these algorithms relevant to Asian population, and can we, or do we manage our patients according to these algorithms? There is little data in our local population regarding presentation and management of pulmonary nodules in lung cancer patients. Our study describes the current pattern of clinical practice in the management of pulmonary nodules in Asian country (Singapore), highlighting the strengths and limitations of using ACCP guidelines in managing them.
Methods
The medical records for all patients diagnosed with lung cancer in 2010 were reviewed. Out of these, all patients presenting with a single SPN or multiple pulmonary nodules (MPN) were selected. A single SPN was defined as radiological opacity measuring up to 3 cm in diameter, fully surrounded by pulmonary parenchyma, in the absence of atelectasis, mediastinal lymphadenopathy or pleural effusion (1,6,7). MPN was defined as a dominant nodule with 1 or 2 more nodules as per the ACCP guidelines (5). All patients presenting with lung masses as defined as >3 cm in diameter were excluded. The study protocol was approved by the NHG Domain Specific Review Board (2015/00932) and was conducted in accordance with the Helsinki Declaration of 1964 (revised 2008). Due to retrospective nature of the study, the consent was waived.
Clinical data including age, gender, symptoms at presentation, and smoking history were collected. Characteristics of the pulmonary nodules including type, shape, size, location and number of nodules were recorded. Histological information, staging investigations, presence of distant metastasis, treatment and survival were also recorded.
We also assessed validity of a tool recommended by ACCP for estimating the probability of malignancy in pulmonary nodules retrospectively as described below (5):
Data analysis
We used software (SPSS, version 17; SPSS, Chicago, Ill, USA) for all statistical analyses. Where applicable, the results were compared using a Wilcoxon two-sample test or Fisher exact test. P values were two sided and considered indicative of a significant difference if less than 0.05.
Results
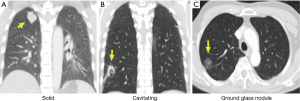
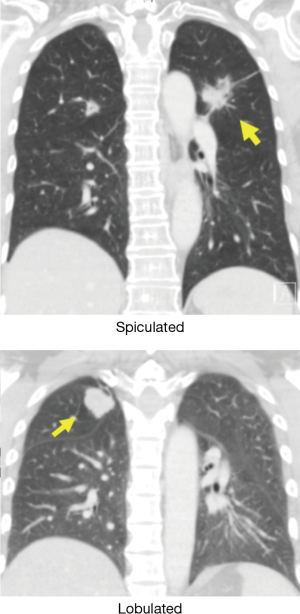
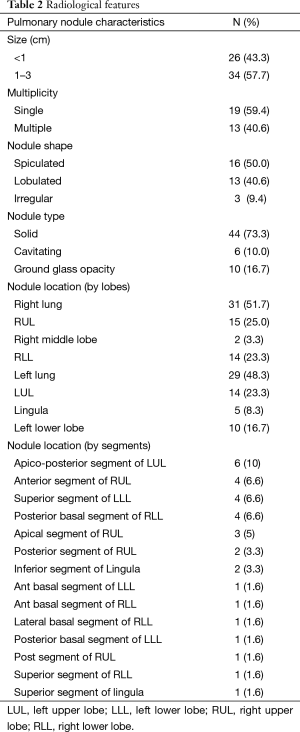
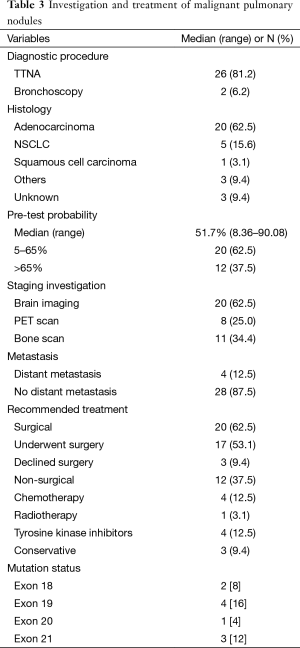
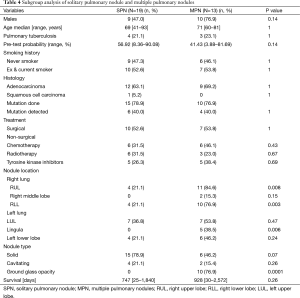
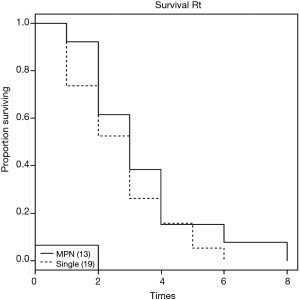
In the absence of any local lung cancer screening program, TB contact screening and pre-employment or employment renewal screening CXR were the most common ways how the malignant nodules were detected incidentally in Singapore. Other ways were the radiograph done to evaluate respiratory symptoms, CT coronary angiogram done to evaluate cardiovascular symptoms, and CT abdomen or CT urogram done to evaluate abdominal and urological symptoms respectively (Table 1). Sixty nodules were detected in 32 patients. Such nodules commonly presented with the size of 1–3 cm in diameter than being sub-centimetre. These nodules were either solid, cavitating, or ground glass in appearance (Figure 1). With regards to location, nodules were more common in the right lung, with right upper lobe (RUL), left upper lobe (LUL) and right lower lobe (RLL) being frequently involved lobes and apico-posterior segment of LUL & apical and anterior segment of RUL being the most frequently involved segments (Table 2, Figure 2). Probability calculator was not used in the management of our patients. However, retrospective analysis revealed that the calculator assigned low/moderate risk of malignancy in 62.5% of patients and high risk in 32.5% of patients. About 37.5% of patients were never smokers in our cohort. Most common histological type of cancer found in such nodules was adenocarcinoma and staging was done by the combination of bone scan and brain scan instead of positron emission tomogram (PET) scan (Table 3). Lung cancer was surgically resectable in 63% of patients. Median survival until death or last follow up was 2.1 years. One year survival was 80% and 5-year survival was 10% with no difference between patients who presented with single or multiple nodules (Table 4, Figure 3).

