A new LigaSure technique for the formation of segmental plane by intravenous indocyanine green fluorescence during thoracoscopic anatomical segmentectomy
Introduction
The frequent detection of ground grass opacity (GGO) and small pulmonary metastases on multi detector-row computed tomography (MDCT) has increased the need for thoracic surgeons to perform resection therapy. Thoracoscopic surgery (TS) is valuable for diagnostic and therapeutic resection because percutaneous CT-guided biopsy or transbronchial biopsy of such peripheral lesions may be difficult. In addition, several authors have reported good outcomes even for sublobar resection of such malignancies (1-3). The absence of randomized controlled trials is the rationale behind the indications for such surgical technique, which has also generated a number of retrospective studies. Segmentectomy, rather than wedge resection, could sometimes secure an adequate margin. However, many thoracic surgeons perform thoracotomy more often than TS. During TS, it is important to proceed with the operation with a clear anatomical understanding (4,5).
In order to solve this problem, we used LigaSure under indocyanine green (ICG) fluorescence system to delineate the anatomical segmental plane during TS. The LigaSure is a technique that has been widely used in pulmonary resection (6); the purpose of this study was to assess its efficacy and safety for segmental plane formation.
Methods
This was a study on consecutive patients who underwent anatomical segmentectomy using ICG fluorescence system (Karl Storz, Tuttlingen, Germany) at Aichi Cancer Center Hospital between June 2013 and December 2014. Total 60 consecutive various bilateral segmentectomies using this device were performed or supervised by more than two senior consultants who have the experience of doing more than 200 procedures (Table 1). In all cases, informed consent for the procedure was obtained. ICG was intravenously administered at 2 mL/body ICG (one fifth of the usual amount in hepatic function test). Patients were scheduled for outpatient control after 3 to 6 months and postoperative follow-up for complications was conducted at a median of 10 months (range, 3–18 months). For postoperative air leak, the term “minor” was defined as no requirement for drainage; the period after removal of tube drainage was defined as “early” when it was performed prior to hospital discharge and as “delayed” when it was performed on outpatient follow-up. The ethics on our institution approved; informed consent was obtained.
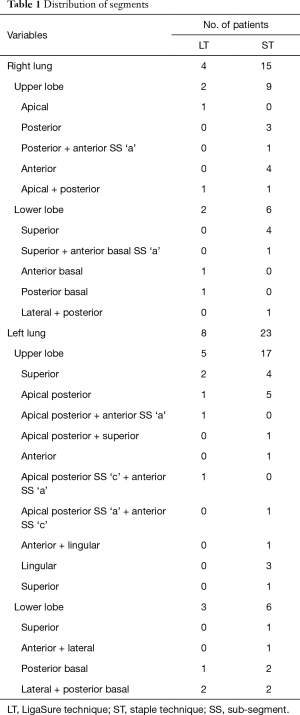
Full table
A new LigaSure technique (LT)
We performed segmentectomy during TS, as well as lobectomy, using four definite ports. The patients were placed on a lateral recumbent position after general anesthesia. The surgeon’s left hand 7-mm port was inserted in the 3nd/4th (for tumor locations in the upper/lower lobe) intercostal space before the scapula, whereas the right hand 20-mm port was inserted in the 5th/6th intercostal space at the post-axillary line. The first assistant manipulated the thoracoscopy instrument through an 11-mm port to establish placement in the 3rd/4th intercostal space before the breast. The role of the second assistant was to take a new port into a 15-mm port to establish placement in the 5th/6th intercostals at the anterior axillary line. We conducted a complete 4-port thoracoscopic anatomical segmentectomy for the purpose of obtaining combined minimal damage to the respiratory muscles and preservation of lung parenchyma.
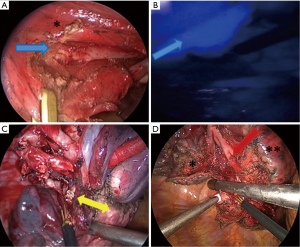
While making the segmental plane, we devised LT the following aspects: (I) extraction of intersegmental veins as the landmarks (Figure 1A); (II) identification of consistency of intersegmental plane and the boundary area using ICG (Figure1B); and (III) to accomplish the formation of segmental plane by using LigaSure (Covidien, Mansfield, MA, USA) for the periphery and Stapler by Powered Echelon Flex (Ethicon Endo-Surgery, Cincinnati, Ohio, USA) for central lung parenchyma instead of the conventional staple technique (ST) (Figure 1C,D).