Full table
Full table
Full table
Full table
Discussion
The clinical practice of managing pulmonary nodules in Asian population differed from the ACCP guidelines. None of the patient had pre-test probability calculated and few had PET scan, the likely reason being endemicity of granulomatous diseases like TB.
In the assessment of a pulmonary nodule, ACCP recommends the calculation of pre-test probability of malignancy (5). However, its performance in the Asian population remains to be determined. This equation or calculator uses “smoking” as one of the predictors of malignancy (5). However, in the current study 37.5% of patients were never smokers. A total of 31.6% of lung cancer patients and 54.5% of lung cancer patients with adenocarcinoma histology have been reported as never smokers in Singapore (8). This data is consistent with other studies in Asian population (9-11). Hence, in one third of the lung cancer patients and half of the adenocarcinoma lung patients, keying in “0” for smoking in the calculator, this predictor may falsely diminish contribution toward predicting malignancy.
Another predictor in the recommended probability calculator is the “upper lobe location” of the nodule (5). In our cohort, nodules were located more frequently in the upper lobes. However TB is also known to affects upper lobes more commonly than the lower lobes (12). This has implications. For example, both the number of new cases, and the age at onset of pulmonary TB and lung cancer are similar in Singapore. New cases of pulmonary TB between year 2009 and 2012 being 937, 951, 977, and 1,206 per year (13), and average number of lung cancer cases being 1,311 patients per year, in the same period (14). Similarly, age at onset for both the diseases is 5th decade onward. Hence, this predictor of “upper lobe location” may falsely contribute toward predicting malignancy in a benign nodule from TB in Asian population.
Another challenge this near equal incidence of the two diseases pose is that, TB being historically a well-known disease, the similarities in the incidence rates, similar clinical presentation such as weight loss, cough etc., and similar radiological features of the involvement of the upper lobes, a primary physician is often led to suspect pulmonary TB first in a patient with a pulmonary nodule, often leading to misdiagnosis of lung cancer as TB. Correspondingly, reporting radiologist mentioning suspicion of TB in their report may also contribute to misdiagnosis since in clinical practice, physicians rely on the “report of the radiograph” more than the radiograph itself. Singh et al. reported 14 out of 70 lung cancer patients being misdiagnosed and treated inappropriately for TB for a mean duration of approximately 4 months (15). In our own audit of a cohort of 54 patients who were referred to respiratory physician for CXR abnormality and were eventually diagnosed with lung cancer, the diagnosis of lung cancer was missed or delayed in 24/54 (44.4%) patients as they were initially suspected to have TB based on the radiograph. Never smoking patients with nodular changes on the CXR, and CXR reported as infective changes suspicious of TB by the reporting radiologists, correlated with the occurrences of misdiagnosis (unpublished data). Since it may take two months (waiting time of TB culture) before TB is confirmed or excluded, this misdiagnosis leads to delay in diagnosis and patient dissatisfaction.
With significant proportion of never smokers developing cancer, and significant proportion of patients with upper lobe involvement secondary to TB in a population with the near equal incidence of TB and cancer, it is arguable that the probability calculator may give inaccurate result in Asian population and assign disproportionately high risk of malignancy to benign nodules.