Predictive score: index of prolonged air leak (IPAL)
We used the method to evaluate predictive probability of postoperative prolonged air leak (PAL) (more than 7 days) according to a previously reported formula to calculate IPAL score. This analysis was calculated as follows: gender (F =0; M =4) − (body mass index−24) + 2 × dyspnea score + pleural adhesion (No =0; Yes =4) + pulmonary resection (segmentectomy =7) + location (lower and middle lobe =0; upper =4) (7).
Statistical analyses
All data were analysed using SPSS version 17.0 (SPSS Institute Incorporated, Chicago, Illinois, USA). Differences between the two groups were calculated using the Mann-Whitney U test, and a value of P<0.05 was considered to indicate statistical significance.
Results
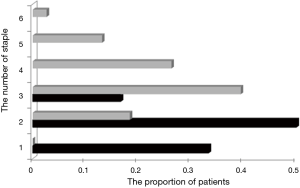
Patient characteristics are summarized in Table 2. The median age was 67 years (range, 37–85 years); 23 were males and 15 were females. During TS and thoracotomy, the median time spent for the formation of the segmental plane with LT was 22.8 min (range, 11–46 min), whereas that for ST was 16.2 min (range, 6–33 min). The border significant difference was found (P=0.05). The procedure of LT that lasted for 46 min included surgery for double sub-segments combined with resection of “left sub-segment S1 + 2a + S3a”. The second longest surgery time was 38 min for the right segment 9+10 anatomical segmentectomy; this included visualization of the boundary line by ICG fluorescence system, marking by electric scalpel, the surgical procedure of LT, and the removal of instruments. The median number of staples for the formation of the segmental plane with LT was 1.8 (range, 1–3), whereas that for the conventional ST was 3.4 (range, 2–6) (Figure 2); this was found to be significantly different (P<0.01). LT provides the miniaturization by the perspective hindrance during TS as possible and the reduction of the performing cost. In particular, in the patient who underwent the formation of two segmental lumens (8), the number of staples was also significantly less in LT than in ST (P<0.01).
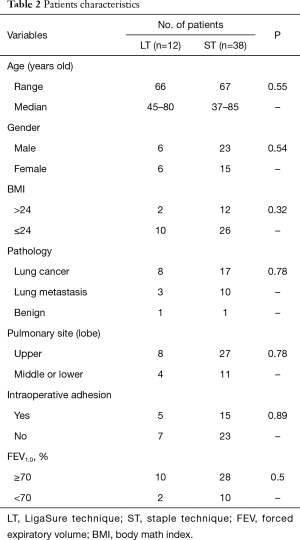
Full table
All patients who underwent LT were able to resume oral intake the following operative day. The average period required for the removal of drainage tube was 1.7 days and patients were discharged after an average of 8.5 days. No patients had PAL. Two patients had early minor air leak at an average of 1.5 operative days after the removal of the drain; these patients were low/middle risk by IPAL score (Table 3). The proportion of patients with moderate risk IPAL score was 16.7% (1/6); there was no air leak in the patients with high risk IPAL score. On the other hand, the average period required for the removal of drainage tube was 1.7 days (P=0.95); the average discharge period of patients receiving staple only was 6.5 days (P=0.04). PAL was diagnosed in 1/38 (2.6%) and delayed minor air leak was seen in 1/38 (2.6%) (Table 3). The patient with PAL was classified in the high risk group by IPAL score and the proportion was 4.5% (1/22). The complication in LT showed a tendency to higher as compared to ST in the patients with low/middle IPAL score, although a significant difference was not obtained (P=0.06). Comparing the two methods, a significant difference was found in terms of the incidence of early air leak (P>0.01) but not in those of PAL (P=0.54) and delayed air leak (P=0.38).
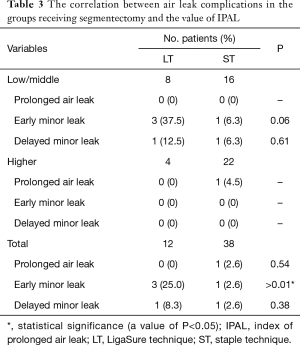
Full table
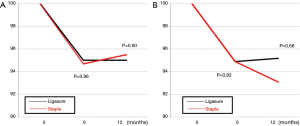
Postoperative half- and 1-year comparative remaining vital capacity (VC) in LT were 95.0% and 95.0%, whereas those in ST were 94.7% and 95.5%, respectively. No significant difference was obtained as for any time (Figure 3A). Postoperative half- and 1-year comparative remaining forced expiratory volume in one second (FEV1.0) in LT were 94.9% and 95.2%, whereas those in ST were 94.9% and 93.1%, respectively. No significant difference was also found (Figure 3B).