Following calculation of the pre-test probability, ACCP guidelines recommend PET scan prior to non-surgical or surgical biopsy for nodules between 8–30 mm (5). This was not practiced in our population. The PET was done in only 25% of patients in our cohort with the intent of staging after the histological diagnosis and it was “true negative” for distant metastasis in all 8 patients. This illustrates that physicians only trusted the PET scan for staging and not for diagnosis. The near equal incidence of TB and lung cancer in Asian population poses challenge to the recommendation of PET scan. In Western countries like USA, the incidence of infective granulomatous diseases such as TB and histoplasmosis, which can lead to confounding radiological changes that mimic malignant nodules is 3.7 in 100,000 and 1 in 100,000 respectively (16,17). Whereas in Singapore, the incidence of TB is 30.7 per 100,000—10 times that of USA (13,18). This pattern increases the chance of false positive PET scan in local population, making the pre-test probability of a positive PET scan being equally from infective and malignant pathology, rendering it less relevant in Asian population.
Regarding screening, although low dose CT screening has shown mortality benefit, however one of the inclusion criteria for lung cancer screening based on National Lung Cancer Screening Trial (NSLT) is smoking history (19). Eligible patients are those between 55 and 74 years of age with a history of cigarette smoking of at least 30 pack-years, and, if former smokers, had quit within the previous 15 years (19). Since 30–40% of Asian population with lung cancer is never-smoker, it makes approximately one third of the lung cancer patients ineligible for screening, limiting the adoption of lung cancer screening to local population (20).
Regarding survival, the reported expected 5-year survival of stage-I disease is 65% (21). However, the 5-year survival in our cohort was 10%. Our local data on 5-year survival in stage-I shows the survival in lung cancer to be 50%. The lower 5-year survival in our cohort can be explained by the 4 (12.5%) patients having metastatic disease, 3 (9.4%) patients refusing surgery despite being resectable, 2 (6.2%) patients underwent surgery but were upstaged at the time of surgery, and 1 (3.1%) patient lung cancer progressed despite surgery. However, there was no difference in survival between our patients presenting with SPN or MPN. They had similar demographics, histology and treatment received. Therefore patient presenting with MPN should not be presumed metastatic and denied appropriate treatment, as survival is equal in comparison to a SPN. This is consistent with recommendations from British Thoracic Society and ACCP that coexistent nodules in patients with lung cancer should not be presumed to be malignant, especially in patients who otherwise would be a surgical candidate (5,22).
In addition to ACCP guidelines, other societies that provide guidelines on the management of pulmonary nodules are Fleischner Society and British Thoracic Society. Fleischner Society, similar to ACCP guidelines, emphasizes distinguishing the patients into high risk and low risk groups based on “smoking” history. No follow-up in ≤4 mm, follow up CT at 1 year in >4–6 mm, or 6 months in >6–8 mm sub-centimetres nodules is recommended for never smokers (6). Similarly, British Thoracic Society stresses on emphysema as one of the items in their proposed prediction model (22). The appropriateness of these criteria is debatable in the context of 50% of patients with adenocarcinoma (most prevalent sub-type of lung cancer) being never smokers in Asian population.
Our study has following limitations. First, it is a retrospective study limited by unavailability of information on certain variables, with small population enrolled. Second, we only included patients with malignant nodules. This may have introduced selection bias. The inclusion of non-malignant nodules would have provided greater information on the false positivity or false negativity and the accuracy of the PET scan and CT scan. However, our aim was to study the pattern of local practice in the management of lung nodules that turn out to be malignant.
Recommendation
We propose the following algorithm when evaluating a patient with pulmonary nodule in our local population. In all patients with a newly discovered pulmonary nodule and no old CXR s or CT scans for comparison, an early CT is recommended for accurate characterization of the nodule’s size, shape, type, and determination of the best diagnostic approach. Two specimens of sputum should also be sent for AFB smear and TB culture at the first visit to exclude pulmonary TB. Pre-test probability of malignancy should be estimated after the CT scan with a validated tool. All patients with moderate or high probability for malignancy should be offered further investigation to obtain histological “and” microbiological samples. Appropriate diagnostic investigation would depend on findings of CT with transbronchial lung biopsy with or without radial probe endobronchial ultrasound preferred for nodules >1 cm with bronchus sign, and transthoracic needle aspiration or assistive technologies like navigation bronchoscopy preferred for peripherally located nodules <2 cm with no bronchus sign, and wedge resection or surveillance CT scans for nodules <1 cm in diameter. With every diagnostic method, specimen should be sent both for AFB smear and TB culture along with histological examination.