Discussion
The formation of segmental plane is the most difficult technique during TS anatomical segmentectomy because the exact landmarks deep within the lung parenchyma are difficult to identify and the incidence of parenchymal vessel injury may be high. LigaSure had good outcomes for treatment by lobectomy and sublobar resection (6) and there are a few articles citing its usefulness for lung parenchyma (6,9). Is this new technique overkilled? Santini et al. reported that the electrothermal bipolar sealing system allowed creation of an appropriate plane through the parenchyma close to the pericyst, minimizing the normal lung exposed to resection as much as possible and reducing the resulting bleeding and air leak in the management of lung hydatid disease (9). Cutting into lung parenchyma using bipolar sealing system might provide the freely deformable flexibility of lung parenchyma while uniformly performing heating and capable of being safely used without over cauterization like the use of electric scalpel. Cutting into lung parenchyma using bipolar sealing system might have a crucial role to play. The formation of subsegmental plane using electric scalpel and/or stapler had been the gold standard for many years, but the stapler is difficult to handle through a thoracoscopy port, at most 20 mm in size, in the thoracic cavity; sometimes, the use of staples for the control of the cutting vector requires a 45-mm size cartridge instead of a 60-mm size cartridge. The large diameter of a 60-mm stapler may obstruct visualization during surgery but most surgeons find it beneficial over the formation of a precise anatomical segmental line by electrocautery in reducing prolonged postoperative air leak. Our new technique of LT was not superior to the conventional staple method in terms of faster time and fewer complications; however, the use of LigaSure may improve surgical facilitation of this situation and reduce cost. This new method expands the options in improving the surgical technique but not paramount.
The following comprehensive proposals on possible visualization of the boundary line between segments and/or subsegments using ICG have been published. First, the injection of ICG through the segmental bronchus facilitates easy identification of intersegmental planes by color change in both the lung surface and parenchyma (10). Second, the injection of ICG through the systemic circulation facilitates easy identification of intersegmental planes only on the surface (11). These techniques enable the identification of the boundary line without lung infiltration and provide a stable working section capable of continuing the process of surgery, TS in particular, while ensuring a sufficient working space. However, Waseda et al. described that the latter method was not applicable to complicated segmentectomies because the duration of intravascular ICG visualization was too short and a large amount of ICG was ideally required (12). How much is an appropriate amount of ICG for TS segmentectomy? As shown in Figure 1, it might be suggested that the formation of a segmental plane was enough with one-fifth quantity. Thus, one of the novel features of a new procedure would be in-depth identification of the exact segmental plane of the entire circumference of lung parenchyma along the ICG fluorescence line during LigaSure and repeated use of ICG (Figure 1). To the best of our knowledge, there have been no reports that focused on this issue. To overcome this disadvantage, we considered that it may be helpful to make one end of the ICG fluorescent area match with the segmental vein (Figure 1). Identification of intersegmental veins is strongly demanded in the formation of thoracoscopic anatomical segmentectomy.
Although there are many articles about PAL after pulmonary resection, there are a few publications about minor leak, especially after sublobar resection. Okada et al. reported that various segmentectomies have presented with PAL in about 7.7% patients, whereas minor leak was observed in 3.8% (13). In our study, the incidence of PAL after various segmentectomies was 2.0% and that for minor leak was 8.0%, which supports their report. In contrast, some recent studies about PAL after lobectomy reported an incidence of approximately 6.8–15.6% (8,14,15). This discrepancy may be accounted for by differences in semantics and the process of the two procedures. Accordingly, the use of surgical sealant techniques by buttress material or staple was routinely required to prevent PAL after segmentectomy in our institution, but not always after lobectomy. The incidence of PAL did not differ significantly between the two methods of segmental plane formation. The sealant potential of LigaSure is typically to close vessels and bronchi from 3 up to 7 mm. In the LigaSure group, all lymphatic ducts and blood vessels, including peripheral lung parenchyma without any suture ligation, underwent the complete work of sealing the small chamber of lung parenchyma in a short time, while avoiding bleeding from the parenchymal vessel and to certainly prevent air leakage after bilateral ventilation. In addition, we consider that the use of ICG allows the resected line at a more precise segmental plane, and it is connected for the decrease in postoperative air leak. However, it is noteworthy that the incidence of early minor leakage was significantly higher in the LigaSure group (P=0.01) regardless of IPAL score. We empirically deduce that postoperative PAL was highly caused by the outflow of air through the staple, whereas the cause of minor leak was rhexis of package by LigaSure. In other words, the problem was caused by surgical technique. Moreover, we consider that this new technique of AC might have possibility to reduce the rate of postoperative air leak that is equal to conventional stapler technique by the improvement of our technique and intraoperative careful caution while reducing the number of staplers in spite of the requirement of more time-consuming. Nevertheless, one advantage of this technique was easy manipulation in the direction of the segmental resected line in the remaining lung parenchyma by combining stapler and ICG fluorescence during TS.