In conclusion, the clinical practice of managing pulmonary nodules in Asian countries may differ from the ACCP guidelines. This is most likely secondary to the local endemicity of TB and its confounding effect on radiological findings of CT scan and PET scan. Factors like being responsible for most number of cancer related deaths in Singapore, reasonably high incidence of ~1,500 patients per year, presentation in late stage, and lack of modifiable factors to prevent cancer, demand an aggressive approach to management of lung cancer. This can be achieved by formation of, first, locally relevant screening programs and guidelines that include never smokers, and incorporate epidemiological patterns of granulomatous infections. Second, developing clinical management protocols to ensure adequacy of tissue to allow mutation analysis for each patient, and promoting increased and easy adoption of mutation analysis due to higher prevalence of EGFR sensitive population in Asia. Third, encourage research to identify methods to distinguish between benign and malignant nodules non-invasively i.e., radiologically with precision.
Acknowledgements
Authors would like to thank Ms. Ivy (Lingling Yu) for her valuable contribution in editing the figures and administrative work.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Wahidi MM, Govert JA, Goudar RK, et al. Evidence for the treatment of patients with pulmonary nodules: when is it lung cancer?: ACCP evidence-based clinical practice guidelines (2nd edition). Chest 2007;132:94S-107S.
- McWilliams A, Tammemagi MC, Mayo JR, et al. Probability of cancer in pulmonary nodules detected on first screening CT. N Engl J Med 2013;369:910-9. [Crossref] [PubMed]
- Edey AJ, Hansell DM. Incidentally detected small pulmonary nodules on CT. Clin Radiol 2009;64:872-84. [Crossref] [PubMed]
- Murrmann GB, van Vollenhoven FH, Moodley L. Approach to a solid solitary pulmonary nodule in two different settings-"Common is common, rare is rare J Thorac Dis 2014;6:237-48. [PubMed]
- Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e93S-120S.
- MacMahon H, Austin JH, Gamsu G, et al. Guidelines for management of small pulmonary nodules detected on CT scans: a statement from the Fleischner Society. Radiology 2005;237:395-400. [Crossref] [PubMed]
- Naidich DP, Bankier AA, MacMahon H, et al. Recommendations for the management of subsolid pulmonary nodules detected at CT: a statement from the Fleischner Society. Radiology 2013;266:304-17. [Crossref] [PubMed]
- Verma A, Lim AY, Tai DY, et al. Timeliness of diagnosing lung cancer: number of procedures and time needed to establish diagnosis: being right the first time. Medicine (Baltimore) 2015;94:e1216. [Crossref] [PubMed]
- Sun S, Schiller JH, Gazdar AF. Lung cancer in never smokers--a different disease. Nat Rev Cancer 2007;7:778-90. [Crossref] [PubMed]
- Subramanian J, Govindan R. Lung cancer in 'Never-smokers': a unique entity. Oncology (Williston Park) 2010;24:29-35. [PubMed]
- Santoro IL, Ramos RP, Franceschini J, et al. Non-small cell lung cancer in never smokers: a clinical entity to be identified. Clinics (Sao Paulo) 2011;66:1873-7. [Crossref] [PubMed]
- Nemec SF, Bankier AA, Eisenberg RL. Upper lobe-predominant diseases of the lung. AJR Am J Roentgenol 2013;200:W222-37. [Crossref] [PubMed]
- World Health Organization. Tuberculosis. Available online: http://www.who.int/tb/country/data/download/en/
- Health Promotion Board, National registry of disease office. Interim Annual registry report Trends in Cancer incidence in Singapore 2009-2013. Available online: www.nrdo.gov.sg
- Singh VK, Chandra S, Kumar S, et al. A common medical error: lung cancer misdiagnosed as sputum negative tuberculosis. Asian Pac J Cancer Prev 2009;10:335-8. [PubMed]
- CDC Fact sheet. Trends in tuberculosis. Available online: http://www.cdc.gov/tb/publications/factsheets/statistics/trends2013.pdf
- Baddley JW, Winthrop KL, Patkar NM, et al. Geographic distribution of endemic fungal infections among older persons, United States. Emerging Infectious Diseases 2011;17:1664-9. [Crossref] [PubMed]
- Tuberculosis. Available online: https://www.moh.gov.sg/content/moh_web/home/diseases_and_conditions/t/tuberculosis.html
- National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011;365:395-409. [Crossref] [PubMed]
- Ebbert JO, Yang P, Vachon CM, et al. Lung cancer risk reduction after smoking cessation: observations from a prospective cohort of women. J Clin Oncol 2003;21:921-6. [Crossref] [PubMed]
- Rena O, Oliaro A, Cavallo A, et al. Stage I non-small cell lung carcinoma: really an early stage? Eur J Cardiothorac Surg 2002;21:514-9. [Crossref] [PubMed]
- Callister ME, Baldwin DR, Akram AR, et al. British Thoracic Society guidelines for the investigation and management of pulmonary nodules. Thorax 2015;70 Suppl 2:ii1-54. [Crossref] [PubMed]