This study has several limitations. It has a small sample size and was conducted in a single institutional study. It serves mostly as a report on the availability of a new technique that seems to have some advantages. Second limitation was the less frequent incidence of complications. We have only operated on 12 patients who underwent apple core under ICG fluorescence navigation system; hence, there is still a lot to improve with regards to the learning curve of this technique.
In conclusion, a combination of ICG fluorescence system and LigaSure may be beneficial for patients who need the formation of the segmental plane during TS. Our new technique allows for a safe and feasible procedure and the LigaSure facilitates peripheral lung parenchyma resection by minimizing the number of staples used and air leak complications. We finally construed that the cause of the minor air leak was a technical problem.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Nomori H, Mori T, Ikeda K, et al. Segmentectomy for selected cT1N0M0 non-small cell lung cancer: a prospective study at a single institute. J Thorac Cardiovasc Surg 2012;144:87-93. [Crossref] [PubMed]
- Okada M, Mimae T, Tsutani Y, et al. Segmentectomy versus lobectomy for clinical stage IA lung adenocarcinoma. Ann Cardiothorac Surg 2014;3:153-9. [PubMed]
- Wolf AS, Richards WG, Jaklitsch MT, et al. Lobectomy versus sublobar resection for small (2 cm or less) non-small cell lung cancers. Ann Thorac Surg 2011;92:1819-23; discussion 1824-5.
- Houck WV, Fuller CB, McKenna RJ Jr. Video-assisted thoracic surgeryVideo-assisted thoracic surgery upper lobe trisegmentectomy for early-stage left apical lung cancer. Ann Thorac Surg 2004;78:1858-60. [Crossref] [PubMed]
- Oizumi H, Kato H, Endoh M, et al. Techniques to define segmental anatomy during segmentectomy. Ann Cardiothorac Surg 2014;3:170-5. [PubMed]
- Shigemura N, Akashi A, Nakagiri T, et al. A new tissue-sealing technique using the Ligasure system for nonanatomical pulmonary resection: preliminary results of sutureless and stapleless thoracoscopic surgery. Ann Thorac Surg 2004;77:1415-8; discussion 1419. [Crossref] [PubMed]
- Brunelli A, Varela G, Refai M, et al. A scoring system to predict the risk of prolonged air leak after lobectomy. Ann Thorac Surg 2010;90:204-9. [Crossref] [PubMed]
- Oizumi H, Kanauchi N, Kato H, et al. Anatomic thoracoscopic pulmonary segmentectomy under 3-dimensional multidetector computed tomography simulation: a report of 52 consecutive cases. J Thorac Cardiovasc Surg 2011;141:678-82. [Crossref] [PubMed]
- Santini M, Fiorelli A, Milione R, et al. The use of an electrothermal bipolar tissue sealing system in the management of lung hydatid disease. Interact Cardiovasc Thorac Surg 2014;19:690-2. [Crossref] [PubMed]
- Oh S, Suzuki K, Miyasaka Y, et al. New technique for lung segmentectomy using indocyanine green injection. Ann Thorac Surg 2013;95:2188-90. [Crossref] [PubMed]
- Tarumi S, Misaki N, Kasai Y, et al. Clinical trial of video-assisted thoracoscopic segmentectomy using infrared thoracoscopy with indocyanine green. Eur J Cardiothorac Surg 2014;46:112-5. [Crossref] [PubMed]
- Waseda R, Oda M, Matsumoto I, et al. A novel fluorescence technique for identification of the pulmonary segments by using the photodynamic diagnosis endoscope system: an experimental study in ex vivo porcine lung. J Thorac Cardiovasc Surg 2013;146:222-7. [Crossref] [PubMed]
- Okada M, Mimura T, Ikegaki J, et al. A novel video-assisted anatomic segmentectomy technique: selective segmental inflation via bronchofiberoptic jet followed by cautery cutting. J Thorac Cardiovasc Surg 2007;133:753-8. [Crossref] [PubMed]
- Brunelli A, Monteverde M, Borri A, et al. Predictors of prolonged air leak after pulmonary lobectomy. Ann Thorac Surg 2004;77:1205-10; discussion 1210. [Crossref] [PubMed]
- Rivera C, Bernard A, Falcoz PE, et al. Characterization and prediction of prolonged air leak after pulmonary resection: a nationwide study setting up the index of prolonged air leak. Ann Thorac Surg 2011;92:1062-8; discussion 1068. [Crossref] [PubMed]


